* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Chemistry: the study of composition, structure, and properties of
Safety data sheet wikipedia , lookup
Catalytic reforming wikipedia , lookup
Biochemistry wikipedia , lookup
Properties of water wikipedia , lookup
Chemical thermodynamics wikipedia , lookup
Gas chromatography–mass spectrometry wikipedia , lookup
Artificial photosynthesis wikipedia , lookup
Registration, Evaluation, Authorisation and Restriction of Chemicals wikipedia , lookup
Chemical bond wikipedia , lookup
Water pollution wikipedia , lookup
Isotopic labeling wikipedia , lookup
Water splitting wikipedia , lookup
Metalloprotein wikipedia , lookup
Stoichiometry wikipedia , lookup
Chemistry: A Volatile History wikipedia , lookup
Condensed matter physics wikipedia , lookup
Freshwater environmental quality parameters wikipedia , lookup
History of chemistry wikipedia , lookup
Electrolysis of water wikipedia , lookup
IUPAC nomenclature of inorganic chemistry 2005 wikipedia , lookup
Sol–gel process wikipedia , lookup
History of molecular theory wikipedia , lookup
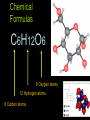
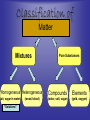
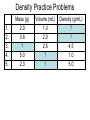
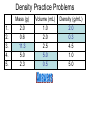
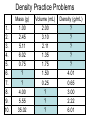
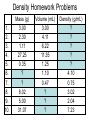
Chemistry: the study of composition, structure, and properties of matter* and the changes it undergoes. *Matter: composed of mass and takes up space. Elements and Compounds • • • All matter is composed of atoms. – Copper pipe is made of copper atoms. – Oxygen gas is made of oxygen atoms. An element is a pure substance made of only one kind of atom. – Carbon is an element made of only carbon atoms. Atoms of two or more elements bond together to make compounds. – CO2 (___ carbon atom and ___ oxygen atoms) – H2O2 (___ hydrogen atoms & ___ oxygen atoms) Chemical Formulas C6H12O6 6 Oxygen atoms 12 Hydrogen atoms 6 Carbon atoms Classification of Matter Mixtures Pure Substances Homogeneous Heterogeneous Compounds Elements (air, sugar in water) (water, salt, sugar) (gold, oxygen) “Solutions” (wood, blood) Mixtures vs. Pure Substances MIXTURES • Blend of two or more types of matter (each retains its own identities and properties.) • Sugar-Water…is both sugar and water. Tastes like sugar and tastes like water. • Examples: grape juice (water and sugar) PURE SUBSTANCES • Every sample has exactly the same properties and came composition. – Pure water is always 11.2% hydrogen and 88.8% oxygen. Homogeneous vs. Heterogeneous Mixtures Homogeneous • The same throughout… every part is evenly distributed. • Examples: Heterogeneous • Not the same throughout… every part is not evenly distributed. • Examples: – vinegar (water and acetic acid) – Salad (tomatoes, lettuce, olives, carrots, etc.) “HOMO” : the same “HETERO” : different Making a Mixture: “Mixing” Miscible: able to be mixed (liquids only) EX. Gasoline is a mixture of about 100 liquids. Immiscible: not able to be mixed (liquids only) EX. Oil and water cannot mix. Gases can mix with liquids also. An example is a carbonated drink like Pepsi. CO2 + sugar + water + flavorings + Awesome Pepsi Separation of Mixtures DISTILLATION FILTRATION 1. Boil mixture. 1. Pour mixture onto a mesh, like filter paper. 2. Liquid boils off, leaving solid minerals behind. 2. Liquid passes through paper. 3. Collect the boiled vapor in a condenser. 3. Solid is left on the paper. 4. Collected liquid is pure(er). Ex: Coffee filters Ex: Seawater Physical Properties A characteristic that can be observed or measured without changing the identity of the substance. Examples: boiling and freezing points Physical Change: does not involve a change in identity of the substance…cutting, melting, boiling, freezing, dissolving. Change of State: physical change from one state to another. • Solid Liquid (Melting) Liquid Solid (Freezing) • Solid Gas (Subliming) Gas Solid (Deposition) • Liquid Gas (Evap./Boil) Gas Liquid (Condensing) SOLID - LIQUID - GAS - PLASMA Physical Properties Other Physical Properties Include: - strength - hardness - magnetism - reflectivity - melting point (temp at which it boils) - melting point (temp at which it melts) - density What is Density? Density: a measurement of how much matter is contained in a certain volume. Calculate Density D = Mass Volume M D V ***Test: Density of Water: 1.00 g/mL Density Practice Problems 1. 2. 3. 4. 5. Mass (g) 2.0 0.6 ? 5.0 2.3 Volume (mL) 1.0 2.0 2.5 ? ? Density (g/mL) ? ? 4.5 1.0 5.0 Density Practice Problems 1. 2. 3. 4. 5. Mass (g) 2.0 0.6 11.3 5.0 2.3 Volume (mL) 1.0 2.0 2.5 5.0 0.5 Density (g/mL) 2.0 0.3 4.5 1.0 5.0 Density Practice Problems 1. 2. 3. 4. 5. 6. 7. 8. 9. 10. Mass (g) 1.00 2.45 5.11 6.02 0.75 ? ? 4.00 5.55 35.02 Volume (mL) 2.00 3.10 2.11 1.35 1.75 1.50 0.25 ? ? ? Density (g/mL) ? ? ? ? ? 4.01 0.65 3.00 2.22 6.01 Density Homework Problems 1. 2. 3. 4. 5. 6. 7. 8. 9. 10. Mass (g) 3.00 2.30 1.11 27.25 0.35 ? ? 8.02 5.00 31.07 Volume (mL) 3.00 4.11 6.22 11.35 1.25 1.10 0.47 ? ? ? Density (g/mL) ? ? ? ? ? 4.10 0.75 3.02 2.04 7.23 Weight vs. Density Density: a measurement of how much matter is contained in a certain volume. Weight: the measurement of gravity on an object’s mass…”how heavy”. Physical States of Matter • SOLID: – Definite Shape – Definite Volume – Particles move slowly and are close together • Examples – Sodium metal (Na) – Ice (H2O) – Gold at room temp. (Au) Physical States of Matter • LIQUID: – No Definite Shape – Definite Volume – Particles move a little faster than solids and more freely. • Examples – Mercury (Hg) – Water (H2O) – Gold at high temp. (Au) Physical States of Matter • GAS: – No Definite Shape – No Definite Volume – Particles move very quickly and are far apart. • Examples – Neon (Ne) – Steam (H2O) – Chlorine gas (Cl2) Physical States of Matter • PLASMA: – High Temperature – Atoms lose electrons easily • Examples – Sun – Fluorescent Bulbs Chemical Properties… …relate how chemicals undergo change. Chemical Change (reaction): when substances are converted into NEW substances. Reactants REACT. Products are PRODUCED. reactant + reactant product carbon + oxygen carbon dioxide C + O2 CO2 Chemical Properties… …relate how chemicals undergo change. Other Chemical Properties Include: - Reactivity (how readily a reaction will occur) - Flammability (ability to burn) - Tarnishing - Reacting with water, oxygen, acids, bases… Chemical Changes …can be detected. Indications of a Chemical Reaction: 1. Change in odor or color. (burnt food) 2. Fizzing or Foaming (Alka-Seltzer) 3. Sound, heat, or light (TNT) Review P. 70-71 (Questions 1 – 21; 24 – 27) - Do not write the question. Just the answer. - Vocabulary on p.69 will be on the test.