* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Rutherford`s Gold Foil Experiment
Standard Model wikipedia , lookup
Photoelectric effect wikipedia , lookup
Double-slit experiment wikipedia , lookup
Introduction to quantum mechanics wikipedia , lookup
Compact Muon Solenoid wikipedia , lookup
Nuclear structure wikipedia , lookup
Elementary particle wikipedia , lookup
Electric charge wikipedia , lookup
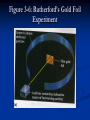
Figure 3-4: Cathode Ray Tube Figure 3.5: Cathode Ray Experiment Found… 1. An object in the tube casts a shadow 2. The wheel rolled from cathode to anode 3. Rays were deflected by a magnetic field like an electric current, known to be negative 4. Rays were deflected from a negative charge. Conclusions: the cathode ray is made of electrons! Electrons have a negative charge Electrons are present in atoms of all elements Millikan Oil Drop Experiment Millikan Oil Drop Experiment Millikan was able to determine the exact charge of an electron by suspending oil droplets in an electric field By determining the mass of the oil droplet he could see the charge required to suspend the drops were discreet multiples of a specific charge. Confirmed electrons are negative Showed they have a tiny mass compared to their large charge J. J. Thomson (cathode ray) 1904 – Model of the atom…figured since the atom is neutral there must be something positive to balance out the negative Called the “Plum Pudding Model” A positive cloud with negative electrons within it. Figure 3-6: Rutherford’s Gold Foil Experiment Figure 3-6: Rutherford’s Gold Foil Experiment Bombarded thin gold foil with alpha particles and expected the particles to pass through. BUT…. Figure 3-6: Rutherford’s Gold Foil Experiment Figure 3-6: Rutherford’s Gold Foil Experiment Particles were deflected greatly and some went straight back to the source Conclusion: the volume of the nucleus is very small compared to the volume of the atom Figure 3-7: Rutherford’s Gold Foil Experiment Ernest Rutherford Rutherford determined there was a small, dense nucleus in the atom because some of the particles were deflected. 1911 – Model of atom includes a nucleus surrounded by electrons Section 3.2 Composition of the Atomic Nucleus Atomic nuclei are made of protons and neutrons Rutherford is typically credited with the discovery of the proton in 1919. James Chadwick is credited with the discovery of the neutron in 1932 Proton has a positive charge equal to the magnitude of the negative charge of the electron. Section 3.2 Simplest hydrogen atom is simply a proton with an electron orbiting. Proton mass is 1836 times greater than the mass of the electron. The number of protons in the nucleus determines the identity of the element and is the atomic number. Section 3.2 Forces in the Nucleus Generally, particles with the same electric charge repel each other However, when two protons are extremely close to each other, there is a strong attraction between them. Nuclear forces: these short-range proton-neutron, proton-proton, and neutron-neutron forces hold the nucleus together. Section 3.2 Forces in the Nucleus http://www.youtube.com/watch?v=Imvbu8aJHHk Section 3.2 The Sizes of Atoms The region occupied by the electrons is an electron cloud. Atomic radius is the distance from the nucleus to the outer portion of the electron cloud. Atomic radii are expressed in picometers ( 1 pm = 10-12 m) Atomic range is 40 to 270 pm. Robert Frost American Poet (1874-1963) Typically wrote pastoral poems about rural life Fireflies in the Garden Here come real stars to fill the upper skies, And here on earth come emulating flies, That though they never equal stars in size, (And they were never really stars at heart) Achieve at times a very star-like start. Only, of course, they can’t sustain the part Robert Frost American Poet (1874-1963) Typically wrote pastoral poems about rural life Very interested in science Curious about the structure of the atom Read Scientific American Attended lectures atomic structure and quantum physics by the chemist Niels Bohr