* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download VIII. Other Types of Notations or Configurations
Renormalization wikipedia , lookup
Density functional theory wikipedia , lookup
Elementary particle wikipedia , lookup
Ferromagnetism wikipedia , lookup
Bremsstrahlung wikipedia , lookup
Molecular orbital wikipedia , lookup
Particle in a box wikipedia , lookup
Quantum electrodynamics wikipedia , lookup
Chemical bond wikipedia , lookup
Double-slit experiment wikipedia , lookup
Hydrogen atom wikipedia , lookup
X-ray fluorescence wikipedia , lookup
Matter wave wikipedia , lookup
Auger electron spectroscopy wikipedia , lookup
X-ray photoelectron spectroscopy wikipedia , lookup
Tight binding wikipedia , lookup
Theoretical and experimental justification for the Schrödinger equation wikipedia , lookup
Wave–particle duality wikipedia , lookup
Atomic orbital wikipedia , lookup
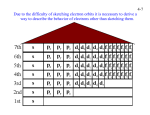
Unit 10 : Light, Quantum Theory and Electron Configurations “Where it all starts to come together” I. Review of Previous Atomic Structure Theories • A. Dalton – 1. All elements are composed of atoms--indivisible and indestructible particles – 2. All atoms of the same element are exactly alike-- they all have the same mass – 3. Atoms can physically mix or chemically combine in simple whole # ratios – 4. Reactions occur when atoms separate, join, or rearrange JJ Thomson • B. J.J. Thompson – 1. First to theorize the existence of the electron. – 2. Atomic Model: • Discovered the e- from work with cathode rays - + + + + -+ + - “Plum Pudding Model” Rutherford • C. Rutherford – 1. First to theorize the existence of the positive nucleus. – 2. Atomic Model: • Gold Foil Experiment with “Alpha particles” • “It is about as incredible as if you had fired a 15-inch shell at a piece of tissue paper and it came back and hit you.” -ER + Rutherford Bohr • D. Bohr – 1. First to theorize the existence of orbiting electrons – 2. Atomic Model: • “Planetary Model” Present Model • E. Present Model – 1. Names this model is known by: • A. Charge Cloud Model • B. Orbital Model • C. Quantum Mechanical Model – 2. Location of electrons • Outside of nucleus • “probable locations of electrons” – Electrons are WAVES! II. The Dual Nature of Light • A. Modern Theory of Light – Light has a dual nature-can behave like waves or particles • B. Light as Waves – 1. Characteristics of Waves • A. Wavelength ( “lambda”) – Distance between any two points in a wave (m) • B. Frequency ( “nu”) – # of peaks that pass a given point each second (cycles/sec Hz) II. The Dual Nature of Light • C. Wave Velocity (speed) – Distance that a peak travels in a unit of time (1 second) Relationship: = c c = 3.00*108 m/s -or- = c/ Real-World Application • Favorite Radio Station? II. The Dual Nature of Light • C. Discoveries – 1. Planck’s Theory • Predicted how the spectrum of light changes with temp • Energy absorbed or emitted is quantized in nature (See IV. A) • E = h (h = Planck’s Constant = 6.626*10-34 J*s) – 2. Photoelectric Effect • Einstein proposed light consists of small packets of energy called photons • Light behaves like a particle each photon carries a certain amount of energy (given by Planck’s equation) II. The Dual Nature of Light – 3. Compton • Discovered and concluded that light has properties of both particles and waves – 4. Louis de Broglie • Stated that particles of matter should behave like waves and exhibit a wavelength (and vice-versa) • Most matter has undetectable wavelengths • Related mass, wavelength, and velocity of matter – =h/m • Led to the development of the electron microscope II. The Dual Nature of Light – 5. Heisenberg’s Uncertainty Principle • Position and momentum of a moving object can’t be simultaneously measured and known exactly • To locate an electron you must strike it with a photon or another particle III. The Emission and Absorption of Radiation • A. Continuous Spectrum – 1. Produced by: • Passing a band of “white light” through a prism – 2. Looks like: • The full array of colors (ROYGBIV) – Red = longest wavelength – Violet = shortest wavelength III. The Emission and Absorption of Radiation • B. Bright-line Spectrum – 1. Produced By: • Passing light through a prism when elements/compounds are heated – 2. Looks Like: • Distinct wavelengths of light given off called “spectral lines” • Unique for each element (Element DNA) – Atomic Line Spectra The electromagnetic spectrum • Complete range of wavelengths and frequencies • Mostly invisible IV. Quantum Theory or Mechanics (Wave Mechanics) • A. What does the word “quantized” mean? – Divide energy into small, but measurable amounts “Continuous” “Quantized” IV. Quantum Theory or Mechanics (Wave Mechanics) • B. How does the concept of “quantized” relate to energy levels? n=4 n=3 n=2 n=1 IV. Quantum Theory or Mechanics (Wave Mechanics) • C. Areas electrons can exist are divided into: – 1. Energy levels – 2. Subshells – 3. Orbitals • D. Orbital—Definition – Region within a sublevel or an energy level where an e- can be found (homes for e-) IV. Quantum Theory or Mechanics (Wave Mechanics) • E. Ground vs. Excited States – Ground state lowest energy an e- occupies – Excited state when an e- is in a higher energy state V. Breakdown of the Electron Cloud: Hotel Analogy • A. Floor – 1. Possibilities • “Principle quantum number” • Corresponds to energy level/row on Periodic Table – 2. Like a ENERGY LEVEL in an atom. • B. Wing or Corridor – 1. Possibilities • Names: – A. s, p, d, f – B. Floor each one starts on “s” – 2. Like SUBLEVELS in an atom. V. Breakdown of the Electron Cloud: Hotel Analogy • C. Room – 1. Possibilities • Names: – A. px, py, pz – B. How many on each corridor number of orbitals = n2 – 2. Like an ORBITAL in an atom. • D. People – 1. How many in a room 2 e– 2. Like a PAIR OF ELECTRONS in an atom. – 3. How do we indicate the different people? • VI. Breakdown of the Electron Cloud: Energy Levels, Sublevels, and Oribitals • A. Principle Energy Level (n) – 1. Describes: • # of principle energy level (row on P.T.) – 2. Energy Order: • 1, 2, 3, 4, … – 3. Number of electron in each energy level • • • • A. n = 1 2 eB. n = 3 8 eC. n = 3 18 eD. General Formula 2 n2 VI. Breakdown of the Electron Cloud: Energy Levels, Sublevels, and Oribitals • B. Energy Sublevels – 1. Types of sublevels Sublevel s Shape # of each Energy Levels? n = 1, 2, 3, 4 p n = 2, 3, 4 d n = 3, 4 f n=4 VI. Breakdown of the Electron Cloud: Energy Levels, Sublevels, and Oribitals – 2. Energy order of sublevels • n =1 < 2 < 3 < 4 – 3. General formula for number of sublevels at each energy level • # sublevels = principle quantum # – 4. Orientation of sublevels in space •s • px, py, pz •d VI. Breakdown of the Electron Cloud: Energy Levels, Sublevels, and Oribitals • C. Orbitals – 1. Maximum number of electrons in an orbital • 2 e- – 2. General formula for number of electrons in a principle energy level • 2 n2 maximum number of e- – 3. Electron pair—definition • Only two e- can occupy an orbital (within a sublevel) at a time. They must have opposite spins. • D. See Summary Pg. 11 – Complete table on Pg. 12 VII. Arrangement or Configuration of Electrons • A. Order is determined by two basic principles: – Pauli Exclusion Principle (2e-/orbital) – Hund’s Rule (half-filled before completely filled orbitals) • B. Energy Order—See Pg. 14 • C. Memory device for energy order—see attachment VII. Arrangement or Configuration of Electrons • D. Regular Electron Configuration – 1. Example Notation • 1s2 2s1 (Pronounced “one-s-two, two-s-one”) • A. What does the coefficient mean? – Principle Quantum # • B. What does the letter mean? – Type of orbital (subshell) • C. What does the exponent mean? – # of electrons in that orbital VII. Arrangement or Configuration of Electrons • E. How to write e- configurations • F. Examples of e- configurations – – – – 1. He 2. N 3. Na 4. Ca 1s2 1s2 2s2 2p3 1s2 2s2 2p6 3s1 1s2 2s2 2p6 3s2 3p6 4s2 • Practice Time! (Pg. 16) VIII. Other Types of Notations or Configurations • A. Orbital Notation or Configuration – 1. How are each of the following represented? • A. Orbital • B. Electron VIII. Other Types of Notations or Configurations – 2. What does each type of sublevel look like in orbital notation? • A. s • B. p • C. d • D. f • *Note* Put brackets around each set of orbitals! VIII. Other Types of Notations or Configurations – 3. How is each sublevel filled? • A. Hund’s Rule – Every orbital in a subshell (s, p, d, f) is singly occupied with one e- before any one orbital is doubly occupied • B. Pauli Exclusion Principle – In order for two electrons to occupy the same orbital, they must have opposite spins – 4. How to write an orbital configuration (Pg. 19) VIII. Other Types of Notations or Configurations – 5. Examples of Orbital Configurations • A. He • B. N • C. Na • D. Ca VIII. Other Types of Notations or Configurations • B. Shorthand Configuration – 1. Where are the noble gases on the periodic table? • Group VIII (far right) – 2. Why are the noble gases special? • Noble gases have 8 e- in their outer shell • Very stable (least reactive) – 3. How can we use noble gases to shorten the regular e- configurations? • Ex. Ba VIII. Other Types of Notations or Configurations – 4. How to write a shorthand configuration (Pg. 22) – 5. Examples of shorthand configuration • A. Na • B. Mn • C. Ca IX. Relating Electron Configurations to the Periodic Table • A. Location of the final electrons in each element: – – – – 1. s (Groups I, II) 2. p (Groups IIIA – VIIIA) 3. d (Transition metals) 4. f (Lanthanides, Actinides) • How to write configurations using the P.T. X. Valence Electrons and Lewis (Electron Dot) Structures • A. Definition – Valence Electrons • Electrons that are in the outermost principle energy level (bonding electrons) • “Where the action is” in Chemistry • B. Outermost principle energy level is called the valence shell – must include at least 1 electron X. Valence Electrons and Lewis (Electron Dot) Structures • C. Inner electrons are called core or kernel electrons • D. Examples – Be: 1s2 2s2 – O: 1s2 2s2 2p4 – Sc: 1s2 2s2 2p6 3s2 3p6 4s2 3d1 2 valence e4 valence e2 valence e- X. Valence Electrons and Lewis (Electron Dot) Structures • E. How to find valence e- easily? – The highest energy level (n = ?): # of e- here! • F. Representing valence e- in atoms using Lewis or Electron Dot structures – 1. Chemical Symbol – 2. Each dot represents valence e– 3. “s” orbital valence e- double up on top, then single egoing clockwise X. Valence Electrons and Lewis (Electron Dot) Structures • G. Examples of Lewis Dot Structures – 1. Hydrogen – 2. Carbon – 3. Phosphorus End of Unit 10!!