* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Thermal Energy and Heat
Survey
Document related concepts
Solar water heating wikipedia , lookup
Heat equation wikipedia , lookup
Thermoregulation wikipedia , lookup
Passive solar building design wikipedia , lookup
Building insulation materials wikipedia , lookup
Copper in heat exchangers wikipedia , lookup
Thermal comfort wikipedia , lookup
Intercooler wikipedia , lookup
Thermal conductivity wikipedia , lookup
Cogeneration wikipedia , lookup
Solar air conditioning wikipedia , lookup
Hyperthermia wikipedia , lookup
Transcript
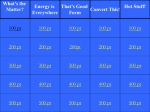
Thermal Energy and Heat Kinetic Theory of Matter ALL particles that make up matter are constantly in motion. ALL particles in matter have kinetic energy. Within a substance, not all particles move at the same rate. Temperature A measure of the average kinetic energy of the individual particles of matter Depends on mass as well as speed Temperature Thermometer is used to measure temperature 3 common scales Fahrenheit Celsius Kelvin Fahrenheit Scale Most common in the U.S. 32o = water freezes 212o = water boils Intervals in between are divided into 180 equal intervals called degrees Fahrenheit (oF) Absolute zero = -459 degrees Celsius Scale Used in most of the 0o = water freezes 100o world = water boils Intervals divided into 100 equal parts called degrees Celsius (oC) Absolute zero = -273 degrees Kelvin Scale Most commonly used in physical science 273 degrees = water freezes 373 degrees = water boils Kelvin scale is designed so that 0 = absolute zero Conversion between Celsius and Fahrenheit Thermal Energy The total energy of all of the particles in a substance or material The more particles a substance has at a given temperature; the more thermal energy it has Thermal Energy Example: 2L of hot chocolate at o 75 has more thermal energy than 1.5L of hot o chocolate at 75 Thermal Energy Thermal energy depends on: temperature # particles in a substance how the particles are arranged Heat Movement of thermal energy from a substance at a higher temperature to another at a lower temperature Heat When energy flows from one object to another; the thermal energy of BOTH objects changes Ex: ice cube in bowl Measuring Heat Calorie Amount of energy needed to raise the temp. of 1g of water by 1 degree Celsius Joule SI unit for energy One calorie = 4.18J Measuring Heat 1 C = 1kilocalorie (1000 calories) Each calorie of food contains 1000 calories of energy Measured by a calorimeter Specific Heat A quantity to measure the relationship between heat and temperature change Amount of energy required to raise the temperature of 1g of a substance by 1oC Every substance has its own specific heat Heat Transfer 3 ways heat can be transferred: Conduction Convection Radiation Conduction Heat is transferred without the movement of matter itself Heat is transferred by touching Convection Transfer of heat by the movements of a fluid Gas or liquid Convection Current Movement of a fluid caused by differences in temperature Radiation Transfer of energy by electromagnetic waves Feel radiation from a bonfire; heat lamp DOES NOT require matter to transfer thermal energy Heat Transfer Heat flows into a substance; thermal energy increases; as thermal energy increases, temperature increases Heat will flow until 2 substances have same temperature Heat Transfer Example: ice cream Heat from ingredients flows to the ice Conductors A material that conducts heat well Metals: silver, copper, stainless steel Insulators A material that does not conduct heat well. Wool, straw, paper, cork Gases: air Changes in State Three states of matter Solid Liquid Gas Changes in State Physical change is a change of state from one form to another Most often occurs between solid & liquid; liquid & gas Changes in State Matter will change from one state to another if thermal energy is absorbed or released. Changes in State The addition or loss of thermal energy changes the arrangement of the particles. The average kinetic energy DOES NOT change. Changes in State Melting: solid to liquid Melting point: temperature at which a solid changes to a liquid Changes in State Freezing: liquid to a solid Freezing point: when a substance loses thermal energy; temperature at which a liquid changes to a solid Changes in State Vaporization: liquid to a gas Evaporation: at the surface Boiling: below the surface Boiling point: temperature at which liquid becomes a gas (boils) Changes in State Condensation: becomes liquid gas Changes in State Thermal Expansion: expanding of matter when it is heated As thermal energy of a substance increases, its particles spread out. True even when a substance is not spreading out Changes in State Examples of thermal expansion: Thermometer Teeth (fillings) Thermostats (heatregulating device) Uses of Heat Heat engine Conversion of thermal energy to mechanical energy requires a heat engine Make use of combustion: burning fuel (coal, gasoline) Heat Engine During combustion, chemical energy is converted to thermal energy Heat engine converts the thermal energy into mechanical energy Heat Engine Classified according to whether combustion takes place outside the engine or inside the engine Heat Engine External combustion engine Fuel is burned outside the engine. Steam engine Heat Engine Internal combustion engine Fuel is burned in cylinders inside the engine Automobiles