* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download What are Valence Electrons
Survey
Document related concepts
Transcript
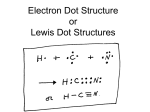
11/4 & 11/5 - 8th Grade Agenda Learning Objective: Valence Electrons and Bonding Collect HW: Reading & Notetaking: p.86 – 87 Chap 4 Test Textbook Reading p.176-182 Reading & Notetaking p.90 – 91 Reminder: Elemental Super Hero Project Due:11/13 (15 Points) Daily Reflection • Prompt: Write 3 ways that we use Radio active elements. Atomic Number tells the number of P___________ • Number of protons (or electrons) in the nucleus of an atom. As we move left to right and top to bottom in the periodic table, the atomic number _________ • Increases What does an atom consist of? • Protons and Neutrons in the nucleus • Surrounded by one or more electrons What charge does a proton have? • Positive Charge What charge does a neutron have? • Neutral charge What are the outer most electrons called? • Valence Electrons What are Valence Electrons • Valence electrons are electrons that have the highest en______ ergy level and are held most loosely ber of • The num______ valence electrons in an atom of an element determines its perties and the pro________ ways it can bond with other atoms In this picture, what is happening to valence electrons? • In a chemical bond, electrons may be transferred from one atom to another or they may be shared between the atoms Electron Dot Diagram • There can be between 1 to 8 Valence elect________. rons • Electron dot diagram includes the symbol for element surr_________ ounded by dots. Electron Dot Diagram How many valence electron in Sulfur? •6 How many valence electrons are needed to make atoms happy? • Atoms are happiest when they have 8 electron in their valence shell. • If the atom’s valence shell happens to be the first shell (hydrogen or Helium) 2 electrons in that shell make them happy Chemical Bond • Atom react in a way that makes each atom more sta______. ble • Number of valence electrons increase to 8 (or 2 for hydrogen) • Chemical bond is the force of attraction that holds two atoms together as a result of rearr___________ angement of electrons between them. Number of Valence Electrons determined by place in the Periodic Table • For elements in groups 1-2 and 13-18, the rons number of valence elect_________ is easy to tell directly from the periodic table. • For elements in group 3-12, determining the number of valence electrons is more compli__________. cated How many Valence electrons does it have?