* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Kinetics & Equilibrium
Process chemistry wikipedia , lookup
Nuclear fusion wikipedia , lookup
Click chemistry wikipedia , lookup
Solar air conditioning wikipedia , lookup
Physical organic chemistry wikipedia , lookup
Rate equation wikipedia , lookup
Chemical reaction wikipedia , lookup
Marcus theory wikipedia , lookup
Heat transfer wikipedia , lookup
Photosynthetic reaction centre wikipedia , lookup
Thermodynamics wikipedia , lookup
Determination of equilibrium constants wikipedia , lookup
Chemical equilibrium wikipedia , lookup
Stoichiometry wikipedia , lookup
Bioorthogonal chemistry wikipedia , lookup
Chemical thermodynamics wikipedia , lookup
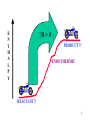
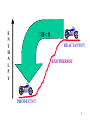
H H C HOH H KINETICS Cl H Cl H OH H C H Cl H H C OH H H OH Cl H C H H H H C OH Cl H H H C H Cl H Cl HH C H CHAPTER 9 H H C H HO H HO H Cl HO H 1 STUDY OF RELATIONSHIP OF CHEMISTRY THERMO DYNAMICS CHEMICAL REACTIONS AND ENERGY st LAW 1st LAW OFTHERMODYNAMICS THERMODYNAMICS OF THERMODYNAMICS 1st1LAW OF DEuniv = DE + DEsur = 0 Efinal - E DE init = - DEsur HEAT ABSORBED BY SYSTEM q > 0, HEAT IS ABSORBED = = q + w DE + WORK DONE ON SYSTEM q < 0, HEAT RELEASED w > 0, WORK ON SYSTEM w < 0, WORK BY SYSTEM q > 0, ENDOTHERMIC q < 0, EXOTHERMIC 2 MOST REACTIONS OCCUR AT CONSTANT TEMPERATURE AND PRESSURE .....BUT SOME ENERGY MAY BE LOST HEAT OF REACTION HEAT ABSORBED IN A ENTHALPY, H CHANGE IN ENTHALPY, DH: REACTION CARRIED OUT AT CONSTANT PRESSURE >0, REACTION ABSORBS HEAT ENDOTHERMIC DH: < 0, REACTION RELEASES HEAT EXOTHERMIC IS A STATE FUNCTION! PROPORTIONAL TO NUMBER OF MOLES OPPOSITE SIGN FOR REVERSE REACTION 3 E N T H A L P Y H>0 PRODUCT!!! ENDOTHERMIC REACTANT!! 4 E N T H A L P Y H< 0 REACTANTS!!! EXOTHERMIC PRODUCTS!! 5 Hcomb = HEAT ABSORBED WHEN 1 MOLE OF A SUBSTANCE REACTS WITH OXYGEN AT CONSTANT P C6H12O6 (s) + 6O2 (g) 6CO2 (g) + 6H2O (g) Hocomb = -2816 kJ STANDARD STATE MOST STABLE FORM AT 1 atm AND THE SPECIFIED TEMPERATURE FOR DISSOLVED SUBSTANCE, 1 M HOW MUCH HEAT IS RELEASED IF 10 g GLUCOSE IS BURNED? mol glucose = 10 g x 1 mol/180 g = 0.056 mol H = -2816 kJ/mol x 0.056 mol = -157.7 kJ 158 kJ of heat is released 6 BOND ENERGY: ENERGY NEEDED TO BREAK 1 MOLE OF BONDS IN THE GASEOUS STATE BREAKING: DHo : ALWAYS > 0 FORMATION DHo : ALWAYS < 0 ESTIMATE: DHo ~ S BE BROKEN - S BE FORMED H2C=CH2 + HCl 1 C=C 1 H-Cl 1 x 612 1 x 431 H3C-CH2Cl 1 C-H TABLE 9.1 1 C-Cl 1 x 413 AND 1 x 234 DHo ~ 1043 - 647 = ~ 396 kJ/mol 7 EVERY PROCESS INCREASES DISORDER IN THE UNIVERSE FOR A SPONTANEOUS PROCESS, SUNIV > 0 q S= J/K T AT WHICH HEAT IS ADDED Sgas > Sliquid > Ssolution > Ssolid 8 DSuniv = DSsur + DS DSuniv > 0 SPONTANEOUS PROCESS NON-SPONTANEOUS PROCESS DSuniv > 0 WHERE THE NUMBER OF MOLES OF GAS INCREASES 9 ESTIMATING ENTROPY CHANGE: COMPARE PRODUCTS TO REACTANTS N2 (g) + 3H2 (g) NaCl (s) 2NH3 (g) Na1+ (aq) + Cl1- (aq) CaCO3 (s) + H301+(aq) H20 (s) H20 (l) CO2 @ 20 oC Ag (s) + NaCl (s) DS <0 >0 Ca2+ (aq) + 3H20 (l) + CO2 (g) >0 H20 (g) >0 >0 CO2 @ 0 oC <0 AgCl (s) + Na (s) ~0 10 DSuniv = DSsur + DS AT CONSTANT P: DSsur = -DH/T -TDS DGuniv = DH - TDS FOR A SPONTANEOUS REACTION: DSuniv > 0 DG < 0 THE ENERGY OF THE PROCESS MUST DECREASE AND THE UNIVERSE MUST BECOME MORE RANDOM!!!! 11 DRIVING FORCES FOR A CHEMICAL REACTION: DH -- ENERGY REQUIRED TO CHANGE TO POTENTIAL ENERGY OF REACTANTS TO THAT OF PRODUCTS -TDS -- ENERGY TO MAKE THE SYSTEM MORE ORDERED RELATE TO DG = DH - TDS DH + + - DS + + - <0 SPONTANEOUS? ALWAYS - AT ANY T NEVER - AT ANY T AT HIGH T AT LOW T 12 ENERGY DIFFERENCES ONLY! WHAT IS POSSIBLE WHAT IS NOT POSSIBLE CONCERNED WITH PATH WHAT HAPPENS HOW FAST IT HAPPENS 13 CH3Br + OH 1- TRANSITION STATE P O T E N T I A L Ea DE H H C H Br OH - CH3OH + Br 1- STERIC EFFECTS: MUST HAVE PROPER ORIENTATION MINIMUM OK OF H-O CAMOUNT ENERGY FOR COLLISION O-H C NR TO ACHIEVE TRANSITION STATE Ea(reverse) NEED COLLISION OF PROPER ENERGY AND ORIENTATION FOR ELECTRONS TO BE SHARED OR TRANSFERRED E 14 RATE OF DISAPPEARANCE M O L L RATE SLOWS WITH TIME RELATED TO NUMBER OF REACTING PARTICLES FOR RATE OFAPPEARANCE Rf = kf[A]X[B]Y SEC 15 X[B]Y R = k [A] f IN IT’S SIMPLEST FORM: f CH3Br + OH 1- <−> CH3OH + Br 1- Rf = kf[CH3Br][OH1-] N2 + 3H2 <−> 2NH3 Rf = kf[N2][H2]3 2NO2 <−> N2O4 Rf = kf[NO2]2 HF (aq) + NH3 (g) <−> NH41+ (aq) + F1- (aq) Rf = kf[HF][NH3] CATALYSTS & INHIBITORS 16 H2 (g) + I2 (g) <−> 2HI (g) RATE 1 OR Rf 2HI (g) <−> H2 (g) + I2 (g) RATE 2 OR Rr H2 (g) + I2 (g) <−> 2HI (g) Rf = Rr Rf [HI]2 K = R = [H ][I ] r 2 2 = [PRODUCTS] [REACTANTS] 17 K= F1- [PRODUCTS] [REACTANTS] (aq) + HNO2 (aq) <−> HF (aq) + NO2 1- 1 [ HF ][ NO (aq) 2 ] K = 1 [F ][ HNO 2 ] [X] = MOLAR CONCENTRATIONS 2HCl (g) <−> H2 (g) + Cl2 (g) K= PH 2 PCl 2 2 (PHCl ) CAN ALSO USE CONCENTRATIONS CaF2 (s) + 2H3O1+ (aq) <−> Ca2+ (aq) + 2HF (aq) + 2H2O (l) [Ca 2 ][ HF]2 K [H 3O1 ]2 18 F1- (aq) + HNO2 (aq) <−> HF (aq) + NO21- (aq) [HF][ NO12 ] K = 1 [F ][ HNO 2 ] HCN (aq) + H2O (l) <−> CN1- (aq) + H3O1+ (aq) [CN1 ][ H3O1 ] K [HCN] PbBr2 (s) <−> Pb2+ (aq) + 2Br1- (aq) K SP= [Pb2+][Br1-]2 19 DGo = - RTlnK K >> 1 K << 1 EXTENSIVE LARGE AMOUNT OF PRODUCT EXOTHERMIC PROCESSES NOT EXTENSIVE SMALL AMOUNT OF PRODUCT ENDOTHERMIC (IF NO CHANGE IN MOLE OF GAS INVOVED) K VARIES ONLY WITH TEMPERATURE!!!!!! 20 REACTANTS <−> PRODUCTS + HEAT K= [PRODUCTS] [REACTANTS] LeCHATELIER’S PRINCIPLE: A SYSTEM AT EQUILIBRIUM WILL RESPOND TO A STRESS IN A WAY TO MINIMIZE THE EFFECT OF THE STRESS ADD PRODUCT: FAVOR REACTANTS DRIVE TO LEFT ADD REACTANT: FAVOR PRODUCTS DRIVE TO RIGHT 21 PbBr2 (s) <−> Pb2+ (aq) + 2Br1- (aq) DHo= 37.2 kJ/MOL b) REMOVING SOME Br1- c) ADDING PbBr2 (s) TO RIGHT OR FAVORS PRODUCTS NO CHANGE!!! d) INCREASING TEMPERATURE e) DOUBLING THE VOLUME f) ADDING Pb2+ 22