Influence of Hydrogen Atoms on the Growth of carbon based
... The flowing afterglow of the second discharge is then used to enhance artificially and in a controllable way the flux of added hydrogen atoms. During this work two different setups of double plasma experiments are used. In the first experimental setup a microwave (MW) expanding plasma at 2.45 GHz is ...
... The flowing afterglow of the second discharge is then used to enhance artificially and in a controllable way the flux of added hydrogen atoms. During this work two different setups of double plasma experiments are used. In the first experimental setup a microwave (MW) expanding plasma at 2.45 GHz is ...
Barrier-free intermolecular proton transfer induced by excess
... one-parameter-method for kinetics 共MPW1K兲 designed by Truhlar et al.28 In both DFT approaches we used the same 6-31⫹⫹G** basis set.29,30 Five d functions were used on heavy atoms. The usefulness of the B3LYP/6-31⫹⫹G** method to describe intra- and intermolecular hydrogen bonds has been demonstrated ...
... one-parameter-method for kinetics 共MPW1K兲 designed by Truhlar et al.28 In both DFT approaches we used the same 6-31⫹⫹G** basis set.29,30 Five d functions were used on heavy atoms. The usefulness of the B3LYP/6-31⫹⫹G** method to describe intra- and intermolecular hydrogen bonds has been demonstrated ...
J. Phys. Chem. 1993,97, 2618
... transfer process is not a function of the solvent relaxation dynamics, but rather is more sensitive to energetic factors of the solute/solvent interactions. The hydrogen bonding effect of protic solventson the proton transfer of 3 H F has been recently extended by Harris et al.17based on femtosecond ...
... transfer process is not a function of the solvent relaxation dynamics, but rather is more sensitive to energetic factors of the solute/solvent interactions. The hydrogen bonding effect of protic solventson the proton transfer of 3 H F has been recently extended by Harris et al.17based on femtosecond ...
PDF w - ACS Publications - American Chemical Society
... conclusion that the rates of these HAT reactions correlate with © 2014 American Chemical Society ...
... conclusion that the rates of these HAT reactions correlate with © 2014 American Chemical Society ...
Selective Gas-Phase Cleavage at the Peptide
... completion of the Human Genome Project,11 MS/MS studies of peptides/proteins based on correlation analysis using DNA/ protein databases could become even more powerful tools in the future. Note that existing algorithms have already taken advantage of the knowledge regarding peptide fragmentation gai ...
... completion of the Human Genome Project,11 MS/MS studies of peptides/proteins based on correlation analysis using DNA/ protein databases could become even more powerful tools in the future. Note that existing algorithms have already taken advantage of the knowledge regarding peptide fragmentation gai ...
Vibrational signatures of Hydrogen bonding in the protonated
... the shared proton (νz) and the symmetric stretching motion of the NH3 subunits (νR) motion, at 743 cm-1. Further, they did find a reasonable agreement of the band positions between the infrared multiple photon dissociation (IRMPD) spectrum of the bare ion and the IR vibrational predissociation spec ...
... the shared proton (νz) and the symmetric stretching motion of the NH3 subunits (νR) motion, at 743 cm-1. Further, they did find a reasonable agreement of the band positions between the infrared multiple photon dissociation (IRMPD) spectrum of the bare ion and the IR vibrational predissociation spec ...
Proton Chemical Shift Tensors and Hydrogen Bond Geometry: A 1H
... chemistry, biochemistry, and biology.1-5 Due to its versatility and applicability to molecular systems in all condensed phases, nuclear magnetic resonance (NMR) spectroscopy is commonly used for studying hydrogen-bonding phenomena. The early discovery of the effect of hydrogen bonding on 1H chemical ...
... chemistry, biochemistry, and biology.1-5 Due to its versatility and applicability to molecular systems in all condensed phases, nuclear magnetic resonance (NMR) spectroscopy is commonly used for studying hydrogen-bonding phenomena. The early discovery of the effect of hydrogen bonding on 1H chemical ...
Molecular simulation with variable protonation states at constant pH
... correct statistical probability for a system in equilibrium with a bath at constant temperature and pH. The method falls in the second category described above: it alternates sampling over configurations for a given protonation state with Monte Carlo moves attempted between physically meaningful, di ...
... correct statistical probability for a system in equilibrium with a bath at constant temperature and pH. The method falls in the second category described above: it alternates sampling over configurations for a given protonation state with Monte Carlo moves attempted between physically meaningful, di ...
CHM134: General Chemistry I
... 8. 4 pts. A room is 6.00 meters long, 3.00 meters high, and 5.00 meters wide, how many liters of volume does this represent? You must show your work: ...
... 8. 4 pts. A room is 6.00 meters long, 3.00 meters high, and 5.00 meters wide, how many liters of volume does this represent? You must show your work: ...
Ionic Strength and Electrostatic Effects in
... models) are approximated. The model showed good agreement with the experimental data of these authors. The advantage of this approach is its generality concerning the size of the molecule, which makes it particularly suited for oligoelectrolytes that show a behavior intermediate between simple ions ...
... models) are approximated. The model showed good agreement with the experimental data of these authors. The advantage of this approach is its generality concerning the size of the molecule, which makes it particularly suited for oligoelectrolytes that show a behavior intermediate between simple ions ...
Relative Atomic Masses
... What does this tell us? It means that if we have a fixed mass of nitrogen, the mass of oxygen which will combine with it cannot be simply any amount. In fact, the opposite is true. There are a few specific masses of oxygen which will combine with the fixed nitrogen, and those specific masses are int ...
... What does this tell us? It means that if we have a fixed mass of nitrogen, the mass of oxygen which will combine with it cannot be simply any amount. In fact, the opposite is true. There are a few specific masses of oxygen which will combine with the fixed nitrogen, and those specific masses are int ...
Solution
... electrons, four of which are involved in covalent bonding and 4 of which are unshared. We can add to this picture, the fact that oxygen has a higher affinity than hydrogen for electrons. Thus in a water molecule, the electron cloud is denser around the oxygen nucleus than around the hydrogen nuclei. ...
... electrons, four of which are involved in covalent bonding and 4 of which are unshared. We can add to this picture, the fact that oxygen has a higher affinity than hydrogen for electrons. Thus in a water molecule, the electron cloud is denser around the oxygen nucleus than around the hydrogen nuclei. ...
A proton wire to couple aminoacyl-tRNA
... for this water was absent from the Hma 50S-subunit preattack struc- A proton wire connects the attacking amine to W1 ture4, it could nevertheless be modeled as a water molecule in all of In all of the structures presented here, a short network of hydrogen our structures on the basis of the following ...
... for this water was absent from the Hma 50S-subunit preattack struc- A proton wire connects the attacking amine to W1 ture4, it could nevertheless be modeled as a water molecule in all of In all of the structures presented here, a short network of hydrogen our structures on the basis of the following ...
Detection of Organic Pollutants with a Pulsed Ion Mobility
... influence of water clusters. A similar problem exists when using the proton affinity to explain certain observations in ion mobility spectrometers, but there at least a similar tendency has been observed, so we assume a similar tendency with repect to the results of the calculations, but the results ...
... influence of water clusters. A similar problem exists when using the proton affinity to explain certain observations in ion mobility spectrometers, but there at least a similar tendency has been observed, so we assume a similar tendency with repect to the results of the calculations, but the results ...
Solvent effects on excited state relaxation phenomena
... In pure solvents effects of electrostatic forces and of specific short range interactions on the spectral position or photophysical properties are not easily distinguished. Shifts attributed to either of these two categories could add up or cancle dependent on the structure of the soluteaolvent clus ...
... In pure solvents effects of electrostatic forces and of specific short range interactions on the spectral position or photophysical properties are not easily distinguished. Shifts attributed to either of these two categories could add up or cancle dependent on the structure of the soluteaolvent clus ...
Can the (M• – X) region in electron capture dissociation provide
... (45.026 Da) has been attributed by Cooper et al. to the loss of CH3NO (45.022 Da) from the amide groups of the asparagine/glutamine side chains. The KH peptide does contain two asparagines. On the other hand, the 59 Da loss has so far only been associated with arginine and attributed to CH5N3 (59.04 ...
... (45.026 Da) has been attributed by Cooper et al. to the loss of CH3NO (45.022 Da) from the amide groups of the asparagine/glutamine side chains. The KH peptide does contain two asparagines. On the other hand, the 59 Da loss has so far only been associated with arginine and attributed to CH5N3 (59.04 ...
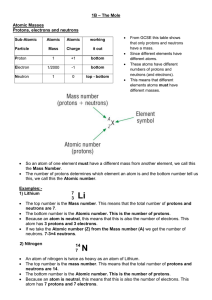
Chapter 5: Chemical Formula Relationships – The Mole Counting by
... energy required to form a stable nucleus, and hold everything together. This is the same nuclear energy which is released in nuclear reactions such as nuclear fission when the nuclei of atoms are broken apart. Note that this nuclear binding energy mass is not a fixed fraction of the atom’s total mas ...
... energy required to form a stable nucleus, and hold everything together. This is the same nuclear energy which is released in nuclear reactions such as nuclear fission when the nuclei of atoms are broken apart. Note that this nuclear binding energy mass is not a fixed fraction of the atom’s total mas ...
Atoms and Molecules
... The chemical formula of a molecular compound is determined by the valency of each element. In ionic compounds, the charge on each ion is used to determine the chemical formula of the compound. Scientists use the relative atomic mass scale to compare the masses of different atoms of elements. Atoms o ...
... The chemical formula of a molecular compound is determined by the valency of each element. In ionic compounds, the charge on each ion is used to determine the chemical formula of the compound. Scientists use the relative atomic mass scale to compare the masses of different atoms of elements. Atoms o ...
Nuclear Magnetic Resonance and Potentiometric
... [9]aneN3, and includes a similar comparison of literature values for the macrocyclic tetraaza ligands DOTA and TETA with [ 12]aneN4and [ 14]aneN4 Although the first protonation constant for NOTA could not be determined in this work by potentiometry, log K,= 11.3 f 0.1 is obtained from the N M R p H ...
... [9]aneN3, and includes a similar comparison of literature values for the macrocyclic tetraaza ligands DOTA and TETA with [ 12]aneN4and [ 14]aneN4 Although the first protonation constant for NOTA could not be determined in this work by potentiometry, log K,= 11.3 f 0.1 is obtained from the N M R p H ...
The Mole
... We can make solutions of known concentration using volumetric flasks. The easiest way of learning this is to try an example. We need 250cm3 of 0.1 mol dm3 solution of sodium hydroxide. Use the formula to calculate the No. of moles of sodium hydroxide – No. of moles = Concentration (Mol dm3) x ...
... We can make solutions of known concentration using volumetric flasks. The easiest way of learning this is to try an example. We need 250cm3 of 0.1 mol dm3 solution of sodium hydroxide. Use the formula to calculate the No. of moles of sodium hydroxide – No. of moles = Concentration (Mol dm3) x ...
WJEC CBAC AS/A LEVEL GCE in Chemistry REVISION AID UNIT 1
... and to forms of leukaemia. Even small increases in the background level of radiation may have significant effects on the population as a whole. This is because the probability for cell mutation is higher when applied to a large population sample. Clusters of childhood leukaemia have been associated ...
... and to forms of leukaemia. Even small increases in the background level of radiation may have significant effects on the population as a whole. This is because the probability for cell mutation is higher when applied to a large population sample. Clusters of childhood leukaemia have been associated ...
CH1 Student Revision Guides pdf
... and to forms of leukaemia. Even small increases in the background level of radiation may have significant effects on the population as a whole. This is because the probability for cell mutation is higher when applied to a large population sample. Clusters of childhood leukaemia have been associated ...
... and to forms of leukaemia. Even small increases in the background level of radiation may have significant effects on the population as a whole. This is because the probability for cell mutation is higher when applied to a large population sample. Clusters of childhood leukaemia have been associated ...
Amino Acid Composition and Wavelength Effects in Matrix
... complexes were examined as a function of their configuration. Investigation of the optimized geometries allowed the location of bifurcated and linear hydrogen bonds between the non-polar analyte (e.g., Gly-Gly) and matrix molecules. In addition to these intermolecular bonds, in the case of His-His a ...
... complexes were examined as a function of their configuration. Investigation of the optimized geometries allowed the location of bifurcated and linear hydrogen bonds between the non-polar analyte (e.g., Gly-Gly) and matrix molecules. In addition to these intermolecular bonds, in the case of His-His a ...
Proton
The proton is an elementary subatomic particle, symbol p or p+, with a positive electric charge of +1e elementary charge and mass slightly less than that of a neutron. Protons and neutrons, each with mass approximately one atomic mass unit, are collectively referred to as ""nucleons"". One or more protons are present in the nucleus of an atom. The number of protons in the nucleus is referred to as its atomic number. Since each element has a unique number of protons, each element has its own unique atomic number. The word proton is Greek for ""first"", and this name was given to the hydrogen nucleus by Ernest Rutherford in 1920. In previous years Rutherford had discovered that the hydrogen nucleus (known to be the lightest nucleus) could be extracted from the nuclei of nitrogen by collision. The proton was therefore a candidate to be a fundamental particle and a building block of nitrogen and all other heavier atomic nuclei.In the modern Standard Model of particle physics, the proton is a hadron, and like the neutron, the other nucleon (particle present in atomic nuclei), is composed of three quarks. Although the proton was originally considered a fundamental particle, it is composed of three valence quarks: two up quarks and one down quark. The rest masses of the quarks contribute only about 1% of the proton's mass, however. The remainder of the proton mass is due to the kinetic energy of the quarks and to the energy of the gluon fields that bind the quarks together. Because the proton is not a fundamental particle, it possesses a physical size; the radius of the proton is about 0.84–0.87 fm.At sufficiently low temperatures, free protons will bind to electrons. However, the character of such bound protons does not change, and they remain protons. A fast proton moving through matter will slow by interactions with electrons and nuclei, until it is captured by the electron cloud of an atom. The result is a protonated atom, which is a chemical compound of hydrogen. In vacuum, when free electrons are present, a sufficiently slow proton may pick up a single free electron, becoming a neutral hydrogen atom, which is chemically a free radical. Such ""free hydrogen atoms"" tend to react chemically with many other types of atoms at sufficiently low energies. When free hydrogen atoms react with each other, they form neutral hydrogen molecules (H2), which are the most common molecular component of molecular clouds in interstellar space. Such molecules of hydrogen on Earth may then serve (among many other uses) as a convenient source of protons for accelerators (as used in proton therapy) and other hadron particle physics experiments that require protons to accelerate, with the most powerful and noted example being the Large Hadron Collider.