* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download electrons - Northside Middle School
Survey
Document related concepts
Transcript
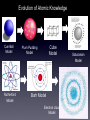
Atomic Structure • • • Elements are made of atoms The word atom is derived from the Greek word atom which means indivisible. The Greeks concluded that matter could be broken down into particles to small to be seen. These particles were called atoms Atoms are composed of three type of particles: protons, neutrons, and electron. Protons and neutrons are responsible for most of the atomic mass. For example: in a 150 person 149 lbs, 15 oz are protons and neutrons while only 1 oz. is electrons. The mass of an electron is very small (9.108 X 10-28 grams). The Structure of Atoms • • • Atoms have 3 components: protons, neutrons, and electrons protons and neutrons are in the nucleus Electrons circle around the nucleus in “electron shells”. Protons, Neutrons, and Electrons • • Protons are found in the nucleus. Protons are positively charged particles. Neutrons are also found in the nucleus. Neutrons are neutral (not electrically charged) charged particles. • Almost all of an atoms mass is found in the nucleus. Electrons don’t really figure into the atomic mass. • Electrons are very light and very small. They are negatively charged and are found in the electron shells surrounding the nucleus Evolution of Atomic Knowledge Cue Ball Model Rutherford Model Cube Model Plum Pudding Model Borh Model Electron cloud Model Saturanian Model • “Pictures” of atoms are often misleading: – Electrons don’t “orbit” the nucleus like planets around the sun. We often draw them that way because it is easier to understand, but they really behave like a cloud around the nucleus, almost like a beehive. Here are some actual “pictures” of atoms: Electron Shells- The Bohr Model • • • • Electrons circle the nucleus at defined positions called shells. The innermost shell of every atom could hold 2 electrons (except hydrogen, which only has 1 electron). The second shell holds 8 electrons, the 3rd holds 18. Electrons fill in from the nucleus out, so the inner shells always fill up first. Electrons fill in from the nucleus out, so the inner shells fill up first.. Build an Atom The outermost electron shell is referred to as the valence shell. Sometimes, atoms do not fill their valence shell all the way. Some spots are empty. Structure of Atoms Elements that have empty spots in their valence shell are reactive, meaning they want to bond with other elements, to try to fill up their empty spots. This is why the alkali metals are sooooo reactive. They all have really empty valence shells. They each only have one electron in their valence shell! Nobel gases are not reactive because they have full valence shells! •All of the elements except the 7 elements considered “noble gases” have empty spots in their valence Structure of Atoms shell. •When elements bond with other elements they form something called a compound. •In a compound, two elements either share their electrons, or one element steals the other elements electrons. Either way, the elements both feel like their valence shell is fuller than it used to be, so they are more stable. •This is why there are so many compounds in our world. Elements form compounds to make a full outer shell. Meet Oxygen

















![Atomic Structure [PowerPoint]](http://s1.studyres.com/store/data/000122096_1-1d100da6540d2f26db122fc51f672fe5-150x150.png)







