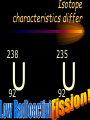* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Energy
Electron configuration wikipedia , lookup
Bohr–Einstein debates wikipedia , lookup
Bremsstrahlung wikipedia , lookup
Hydrogen atom wikipedia , lookup
Tight binding wikipedia , lookup
Elementary particle wikipedia , lookup
Double-slit experiment wikipedia , lookup
Particle in a box wikipedia , lookup
Molecular Hamiltonian wikipedia , lookup
Mössbauer spectroscopy wikipedia , lookup
X-ray photoelectron spectroscopy wikipedia , lookup
Ultrafast laser spectroscopy wikipedia , lookup
Rutherford backscattering spectrometry wikipedia , lookup
Matter wave wikipedia , lookup
Electron scattering wikipedia , lookup
X-ray fluorescence wikipedia , lookup
Wave–particle duality wikipedia , lookup
Atomic theory wikipedia , lookup
Theoretical and experimental justification for the Schrödinger equation wikipedia , lookup
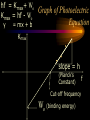
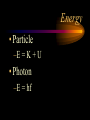
The photon • A “particle” of light • A “quantum” of light energy • The energy of a given photon depends on the frequency (color) of the light But light is also a wave! • Travels at constant speed c in a vacuum. • c = lf 8 –c: 3 x 10 m/s – l: wavelength (m) – f: frequency (Hz) Calculating photon energy •E = hf –E: energy (J or eV) –h: Planck’s constant • 6.62510-34 J s or 4.14 10-15 eV s –f: frequency of light (s-1, Hz) The “electron-volt” (eV) is an energy unit • Useful on the atomic level. • If a moving electron is stopped by 1 V of electric potential, we say it has 1 electron-volt (or 1 eV) of kinetic energy! Converting eV to Joules (J) 1 eV = -19 1.60210 J Photoelectric Effect experiment light Collector (-) At a certain voltage, PhotoVs, the current can’t Diode e- e- e- e- e- eflow anymore! (+) e- e- e- e- V e- ee- e- e- Ae- e- Anomalous Behavior in Photoelectric Effect • Voltage necessary to stop electrons is independent of intensity (brightness) of light. • Photoelectrons are not released below a certain frequency, regardless of intensity of light. • The release of photoelectrons is instantaneous, even in very feeble light, provided the frequency is above the cutoff. Voltage current for different intensities of light. i I3 > I2 > I1 I3 I2 I1 Vs Stopping potential is unaffected! V Voltage versus current for different frequencies of light. i f3 > f2 > f1 f3 f2 f1 V Vs,3 Vs,2 Vs,1 Stopping potential becomes more negative at higher frequencies! Photoelectric Effect • Ephoton = Kmax + Wo –Ephoton = hf (Planck’s equation) –Kmax: maximum kinetic energy of electrons –Wo: binding energy or “work function” • hf = Kmax + Wo hf = Kmax+ Wo Kmax = hf - Wo y = mx + b Graph of Photoelectric Equation Kmax slope = h (Planck’s Constant) Cut-off frequency Wo (binding energy) f Absorption Spectrum Photon is absorbed and excites atom to higher quantum energy state. 0 eV DE hf Ground state -10 eV Absorption Spectrum Absorption spectra always involve atoms going up in energy level. ionized 0 eV -10 eV Emission Spectrum Photon is emitted and atom drops to lower quantum energy state. Excited state 0 eV DE hf -10 eV Emission Spectrum Emission spectra always involve atoms going down in energy level. ionized 0 eV -10 eV A typical nucleus 12 Atomic mass: protons plus neutrons C 6 Element name Atomic number: protons Isotope characteristics differ 238 235 92 92 U U Binding energy • Energy released when a nucleus is formed from protons and neutrons. • Mass is lost. 2 • E = mc –where m is the lost mass Nuclear Particles 1 p 1 1 n 0 • Nucleons – Proton • Charge: +e • Mass: 1 amu – Neutron • Charge: 0 • Mass: 1 amu Nuclear reactions • Nuclear Decay –Alpha decay –Beta decay • Beta Minus • Positron • Fission • Fusion Decay Particles 4 He •Alpha 2 0 e -1 0 e 1 •Beta •Positron Alpha Decay • Occurs only with very heavy elements. • Nucleus too large to be stable. 226 Ra 88 222 4 86 2 Rn He Beta Decay • Occurs with elements that have too many neutrons for the nucleus to be stable. 40 40 19 20 K Ca 0 e -1 n antineutrino Positron Decay • Occurs with elements that have too many protons for the nucleus to be stable. 2 2 2 1 He H 0 e 1 n neutrino Neutrino and Anti-Neutrino • Proposed to make beta and positron decay obey conservation of energy. • No mass, no charge. • Energy and spin. • Does not react easily with matter. • Hard to detect. Gamma Radiation, g • Released by atoms which have undergone a nuclear reaction. • Results when excited nuclei return to ground state. • High energy! E = hf! Fission • Occurs only with very heavy elements. • Nucleus too large to be stable. • Induced by neutrons. 239 1 92 144 94 0 38 56 Pu n Sr Ba 4 1 n 0 Fusion • The largest amount of energy available. • Energy produced in the sun. • Fusion of light elements results in nonradioactive waste. 1 1 2 1 1 2 H H He Summary of Wave-Particle Duality Waves are particles and particles are waves Energy • Particle –E = K + U • Photon –E = hf Momentum • Particle –p = mv • Photon –p = h/l Wavelength • Photon – l = c/f • Particle – l = h/p – deBroglie wavelength Compton Scattering • Proof of the momentum of photons. • High-energy photons collided with electrons. • Conservation of momentum. • Scattered photons examined to determine loss of momentum. Davisson-Germer Experiement • Verified that electrons have wave properties by proving that they diffract. • Electron diffraction