* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download No Slide Title
Discovery and development of direct thrombin inhibitors wikipedia , lookup
Discovery and development of tubulin inhibitors wikipedia , lookup
Polysubstance dependence wikipedia , lookup
Discovery and development of proton pump inhibitors wikipedia , lookup
Compounding wikipedia , lookup
Orphan drug wikipedia , lookup
Psychopharmacology wikipedia , lookup
Neuropsychopharmacology wikipedia , lookup
Drug discovery wikipedia , lookup
Drug design wikipedia , lookup
Pharmacognosy wikipedia , lookup
Pharmaceutical industry wikipedia , lookup
Prescription drug prices in the United States wikipedia , lookup
Neuropharmacology wikipedia , lookup
Prescription costs wikipedia , lookup
Drug interaction wikipedia , lookup
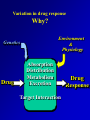
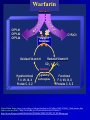
INTERPATIENT VARIABILITY OF DRUG DISPOSITION & DOSAGE ADJUSTMENT Readings (Applied Biopharm & PK 5th Ed.): Chp 12. Pharmacogenetics p 355-68. Chp 21. Renal & Hepatic Disease p 673-714 Chp 20. Peds/Geriatrics/Obesity p 634-42. Several articles will be posted on-line. Objectives • Identify variation in response • Understand underlying genetic, environmental and pathophysiological factors responsible for patient differences in drug response. • Evaluate clinical significance • Individualize drug therapy based on specific patient factors (using knowledge, logic and available equations). “Personalized Medicine” 20th Century Medicine: One Size (or Dose) Fits All • Currently use “trial and error” method of prescribing IS IT EFFECTIVE? Medicine’s Dirty Little Secretonly 50% of patients respond to major drug groups. Drugs Non-responders TNFa inhibitors Tricyclic antidepressants SSRI antidepressants Beta-blockers ACE inhibitors 5-HT1 blockers (migraine) HMG CoA red. inhibitors Interferons Anti-neoplastics >40% 20-50 % 10-25 % 15-35 % 10-30 % 20-45 % 10-30 % 60-90 % 50-70 % IS IT SAFE? • Adverse drug reactions (ADRs) represent the 4th leading cause of hospitalization and is responsible for 100,000 deaths/yr in the U.S. 2 million hospitalizations/yr in US • U.S. Health Management Organization (HMO) data suggest that the healthcare cost of treating drug ADRs exceeds the cost of providing the medications themselves Cost estimates range between 30-150 billion/ yr in US. Interpatient variability of drug response • Inter-patient variability in response to drug therapy is the rule, not the exception for almost all medications. • Research in the past 5- 15 years has identified many sources of inter-patient variability- which can be used for drug and dosage selection. • New knowledge, particularly in the area of pharmacogenetics, is progressing at a rapid pace. Variation in drug response Why? Environment & Physiology Genetics Drug Absorption Distribution Metabolism Excretion Target Interaction Drug Response Warfarin Sw ar fa rin rin rfa wa S- CYP1A1 CYP1A2 CYP3A4 Rwa rf a rin Warfarin in r a rf a w RVitamin K Reductase Oxidized Vitamin K CYP2C9 Reduced Vitamin K CO2 O2 Calumenin Hypofunctional F. II, VII, IX, X Protein C, S, Z γ-glutamyl carboxylase Functional F. II, VII, IX, X Proteins C, S, Z From Brian Gage; http://www.fda.gov/ohrms/dockets/ac/05/slides/2005-4194S1_Slide-Index.htm Other relevant slides: http://www.fda.gov/ohrms/dockets/ac/05/slides/5 http://www.fda.gov/ohrms/dockets/ac/05/slides/2005-4194S1_02_02-Huang.ppt Factors which impact warfarin dose requirements Environmental Age Gender Drugs Body wt Race Diet Others UNKNOWN CYP2C9 VKORC1 Genetic <Caldwell M., CPSC Advisory Committee Meeting, November 14, 2005> http://www.fda.gov/ohrms/dockets/ac/05/slides/8> ENVIRONMENTAL & PHYSIOLOGICAL FACTORS - Exposure to drugs / toxins/ pollutants - Diet - Disease - Age - Weight - Gender/ hormones - Exercise - Others? Lets look at several important examples of environmental factors which impact the absorption / distribution / elimination of drugs. Multiple factors can play a role. A. Absorption Influenced by: Permeability Motility Active Transporters Metabolic Enzymes 1. May be altered in diseases of GIT • Colitis- diarrhea, flu motility; absorption • Inflammation (Crohn’s, IBD) scar tissue: absorption • Cystic fibrosis Mucus & electrolyte changes, Malabsorption • Malnourishment F of Vitamin & minerals • Achlorhydria pH -Dose dumping of Enteric coated 2. May be altered by diet. • Grapefruit juice – CYP3A4 & PGP in gut (up to 3X) drug concentrations: cyclosporin A (CsA) , terfenadine, midazolam, felodipine, Ca++ channel blockers, talinolol - uptake transporters : oatp - Altered bioavailability of substrates such as fexofenadine, digoxin, pravastatin, atorvastatin Fexofenadine [C] ng/ml Example Effect of Grapefruit juice (300 ml- taken with drug) on bioavailability of fexofenadine. 400 300 Water 200 GFJ 100 0 0 2 4 6 8 Hours GFJ decreases intestinal expression of OATP- an active transporter involved in the uptake (absorption) of fexofenadine. Bioavailability reduced by half. 3) May be altered by drugs or natural products • Herbal products – Induction of intestinal CYP3A and PGP by St John's Wort. • Decreased oral availability of drug substrates. (CsA, indinavir, digoxin) Cyclosporin- has resulted in numerous cases of organ transplant rejection. • Decreased effectiveness of oral contraceptives. -Potential for unplanned pregnancy B. Distribution 1) Disease-associated changes in plasma protein concentrations. albumin: binding: Vd - NSAIDs α1-acid glycoprotein: binding, Vd -propafenone, propranolol 2) Obesity distribution of fat soluble drugs 3) Pregnancy fat, water, weight, placenta 4) Age Changes in body composition 5) Altered blood-brain barrier Disease-induced changes in expression of drug transporters at BBB Altered permeability of membrane * Will cover in more detail in future lectures C. Elimination There are numerous examples where hepatic and renal elimination is affected by environmental or physiological changes. 1) Environmental Toxins 2) Food 3) Drugs 4) Disease 5) Age 6) Pregnancy Environmental Pollutants: Polycyclic Hydrocarbons induce P450s • Smoking • Charcoal Broiling • Pollutants Increased drug clearance: theophylline, phenacetin Food: High protein diet: creatinine Alcohol: P450 Red Wine: Cyclosporin A levels DRUGS http://medicine.iupui.edu/ flockhart/table.htm A. Induction of Metabolism Some known CYP P450 Inducers: CYP 1A2 cigarette smoke, omeprazole, phenobarbitone CYP2D6 dexamethasone, rifampin CYP2E1 Ethanol, isoniazid CYP3A Barbiturates, carbamazepine, ethosuximide, glucocorticoids, phenobarbital, phenytoin, rifampicin, ….. DRUGS B. Inhibition of Metabolism Some known CYP P450 Inhibitors: CYP 1A2 - cimetidine, fluoroquinolones CYP2D6 - fluoxetine, quinidine, paroxetine CYP2E1 - cimetidine, disulfiram CYP3A - eg. HIV protease inhibitors, antimicrobials (clarithromycin, erthryomycin, ketoconazole) - many more DRUGS C. Inhibition of Hepato-Biliary Secretion P-glycoprotein (efflux transporter) Quinidine/ quinine + digoxin: - CLbile digoxin (50-60%) Oatp (influx transporter) Gemfibrozil + statins: - 2X ↑ AUC pravastatin (ed hepatic uptake) - 4 X ↑ AUC cerivastatin Cyclosporin A + statins: - 4X ↑ AUC cerivastatin - 7 X ↑ AUC rosuvastatin (ed hepatic uptake) DRUGS D. Inhibition of Renal Secretion P-glycoprotein (efflux transporter) Quinidine + digoxin: - CLr digoxin (50-60%) Ritonavir + digoxin: - CLr digoxin Oatp (influx transporter) Probenecid + Cephalosporins: - CLr - 1.8X CLr with 2.4 X ↑ AUC cephradine OCT (organic cation transporter) Cimetidine : - CLr procainamide from 347 to 196 ml/min (↑AUC procainamide) - CLr metformin from 527 to 378 ml/min Diseases Drug metabolism and secretion is decreased in a variety of diseases which are associated with an inflammatory response. – infection, arthritis, Crohn’s disease, renal disease, cancer etc.. Altered drug PK and drug response is seen both clinically and in experimental animal disease models. Cancer • Inflammatory response induced by tumor growth has been shown to decrease activity of drug metabolizing enzymes in Cancer patients. (14C- Erythromycin Breath test) in Cancer Patients CYP 3A Enzyme Activity Levels of Inflammation Marker (C-reactive Protein) Arthritis Propranolol Arthritic vs Control Rats 10000- Arthritic Conc 1000- 100- Control TIME CYP P450 Activity Protein binding Bacterial Infection - Altered disposition of P-Glycoprotein Substrate (99Tc-Sestamibi) in Pregnant Rats Fetal: Placenta Ratio (% Control) Increased Fetal Accumulation Accumulation of 99 Tc-sestamibi in Fetus 500 Control 400 LPS 300 200 100 *0 Increased Maternal Accumulation Effect of LPS on 99mTc-sestamibi Disposition (4h) in Pregnant Rats 1800 1600 -Infection 1400 Tc-MIBI 1200 1000 %Control 800 600 400 200 0 Brain Liver Kidney Intestine Placenta Altered Maternal and Fetal Disposition- due to decreased expression and activity of P-glycoprotein Renal Disease Advanced kidney disease can impact the metabolism, intestinal and/or hepatobiliary elimination of non-renally cleared drugs 1) Pgp and CYP3A in intestine: ↑ oral bioavailability of Pgp/CYP3A substrates. - erythromycin, propranolol, tacrolimus 2) CYP3A & CYP2C11 in Liver: hepatic metabolism of substrates. 3) Hepatic expression of Oatp uptake transporter: hepatobiliary CL? Ex. Repaglinide in Renal Disease - non-renally cleared oral hypoglycemic (<8% Clr) - Excreted via bile: - extensively metabolized (glucuronidation, CYP3A, CYP2C8) - active transport via Oatp1B1 and ABCB1 Mild/mod disease Severe disease Increased AUC due to decreased hepatobiliary clearance: OATP & CYP3A