Thermodynamics and Heat Powered Cycles
... This integrated, engineering textbook is the result of fourteen semesters of CyclePad usage and evaluation of a course designed to exploit the power of the software, and to chart a path that truly integrates the computer with education. The primary aim is to give students a thorough grounding in bot ...
... This integrated, engineering textbook is the result of fourteen semesters of CyclePad usage and evaluation of a course designed to exploit the power of the software, and to chart a path that truly integrates the computer with education. The primary aim is to give students a thorough grounding in bot ...
Role of the Sarcoplasmic Reticulum Ca2+-ATPase
... Ca2+ concentration is high inside the reticulum and low in the cytosol and the pump operates forward and backwards, cleaving and synthesizing ATP continuously. In the bibliography, the simultaneous synthesis and hydrolysis of ATP measured in steady state conditions is referred to as the ATP ↔Pi exch ...
... Ca2+ concentration is high inside the reticulum and low in the cytosol and the pump operates forward and backwards, cleaving and synthesizing ATP continuously. In the bibliography, the simultaneous synthesis and hydrolysis of ATP measured in steady state conditions is referred to as the ATP ↔Pi exch ...
application notes - Endot Industries
... Endot produces three types of polyethylene water pipe, ID controlled pipe, manufactured to ASTM specification D2239, commonly called IPS (Iron Pipe Size) pipe, OD controlled IPS pipe manufactured to ASTM D3035 and ASTM D2447, and OD controlled tubing manufactured to ASTM specification D2737, known a ...
... Endot produces three types of polyethylene water pipe, ID controlled pipe, manufactured to ASTM specification D2239, commonly called IPS (Iron Pipe Size) pipe, OD controlled IPS pipe manufactured to ASTM D3035 and ASTM D2447, and OD controlled tubing manufactured to ASTM specification D2737, known a ...
CHAPTER 6 THERMOCHEMISTRY
... a. The ∆H value for a reaction is specific to the coefficients in the balanced equation. Because the coefficient in front of H2O is a 2, 891 kJ of heat is released when 2 mol of H2O is produced. For 1 mol of H2O formed, 891/2 = 446 kJ of heat is released. b. 891/2 = 446 kJ of heat released for each ...
... a. The ∆H value for a reaction is specific to the coefficients in the balanced equation. Because the coefficient in front of H2O is a 2, 891 kJ of heat is released when 2 mol of H2O is produced. For 1 mol of H2O formed, 891/2 = 446 kJ of heat is released. b. 891/2 = 446 kJ of heat released for each ...
Anti - Corrosion Management, Environment and Quality at the Oil
... Many corrosion problems have occurred with related ecological damage, during the 90 years of the existence of the oil refining industry. Many corrosion problems have been solved. Some of them have not. Why? Sometimes corrosion damages occur at certain facilities, but nor at others. There are many co ...
... Many corrosion problems have occurred with related ecological damage, during the 90 years of the existence of the oil refining industry. Many corrosion problems have been solved. Some of them have not. Why? Sometimes corrosion damages occur at certain facilities, but nor at others. There are many co ...
Chapter 7 ENTROPY
... energy changes are negligible. 3 Air is an ideal gas. 4 The process involves no internal irreversibilities such as friction, and thus it is an isothermal, internally reversible process. Properties Noting that h = h(T) for ideal gases, we have h1 = h2 since T1 = T2 = 25°C. Analysis We take the compre ...
... energy changes are negligible. 3 Air is an ideal gas. 4 The process involves no internal irreversibilities such as friction, and thus it is an isothermal, internally reversible process. Properties Noting that h = h(T) for ideal gases, we have h1 = h2 since T1 = T2 = 25°C. Analysis We take the compre ...
Photothermal effects in micro/nano electromechanical systems
... sensors to obtain spectroscopic information from trace amounts of mass (≈ fg) and weak IR absorbing analytes such as liquids. To achieve this goal, two types of unexplored micro/nano mechanical resonators have been investigated; namely, a novel design of biii ...
... sensors to obtain spectroscopic information from trace amounts of mass (≈ fg) and weak IR absorbing analytes such as liquids. To achieve this goal, two types of unexplored micro/nano mechanical resonators have been investigated; namely, a novel design of biii ...
Geothermal
... Even with minimal exploration activity between 1985 and 2000, understanding of extensional geothermal systems has changed, especially regarding deep fluid circulation in the absence of magmatism. In particular, recognition of temperatures exceeding 285°C in Dixie Valley (Blackwell et al., 2000), as ...
... Even with minimal exploration activity between 1985 and 2000, understanding of extensional geothermal systems has changed, especially regarding deep fluid circulation in the absence of magmatism. In particular, recognition of temperatures exceeding 285°C in Dixie Valley (Blackwell et al., 2000), as ...
Thermal Modeling and Management of Discrete Surface
... as notebook computers and cellular phones, the designers are faced with new issues and challenges as to “how to get the heat out.” Although, thermal management of power semiconductors has always been an issue, the problem of higher power in smaller packages is a major issue more than ever and will c ...
... as notebook computers and cellular phones, the designers are faced with new issues and challenges as to “how to get the heat out.” Although, thermal management of power semiconductors has always been an issue, the problem of higher power in smaller packages is a major issue more than ever and will c ...
2002 Lyman ETFS
... Louvered fins, typically found in many compact heat exchanger designs, increase the average heat transfer by interrupting the boundary layer formation and by providing more surface area. In contrast to a continuous fin, which allows an uninterrupted growth of the boundary layer, a louvered fin provi ...
... Louvered fins, typically found in many compact heat exchanger designs, increase the average heat transfer by interrupting the boundary layer formation and by providing more surface area. In contrast to a continuous fin, which allows an uninterrupted growth of the boundary layer, a louvered fin provi ...
MASSACHUSETTS INSTITUTE NUCLEAR ENGINEERING OF TECHNOLOGY by
... and breeding material because of its low melting point, high boiling point, low vapor pressure, low density, high heat capacity, high thermal conductivity, and low viscosity. ...
... and breeding material because of its low melting point, high boiling point, low vapor pressure, low density, high heat capacity, high thermal conductivity, and low viscosity. ...
Unsteady coupled convection, conduction and radiation simulations
... A.3 The PALM units for multi-physics . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 252 A.3.1 The unit AVBP . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 252 A.3.2 The unit PRISSMA . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . ...
... A.3 The PALM units for multi-physics . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 252 A.3.1 The unit AVBP . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 252 A.3.2 The unit PRISSMA . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . ...
Unsteady coupled convection, conduction and radiation simulations
... A.3 The PALM units for multi-physics . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 252 A.3.1 The unit AVBP . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 252 A.3.2 The unit PRISSMA . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . ...
... A.3 The PALM units for multi-physics . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 252 A.3.1 The unit AVBP . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 252 A.3.2 The unit PRISSMA . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . ...
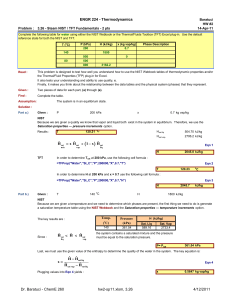
hw2-sp11 - Learn Thermo HOME
... The key to determining properties for state 1 is that we know both the pressure and temperature. So, we can determine every other property, including the specific volume and internal energy. The key for states 2a and 2b is whether the piston has fallen all the way down to rest on the stops or not. S ...
... The key to determining properties for state 1 is that we know both the pressure and temperature. So, we can determine every other property, including the specific volume and internal energy. The key for states 2a and 2b is whether the piston has fallen all the way down to rest on the stops or not. S ...
Refrigeration and Air- Conditioning
... Assertion (A): The coefficient of performance of a heat pump is greater than that for the refrigerating machine operating between the same temperature limits.[IES-2002; IAS-2002] Reason (R): The refrigerating machine requires more energy for working where as a heat pump requires less. (a)Both A and ...
... Assertion (A): The coefficient of performance of a heat pump is greater than that for the refrigerating machine operating between the same temperature limits.[IES-2002; IAS-2002] Reason (R): The refrigerating machine requires more energy for working where as a heat pump requires less. (a)Both A and ...
chapter 1
... 6-22E Steam is leaving a pressure cooker at a specified pressure. The velocity, flow rate, the total and flow energies, and the rate of energy transfer by mass are to be determined. Assumptions 1 The flow is steady, and the initial start-up period is disregarded. 2 The kinetic and potential energies ...
... 6-22E Steam is leaving a pressure cooker at a specified pressure. The velocity, flow rate, the total and flow energies, and the rate of energy transfer by mass are to be determined. Assumptions 1 The flow is steady, and the initial start-up period is disregarded. 2 The kinetic and potential energies ...
Numerical Simulation of Residual Stress and Strain
... Welding, among all mechanical joining processes, has been employed at an increasing rate for its advantages in design flexibility. In addition to that, cost savings, reduced overall weight and enhanced structural performance. The highly localized transient heat and strongly nonlinear temperature fie ...
... Welding, among all mechanical joining processes, has been employed at an increasing rate for its advantages in design flexibility. In addition to that, cost savings, reduced overall weight and enhanced structural performance. The highly localized transient heat and strongly nonlinear temperature fie ...
A Heat Transfer Textbook by John H. Lienhard IV and John H
... Verne’s description in Around the World in Eighty Days in which, to win a race, a crew burns the inside of a ship to power the steam engine. The combustion of nonrenewable fossil energy sources (and, more recently, the fission of uranium) has led to remarkably intense energy releases in power-generat ...
... Verne’s description in Around the World in Eighty Days in which, to win a race, a crew burns the inside of a ship to power the steam engine. The combustion of nonrenewable fossil energy sources (and, more recently, the fission of uranium) has led to remarkably intense energy releases in power-generat ...
Thermodynamics Demystified
... students to develop their problem-solving skills. All examples and problems are presented using SI metric units. English-unit equivalents are given in App. A. The mathematics required to solve the problems is that used in several other engineering courses. The more-advanced mathematics is typically ...
... students to develop their problem-solving skills. All examples and problems are presented using SI metric units. English-unit equivalents are given in App. A. The mathematics required to solve the problems is that used in several other engineering courses. The more-advanced mathematics is typically ...
agroekologi temperatur tanah
... http://info.cycadpalm.com/bid/57663/How-to-Fertilize-Cycads-Part-2 diunduh 15/2/2012 ...
... http://info.cycadpalm.com/bid/57663/How-to-Fertilize-Cycads-Part-2 diunduh 15/2/2012 ...
Protection against cold in prehospital care: thermal insulation
... move the patient into shelter, remove wet clothing if possible, insulate the patient from the ground, and contain endogenous heat production within adequate wind- and waterproof insulation ensembles. In addition, depending on the victim’s physiological status, available resources, and expected evacu ...
... move the patient into shelter, remove wet clothing if possible, insulate the patient from the ground, and contain endogenous heat production within adequate wind- and waterproof insulation ensembles. In addition, depending on the victim’s physiological status, available resources, and expected evacu ...
2007 Lynch TE
... presented results of endwall heat transfer influenced by secondary flows. Measurements of heat transfer coefficients indicated regions of high heat transfer at the blade leading edge, and adjacent to the suction side of the airfoil downstream of the passage throat. All of the investigators attribute ...
... presented results of endwall heat transfer influenced by secondary flows. Measurements of heat transfer coefficients indicated regions of high heat transfer at the blade leading edge, and adjacent to the suction side of the airfoil downstream of the passage throat. All of the investigators attribute ...
Thermodynamics
... Concept of Continuum The concept of continuum is a kind of idealization of the continuous description of matter where the properties of the matter are considered as continuous functions of space variables. Although any matter is composed of several molecules, the concept of continuum assumes a conti ...
... Concept of Continuum The concept of continuum is a kind of idealization of the continuous description of matter where the properties of the matter are considered as continuous functions of space variables. Although any matter is composed of several molecules, the concept of continuum assumes a conti ...
Heat recovery with hybrid ventilation in office
... Netherlands have to fulfil the demands of Dutch government. The goal of the Dutch government is to create almost zero energy buildings at around 2018 (Agentschap NL, 2013). Not only in the production of energy but also in the consumption of energy. One of the main energy users in the building is the ...
... Netherlands have to fulfil the demands of Dutch government. The goal of the Dutch government is to create almost zero energy buildings at around 2018 (Agentschap NL, 2013). Not only in the production of energy but also in the consumption of energy. One of the main energy users in the building is the ...