* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Lecture 4: Antigen Presentation by T lymphocytes
Survey
Document related concepts
Transcript
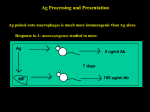
Lecture 4: Antigen Presentation by T lymphocytes Questions to Consider What is the structural basis by which MHC molecules present peptides to the T cell receptor? How are endogenous peptides targeted to MHC Class I molecules and exogenous peptides targeted to MHC Class II molecules? How does the T cell receptor see the peptide and MHC molecule? What is the structural basis for CD4 T cells/MHC Class II and CD8 T cell/MHC Class I restriction? Presentation of Peptide to CD8 or CD4 T Cell by Class I MHC or Class II MHC Molecules, Respectively Number of alleles/locus The Three Loci Encoding MHC Class I (A, B and C) or MHC Class II (DP, DQ or DR) Genes Are Highly Polymorphic Expression of MHC Alleles is Codominant Class I MHC molecules can present a diverse yet limited number of peptides sized 8 – 10 amino acids long. What is the structural basis that limits the peptides that the MHC molecule can present? Structure of MHC Class I Molecule Heterodimer of membrane- spanning -chain and 2-microglobulin The -chain is polymorphic while the 2-microglobulin is the same for everyone The 1 and 2 domains form a cleft or pocket able to noncovalently bind peptides Peptides Are Bound Within MHC Class I Molecules by Hydrogen Bonds and Ionic Interactions Between Amino Acids in the Peptide Ends and the MHC Molecule Polymorphism in the MHC Molecules is Restricted to the Peptide-Binding Cleft Peptides Bind to MHC Class I Molecules Through Anchor Residues Unique for Each MHC Molecule Structural Basis For the Tight Binding of Peptides: Limited in Length Within the MHC Class I Cleft Some Residues of the Peptide in the MHC Molecule Are Aligned Toward MHC Binding Clefts and Others Toward the T Cell Receptor From Dr. Stanley Nathenson Structural Representation of Anchor Residue Binding of Peptides Within the MHC Cleft T cell epitopes Peptide MHC Class I molecule From Dr. Stanley Nathenson Anchor Residues MHC Class II molecules can present a diverse yet limited number of peptides sized 13 – 17 amino acids long. What is the structural basis permitting MHC Class II molecules to present longer peptides than MHC Class I molecules? Structure of MHC Class II Molecule Heterodimer of membrane-spanning -chain and -chain The -chain and chain are polymorphic The 1 and 1 domains form a cleft or pocket able to noncovalently bind peptides Part of the Peptide Is Bound to MHC Class II Molecules by Hydrogen Bonds and Ionic Interactions Between Amino Acids in the Peptide and the MHC Molecule Peptides of Variable Length Bind to MHC Class II Molecules Through Structurally Related Anchor Residues At Various Distances From the Ends of the Peptide Position 1 has hydrophobic residues Position 4 is negatively Position 9 is hydrophobic tyrosine (Y), leucine (L), charged aspartic acid (D) or glutamic acid (E) proline (P) or phenylalanine (F). Class I MHC or Class II MHC Molecules Present Peptides to CD8 or CD4 T Cells Respectively MHC Molecules Contain Binding Sites For Either CD4 or CD8 Structural Differences between Class I MHC and Class II MHC Molecules and Their Consequences Class I MHC Class II MHC Structure -chain and 2-microglobulin -chain and -chain Peptide size 8-9 amino acids 13-17 amino acids Cleft Peptide must be within cleft Ends of peptide can dangle outside of cleft Binding affinity Peptide tightly bound Peptide is bound looser T cell interaction CD8+ T cell CD4+ T cell How do peptides get into those clefts and what are the functional ramifications of this process? Remember that presentation of a foreign peptide in a Class I MHC molecule to a CD8 T cell is a death sentence Cells Contain Two Intracellular Compartments: The Vesicular Which Communicates With the Extracellular Fluid and Cytosol Which Does Not The Compartmental Localization of Pathogen Determines the Destination of Its Peptides Peptides Presented by MHC Class I Molecules Are Derived From Intracellular Proteins The Proteosome Generates Peptides of Equivalent Size From Proteins The TAP Molecule Transports Peptides Into the Lumen of the Endoplasmic Reticulum Cytosolic Proteins Are Degraded and Transported Into the ER Where They Can Bind to MHC Class I Molecules Peptides Presented by MHC Class II Molecules Are Derived From Extracellular Proteins The Phagolysosome Generates Peptides of Different Sizes From Proteins MHC Class II Molecules Are Exported From the ER With Its Cleft Containing the Invariant Chain Processing of Invariant Chain to CLIP Peptide Peptides Derived From Exogenous Antigen Replace the CLIP Peptide in the MHC Class II Molecule Cleft in the Endosome Class I MHC or Class II MHC Molecules Present Peptides to CD8 or CD4 T Cells Respectively The T Cell Receptor Specifically Recognizes Sequences in the MHC Molecule and the Peptide it is Presenting Alloreactivity May Be Due to Heightened Affinity of a T Cell Receptor to a Different Nonself Peptide Alone or a Nonself Foreign MHC Molecule Alone Differences Between Peptide Processing of Class I and Class II MHC Molecules Class I MHC Class II MHC Peptide Source Endogenous Exogenous Peptide loading Endoplasmic reticulum Endosome Peptide used for folding Antigen-derived peptide CLIP peptide T cell interaction CD8+ T cell CD4+ T cell Cellular sequela of presentation Death Activation Tetramers Can Identify and Quantify Ag-specific T Cells MMWR May 23 1980 (1980; 29: 229-30) National surveillance data, first MMWR report 55 cases of TSS from 8 states; 31 from Wisconsin 52 (95%) cases in women 38 (95%) of 40 (known history) onset during menses 33 (73%) of 45 had S. aureus isolated from mucosal site Case fatality rates: 13% overall: 3.2% (1/31) in Wisconsin, 25% (6/24) in 7 other states Necrotizing Rash Associated With Toxic Shock Syndrome Superantigens Bind Directly to the TCR and Activate T Cells Immunological Synapse From Grakoui, et al Science ,1999 Vol 285, 221-227 The Immunological Synapse is Characterized by a Ring of Adhesion Molecules Surrounding T cell Receptor-associated Molecules HIV Co-opts The Immunological Synapse to Enhance Cell-to-cell Transmission Env J Clin Invest. 2004; 114(5):605 Questions to Consider What is the structural basis by which MHC molecules present peptides to the T cell receptor? How are endogenous peptides targeted to MHC Class I molecules and exogenous peptides targeted to MHC Class II molecules? How does the T cell receptor see the peptide and MHC molecule? What is the structural basis for CD4 T cells/MHC Class II and CD8 T cell/MHC Class I restriction?