* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download atoms
Survey
Document related concepts
Transcript
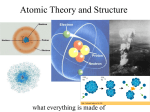
Atomic Theory Ancient Greeks were the first to think about the nature of matter Democritus proposed the idea of matter as small pieces Atom: the smallest particle of an element As more evidence was collected over time, the theory and models were revised Atomic Theory Dalton’s Atomic Theory: atoms were like smooth, hard spheres that could not be broken into smaller pieces All elements are composed of atoms that cannot be divided Atoms cannot be created or destroyed in a chemical change Compounds are composed of more than one element Atomic Theory Thompson found smaller parts of atoms--atoms contain negatively charged particles (electrons) e Berries in a muffin Rutherford and the Nucleus: Gold Foil Experiment A few particles deflected strongly Some bounced back!! Neutrons (no charge): located in center of atom Protons (+): positively charged particles inside the nucleus Atomic Theory Bohr’s Model: showed electrons could only have specific amounts of energy, leading them to move in certain orbits Electron Cloud Model: According to this model, the electrons move rapidly in every direction around the nucleus The Modern Atomic Model The Modern Atomic Model: Chadwick Discovered Neutron= same mass as proton Describes an atom as consisting of a nucleus that contains protons and neutrons surrounded by electrons Particle Charges: Protons charge is + Electrons charge is – # of protons = # of electrons making the atom neutral # of neutrons does not always match #of P+ and ebecause they have no charge The Modern Atomic Model Comparing Particle Mass: atomic mass unit= amu (1 proton is one amu) Protons and neutrons make up nearly the entire mass of the atom Scale and Size of Atoms The tiniest visible speck of dust may contain 10 million billion atoms The Modern Atomic Model Atomic Number # of protons in the nucleus of an atom Identifies an element Isotopes and Mass Number Isotopes: different number of neutrons Mass number: sum of protons and neutrons Model of an Atom