* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Atom QuizO
Molecular Hamiltonian wikipedia , lookup
Mössbauer spectroscopy wikipedia , lookup
Degenerate matter wikipedia , lookup
Particle-size distribution wikipedia , lookup
Atomic orbital wikipedia , lookup
Electron scattering wikipedia , lookup
Chemical bond wikipedia , lookup
Electron configuration wikipedia , lookup
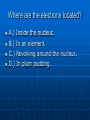
Atom Quiz Olivia, Julia, Jaclyn, Kelly What is a proton? A.) a subatomic particle with no charge B.) a subatomic particle with a negative charge C.) A subatomic particle that has a positive charge that revolves around the nucleus D.) A subatomic particle that has a positive charge that is located in the nucleus What is an electron? A.) A subatomic particle that revolves around the nucleus with a negative charge B.) An atom of the same element that have a different number of neutrons C.) An element that has a negative charge D.) A subatomic particle that has no charge What is a neutron? A.) A element with a positive charge B.) A subatomic particle in the nucleus with no charge C.) A element with no charge and in the nucleus D.) An ingredient used in Pepsi. Where are the electrons located? A.) Inside the nucleus. B.) In an element. C.) Revolving around the nucleus. D.) In plum pudding. How do you find the mass number? A.) You subtract the atomic mass from the number of protons. B.) It is the number of protons. C.) You add the number of protons and neutrons together. D.) You subtract neutrons from electrons What is the atomic number for Thulium? A.) 65 B.) 69 C.) 176 D.) None of the above What is one thing atoms of the same element share? A.) The same atomic number B.) The same shape C.) Julia Berv D.) The same school What can be used to see individual atoms? A.) binoculars B.) 3-D glasses C.) times 500 microscope D.) scanning tunneling microscope What scientist did the gold foil experiment? A.) Darwin B.) Rutherford C.) Thomson D.) Dr. Chrisophy How are ions formed? A.) by adding the electrons to the protons B.) subtracting the protons from the neutrons C.) by atoms gaining or losing electrons D.) by combining two atoms. Your score 10 right- you are a genius 8 right- you are extremely smart 6 right- You are doing okay 4 right – Keep trying! 2 right- You need to study more 0 right- Good Luck passing chemistry!