* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download environmental science & services - level 1
Survey
Document related concepts
Surveys of scientists' views on climate change wikipedia , lookup
Scientific opinion on climate change wikipedia , lookup
Fred Singer wikipedia , lookup
Global Energy and Water Cycle Experiment wikipedia , lookup
Instrumental temperature record wikipedia , lookup
Global commons wikipedia , lookup
Transcript
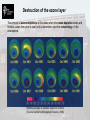
Low Carbon Buildings and Sustainability By Dr David Johnston – licensed under the Creative Commons Attribution – Non-Commercial – Share Alike License http://creativecommons.org/licenses/by-nc-sa/2.5/ ENVIRONMENTAL SCIENCE & SERVICES LEVEL 1 Introduction Dr. David Johnston Module aim The aim of this module is to: Develop an understanding of the scientific principles involved in the environmental design of buildings, together with the provision of building services systems. Learning outcomes and competencies On completion of this module, students should be able to: Define and use appropriate technical and scientific terminology. Identify the basic principles of environmental design. Apply these basic principles to the design of simple elements to meet environmental requirements within the current statutory framework. Describe different types of building services installations applied to a range of building types and the principles involved in their design. Module content The module content will encompass the following subjects: Global Environmental Issues The Impact of Buildings on the Environment Thermal Comfort Heat Loss and Thermal Performance of Buildings Thermal Modelling of Buildings Condensation Acoustics Energy Resources and Supply CHP and District Heating Systems Space Heating Systems Hot Water Supply Systems Active and Passive Solar Systems Natural and Mechanical Ventilation Air Conditioning Natural and Artificial Lighting Key skills In addition to developing subject specific knowledge and understanding, the module will also help you to develop a number of generic key skills. Specific key skills which this module will help to develop are outlined below. ● Technical Skills (developed throughout module and assessed in end examination). ● Theory and principles (developed throughout module and assessed in phase examination and end examination). ● Analysis and Reflection (developed throughout module and assessed in end examination). ● Application and Reflection (developed throughout module and assessed in end examination). ENVIRONMENTAL SCIENCE & SERVICES LEVEL 1 Impact of buildings on the global climate Dr. David Johnston Objectives By the end of this lecture you should be able to: Identify the ways in which the built environment can have an adverse effect on the natural environment. Identify the ways in which the built environment can contribute to global atmospheric pollution. Understand the processes involved in: the greenhouse effect. the destruction of the ozone layer. acid deposition. Identify the main contributors to these processes. Introduction Introduction Buildings can have a number of adverse effects on the natural environment. These include: Depletion of natural resources – Over 90% of non-energy minerals (mainly aggregates) extracted in the UK are supplied as construction materials. The construction and demolition industry contributes 33% to the volume of the UK’s waste. Changes in land use – Supply of land is limited. Although the emphasis of current Government policy is to build new housing on brownfield land, in 2005 23% of dwellings were built on undeveloped (green) land. This proportion is likely to increase as demand for housing grows. Destruction of habitats and ecosystems – In the last century, over 100 species have been lost and many more species and habitats are in danger of disappearing. Changes to the global climate – Main changes include: Global warming. Destruction of the ozone layer. Acid deposition. Global warming Global climate The greenhouse effect is a natural phenomenon in which short wave radiation emitted from the sun penetrates the Earth's atmosphere and warms the Earth's surface. Infra-red (longwave) radiation is then emitted from the Earth’s surface, some of which is absorbed by gases in the atmosphere (greenhouse gases). This warms the Earth and the lower atmosphere. Short wave radiation Natural greenhouse gases: Infra-red radiation •Water vapour •CO2 •CH4 The greenhouse effect [Source: Royal Commission on Environmental Pollution, 2000] The ‘natural’ greenhouse effect keeps the Earth 33OC warmer than it would otherwise be, at an average of 15OC, allowing life as we know it on Earth to exist. In contrast, the moon has an average surface temperature of -15OC. Global climate Over the last 250 years, various anthropogenic (human generated) activities have altered the composition of the global atmosphere mainly as a direct result of industrial activity. The release of these gases has led to an increase in the concentrations of the earth’s greenhouse gases. Therefore, more of the infra-red radiation emitted by the Earth’s surface is being trapped. Anthropogenic activities have led to increased concentrations of greenhouse gases. The greenhouse effect [Source: Royal Commission on Environmental Pollution, 2000] This change has been commonly referred to as the ‘enhanced’ greenhouse effect. Anthropogenic greenhouse gases The balance of current scientific opinion is that changes to the global climate are taking place, and these changes are taking place primarily due to the increase in the production of anthropogenic greenhouse gases As anthropogenic greenhouse gas concentrations in the atmosphere increase, more heat is trapped in the Earth’s atmosphere resulting in the enhanced greenhouse effect and global warming. The IPCC stated in their Fourth Assessment Report ‘Climate Change 2007’ that: “Most of the observed increase in global average temperatures since the mid20th century is very likely due to the observed increase in anthropogenic greenhouse gas concentrations” (IPCC, 2007 p.10). Comparison of observed continental- and globalscale changes in surface temperature [Source: IPCC, 2007] Greenhouse gases Greenhouse gases are gases that have a large influence on the greenhouse effect. The most important greenhouse gases are : Water vapour – causes about 36-70% of the greenhouse effect. Carbon dioxide (CO2) – causes about 9-26% of the greenhouse effect. Methane (CH4) – causes about 4-9% of the greenhouse effect. Ozone – causes about 3-7% of the greenhouse effect. Other powerful greenhouse gases include: Nitrous oxide (N2O). Sulphur hexafluoride (SF6). Chlorofluorocarbons (CFCs). Hydrofluorocarbons (HFCs). Hydrochlorofluorocarbons (HCFCs). Perfluorocarbons (PFCs). Related bromide compounds. Many Volatile Organic Compounds (VOCs). Some of these gases occur naturally, some occur only as a result of human activity, and others occur both naturally and as a result of human activity. Greenhouse gases The ability of each greenhouse gas to contribute to global warming varies considerably and is dependent upon: Atmospheric lifetime – how many years the gas remains in the atmosphere. Global Warming Potential (GWP) – a relative measure of how effective the gas is at preventing the passage of infra-red radiation. Atmospheric concentration – usually measured in parts per million (ppm). The GWPs1 for some of the most important greenhouse gases are as follows: Carbon dioxide (CO2) Methane (CH4) Nitrous oxide (N2O) CFC-12 HCFC-22 1 Time Horizon 20 years 100 years 1 1 62 23 275 296 7900 8500 4300 1700 500 years 1 7 156 4200 520 GWP is the ratio of global warming from one unit mass of a greenhouse gas to that of one unit mass of CO2 over a set time period. Greenhouse gases Water vapour is the most important greenhouse gas, although its concentration in the atmosphere is not directly influenced by human activity. However, its concentration is influenced indirectly as any increase in global temperatures will increase concentrations, as warmer air can hold more water. Although CO2 is not the most potent greenhouse gas, it is causing the greatest concern, due to: Its vast concentration in the atmosphere – Concentration has increased sharply due to human activities. Its long atmospheric lifetime – CO2 remains in the atmosphere for 50-200 years. 95% of CO2 emissions come from the combustion of fossil fuels. The combustion of fossil fuels releases CO2 into the atmosphere which had been previously been fixed by the biosphere over many millions of years, and thus increases the concentration of CO2 in the atmosphere.. CO2 emissions contribute around 70% of the potential global warming effect of anthropogenic emissions of greenhouse gases. UK greenhouse gas emissions In 2006, UK emissions of the basket of six greenhouse gases covered by the Kyoto Protocol were estimated to be equivalent to 653 million tonnes of carbon dioxide. UK total greenhouse gas emissions [Source: DEFRA, 2009] Emissions have reduced by 15% over the period 1990 to 2006. Emissions in 2006 were 0.5% less than they were in 2005. In the UK, CO2 is the main greenhouse gas, accounting for around 85% of total greenhouse gas emissions in 2006. UK greenhouse gas emissions CO2 emissions by source: 1990-2006 [Source: DEFRA, 2009] NO2 emissions by source: 1990-2006 [Source: DEFRA, 2009] Methane emissions by source: 1990-2006 [Source: DEFRA, 2009] UK CO2 emissions One of the sectors which contributes substantially to CO2 emissions is the built environment. In the UK, as in most industrialised countries, the domestic sector contributes substantially to national CO2 emissions. There are over 24 million dwellings in the UK, and in 2006 they were responsible for 27% of the UK’s total CO2 emissions by end user (DEFRA, 2009). Increased by 12% over period Reduced by 4% over period CO2 emissions by end use: 1990-2004 [Source: DEFRA, 2009] Important to realise that the UK housing stock is also characterised by long physical lifetimes and slow stock turnover. Global carbon cycle The concentration of CO2 within the atmosphere is the outcome of a natural cycle. CO2 is continuously released into the atmosphere and re-absorbed by rocks, organic material and the oceans. The global carbon cycle [Source: Royal Commission on Environmental Pollution, 2000] Over the last 250 years: Mining and burning of fossil fuels Oceans have absorbed about Forest clearance, changes in land use Net increase is +260GtC -140GtC +50GtC +170GtC Global carbon cycle Over the last 400,000 years, natural variations have occurred in the carbon cycle. The figure below shows how natural variations In CO2 concentration relate to changes in temperature. Changes in CO2 Changes in temperature CO2 concentration and temperature evidence from ice cores [Source: Royal Commission on Environmental Pollution, 2000] All the significant changes that have occurred in the CO2 concentration have been associated with a significant change in temperature. Global carbon cycle Monthly mean atmospheric CO2 concentration at Mauna Loa Observatory, Hawaii. [Source: Earth System research Laboratory, 2009] The present concentration of CO2 in the atmosphere is around 380ppmv, which is much greater than anything that has been recorded over the last half million years. Pre-industrial concentration was around 280ppmv in 1750. Global warming The precise effect of the increase in CO2 concentration on the global climate cannot be predicted with certainty. However, most analyses indicate that it has probably lead to global warming. Combined global land and marine surface temperature record from 1856-2007 [Source: Climatic Research Unit, 2009] 2007 was the eighth warmest year on record, exceeded by 1998, 2005, 2003, 2002, 2004, 2006 and 2001. The 1990s were the warmest complete decade in the series. Twelve of the thirteen warmest years in the series have occurred in the past thirteen years (1995 – 2007). Global warming Model simulations also suggest that the observed increase in the CO2 concentration has probably contributed to most of the observed warming over the last 50 years. Temperature change model simulations using the latest Met Office Hadley Centre model [Source: Met Office, 2009a] Global warming myths Two main global warming myths: Global warming is caused by changes in the Sun’s energy. The intensity of cosmic rays changes the climate by altering cloud cover. Solar activity 1850-2007 [Source: Met Office, 2009a] Changes in solar activity do affect global temperatures, but recent changes in global temperatures cannot be linked to solar activity alone. No evidence that cosmic rays have decreased over the last 30 years resulting in less cloud cover. Effects of global warming We are already experiencing some of the effects of global warming: The number of Category 4 and 5 hurricanes has almost doubled in the last 30 years. Malaria has spread to higher altitudes in places like the Columbian Andes, 7000 feet above sea level. The flow of ice from glaciers in Greenland has more than doubled over the last decade. At least 279 species of plants and animals are already responding to global warming, moving closer to the poles. (An Inconvenient Truth, 2006) Effects of global warming IPCC projections indicate that by the end of the 21st century, global warming is likely to result in: An increase in the globally averaged surface temperature of 1.8 to 4ºC. An increase in global mean sea level of 18 to 59cm. A decrease in snow cover and shrinkage in sea-ice extent in both the Artic and Antarctic. It is likely that future tropical cyclones (typhoons and hurricanes) will become more intense. It is very likely that there will be an increase in the amount of precipitation in high latitudes and likely that precipitation will decrease in most subtropical land regions (by as much as 20% by 2100). Both past and future anthropogenic CO2 emissions will contribute to global warming and sea level rise for more than a millennium, due to the timescales involved. (IPCC, 2007) Predicted temperature rise and sea ice coverage [Source: Met Office, 2009b] Effects of global warming The UK is unlikely to experience some of the more severe effects associated with climate change. Nevertheless, significant changes are still expected to occur: A 2 to 3.5ºC increase in average annual temperature by the 2080s. An increase in the temperature of UK coastal waters. An increase in the frequency of high summer temperatures. An increase in winter rainfall. A rise in the relative sea level around most the UK’s shoreline. An increase in the frequency of extreme sea levels. A weakening of the Gulf Stream. (Hulme, Jenkins, Lu, Turnpenny, Mitchell, Jones, Lowe, Murphy, Hassell, Boorman, McDonald & Hill, 2002) Response to global warming Various attempts have been made to estimate the scale of the reductions that will be required to stabilise atmospheric CO2 concentrations over various time-scales. The estimates vary from approximately a 60% to a 90% reduction in current CO2 emissions by the year 2050. Fossil carbon emissions (GtC/year) 7 Industrialised Countries 6 Developing Countries World 5 4 3 2 1 0 1980 1990 2000 2010 2020 2030 2040 2050 2060 2070 2080 2090 2100 Year Carbon emission rates for the 300GtC global fossil carbon budget [Source: Krause, Bach & Koomey, 1990] Suggest that target should be achieved by dividing the reductions between the industrialised and developing countries. Intended to address the highly asymmetrical distribution of emissions per capita (~11:1). Response to global warming The IPCC have also defined a number of CO2 emission profiles that will result in stabilisation of the atmospheric CO2 concentration. 1100 CO2 concentration (ppm) 1000 900 800 Only emission profile to result in CO2 stabilisation by the end of the 21st century. 700 600 500 400 450 ppm scenario 550 ppm scenario 650 ppm scenario 750 ppm scenario 1000 ppm scenario 300 200 100 0 2000 2050 2100 2150 2200 2250 2300 Year IPCC atmospheric CO2 emission stabilisation scenario [Adapted from: IPCC, 2001] Only the 450ppm/650GtC profile will result in the stabilisation of the atmospheric concentration by the end of the 21st century. Assuming that the reductions are equally divided between the industrialised and the developing countries, CO2 emission reductions of approximately 60% will be required in the industrialised countries by the year 2050. These sorts of reductions have been proposed by the Royal Commission on Environmental Pollution (Royal Commission on Environmental Pollution, 2000). UK’s response to global warming In 1997, the Kyoto Protocol was ratified, committing the UK to a legally binding target of reducing emissions of the six main greenhouse gases by 12.5%, based on 1990 levels, between 2008 and 2012. Following the Kyoto Protocol, the UK Government set a more ambitious target of seeking national reductions in CO2 emissions of 20% by the year 2010. In 2000, the Royal Commission on Environmental Pollution (RCEP) proposed that the UK should reduce its CO2 emissions by 60% by 2050. This was accepted by the UK Government and was used for policy making. The Climate Change Bill was introduced into Parliament in November 2007 and became law (the Climate Change Act) on the 26th November 2008. The Act sets a series of clear targets for reducing UK CO2 emissions. The target for 2050 is at least an 80% reduction in CO2 emissions, based on 1990 levels. Also a reduction in CO2 emissions of at least 34% by 2020. In May 2009, carbon budgets were set for 3 separate time periods: % reduction on 1990 levels 2008-12 22 2013-17 28 2018-22 34 Destruction of the ozone layer The ozone layer Ozone (O3) occurs naturally throughout the atmosphere. The majority of ozone (about 90%) is found in the stratosphere, a region that begins about 10km above the Earth’s surface and extends up to about 50km. Most ozone resides in the stratosphere in what is commonly known as the ozone layer. The remaining ozone, about 10%, is found in the troposphere, the lowest region of the atmosphere. 90% of ozone found in this zone. The Earth’s atmosphere [Source: Centre for Atmospheric Science, 2006] The ozone layer in the stratosphere is thinnest in the tropics (around the equator) and denser towards the poles. The ozone layer Ozone helps to protect human beings, other biological systems and man-made materials from the harmful effects of certain wavelengths of sunlight. The ozone layer in the stratosphere helps to absorbs potentially harmful UV-B radiation (280-315 nm wavelength) coming from the Sun, allowing only reduced amounts to penetrate to the Earth’s surface. UV-A (315-400 nm wavelength) and other solar radiation are not strongly absorbed by the ozone layer. UV protection by the ozone layer [Source: Fahey, 2002] Without the ozone layer, life on Earth would not be possible. The ozone layer The ozone in the stratosphere is created when ultraviolet radiation (sunlight) strikes the stratosphere, dissociating (or splitting) oxygen molecules (O2) to form atomic oxygen (O). The atomic oxygen quickly combines with further oxygen molecules to form ozone (O3): Stratospheric Ozone Production [Source: Fahey, 2002] The amount of ozone above a particular point on the Earth’s surface is measured using Dobson units (DU). Ozone is typically around 260 DU near the tropics and higher elsewhere, although there are seasonal fluctuations. Destruction of the ozone layer Recent evidence indicates that the ozone layer is thinning in certain areas, and ozone ‘holes’ have developed. It is the ozone in the stratosphere over Antarctica that is giving the greatest cause for concern – owing to climatic conditions, the ozone over the Antarctic is particularly susceptible to ozone depletion at certain times of the year. Ozone depletion is most marked in the Antarctic Spring, around October. There is also evidence that the ozone layer is thinning, albeit less extensively, throughout the world. Total Ozone above Halley Bay Station, Antarctica [Source: Centre for Atmospheric Science, 2006] Destruction of the ozone layer TOMS (Total Ozone Mapping Spectrometer) satellite measurements of ozone levels over Antarctica. Changes in Antarctic Ozone Levels [Source: Centre for Atmospheric Science, 2006] Destruction of the ozone layer The amount of ozone depleted and the dates when the ozone depletion starts and finishes varies from year to year and is dependent upon the meteorology of the atmosphere. Monthly averages in Antarctic ozone for October [Source: Centre for Atmospheric Science, 2006] Destruction of the ozone layer Size and depth of this years ozone hole [Source: NASA, 2007] Destruction of the ozone layer The ozone in the stratosphere is continually being created and destroyed by natural chemical reactions and processes that involve sunlight (photochemical). Until recently, this process has been in equilibrium: the amount of ozone created ≈ amount of ozone destroyed However, recent anthropogenically produced catalysts and trace gases have been accumulating in the atmosphere, and are thought to be altering and destroying the ozone layer without any compensating increase in the rate at which it is produced. Gases of particular concern include a group of synthetically produced chlorine containing chemical substances known as CFCs and HCFCs. These have been used for: Refrigeration systems. Air conditioning systems. Aerosol propellants. Solvents. Blowing agents for foams Cleaning of metals and electronic components. Other compounds containing bromide, other related halogen compounds and also nitrogen oxides (NOx) also contribute to ozone depletion. Destruction of the ozone layer CFCs and HCFCs have been used due to a number of important properties: Their chemical stability - they are non-inflammable, non-toxic, non-poisonous. Cost-effective. Non-corrosive. However, CFCs and HCFCs have such long residence times in the atmosphere that only a fraction of the amount released each year is broken down and taken out of circulation by natural processes. Atmospheric lifetimes, emissions and ozone depletion potentials of halogen gas sources [Source: Fahey, 2002] Destruction of the ozone layer Schematic illustrating the lifecycle of CFCs [Source: Centre for Atmospheric Science, 2006] Destruction of the ozone layer When the CFCs and HCFCs are broken down in the stratosphere by sunlight, various products are produced. The most important of these are a number of long-lived inorganic carriers of chlorine; hydrochloric acid (HCl) and chlorine nitrate (ClONO2). Evolution of ClONO2 over North Pole during winter 1994 [Source: Centre for Atmospheric Science, 2006] Destruction of the ozone layer Depletion of stratospheric ozone can lead to damaging effects on human health and the environment: Directly - increased penetration of UV-B radiation. Indirectly - changes in the distribution of stratospheric ozone and water vapour that contribute to global warming effects and altered climatic conditions. The effects of an increase in the amount of UV-B radiation reaching the Earth’s surface are serious: Human health: - Greatest threat is an increase in skin cancer. Each 1% loss of total ozone leads to a 3 to 5% increase in skin cancer cases. - Eye disease, such as cataracts and temporary disorders such as ‘snow blindness’. - UV-B suppresses the immune system. Marine environment: - Likely to be a reduction in the numbers of photoplankton and photosynthesising organisms in the sea. - UV-B causes direct damage to young fish, shrimp and crab larvae, and other small animals. Results in a reduction in food stocks further up the food chain. Agriculture and plant life: - High concentrations of UV radiation can stunt the growth and leaf development of most plant varieties. - UV sensitive plants would be more susceptible to pests and disease. Material degradation: - Useful life of plastics can be reduced when exposed to UV radiation. Destruction of the ozone layer UV radiation at the Earth’s surface that causes sunburn has increased over the globe between 1979 and 1992. Changes in surface UV radiation [Source: Fahey, 2002] Destruction of the ozone layer In response to fears about damage to the ozone layer, the production of ozone depleting gases is regulated under a 1987 international agreement known as the ‘Montreal Protocol on Substances that Deplete the Ozone Layer’ and its subsequent Amendments. The Protocol, now ratified by over 180 nations, establishes legally binding controls on the national production and consumption of ozone depleting gases. Production and consumption of all principal halogen containing gases by developed and developing nations will be significantly phased out before the middle of the 21st century. Effect of the Montreal Protocol [Source: Fahey, 2002] Destruction of the ozone layer Past and future abundance of atmospheric halogen source gases [Source: Fahey, 2002] Acid deposition Acid deposition This is a term used to refer to the various processes by which acidic gases and particles are deposited on land and water. The acidic pollutants reach the earth’s surface via two pathways: Dry deposition – the acidic pollutants are deposited on plants, trees, soils and water surfaces via gases or dust. Combines with fog and dust to produce smog. About half of the acidity in the atmosphere falls back to earth through dry deposition. Wet deposition - when the pollutants fall to the ground through incorporation in rain drops, snow, sleet, mist or fog. Dry and wet deposition [Source: US Environmental Protection Agency, 2006] During the 1980’s, acid rain was a major environmental issue. Acid deposition Acid deposition occurs both naturally and as a result of human activities. Natural Absorption of CO2 by water leading to the formation of mild carbonic acid. Thunderstorms which provide the energy to generate NOX from Nitrogen and Oxygen. Volcanic activity and fires which produce SO2. Anthropogenic The burning of coal and oil for heating, electrical energy generation and transport. Smelting processes. Industrial activities. Main contributors are SOX and NOX from fossil fuel combustion and road transport. Acid deposition Since the emission of pollutants is heavily concentrated in the northern hemisphere, particularly Europe and North America, precipitation is generally acidic in these countries. The distances that the acidic pollutants can travel means that acid deposition is a transboundary problem. For instance, industrial pollution from the UK is blown across the sea and falls as acid rain in Norway and Sweden. Acid deposition can lead to the following consequences: it can damage or kill trees. it can inhibit plant growth and reduce crop yields. freshwater can be made toxic. it can cause disruption to the food chain. it can damage buildings by eroding away limestone and marble and corroding steel reinforcement within concrete structures. In Sweden, over 18,000 lakes have become so acidic due to acid rain that all the fish have died. In parts of Germany, more than 70,000 square kilometres of forests have died because of acid rain. Acid deposition Materials such as limestone, marble and sandstone are particularly vulnerable to acid deposition. The effects of acid deposition are therefore greater on ancient monuments, rather than on modern buildings. York Minster and Westminster Abbey have been severely eroded in recent years. Other buildings, such as the Taj Mahal in India, the Statue of Liberty in New York and the Colosseum in Rome, have also suffered deterioration due to acid deposition. Masonry dissolved by acid rain. Acid deposition In recent years, the UK and other Governments have taken steps to reduce acid gas emissions. Steps have included: Fitting desulphurisation plant to many power stations. Catalytic converters on new motor vehicles. Unfortunately, both of these measures result in a reduction in fuel efficiency, resulting in higher CO2 emissions for a given energy output. Reductions in emissions have also been achieved by switching to ‘cleaner’ fuels, such as natural gas. Although this reduces SO2 emissions, it does not reduce NOx emissions. Examples of dwellings that minimise their impact on the global environment Examples The Hockerton Housing Project, Southwell, Nottinghamshire The UK’s first earth-sheltered, self-sufficient ecological housing development. A terrace of five single storey earth-sheltered dwellings (four 3 bedroom with internal floor area of 122m2 and one 4 bedroom with an internal floor area of 140m2). Superinsulated and airtight construction incorporating passive solar design. Environmentally friendly materials used throughout. Rainwater is filtered and collected for drinking. Sewage is treated in a reed bed. Wind turbine. Areas for crop cultivation and rearing of animals. Only one fossil-fuelled car permitted per household. Examples The Autonomous Urban House, Southwell, Nottinghamshire The UK’s first ‘autonomous’ house. 2½ storey, four bedroom detached house with an internal floor area of 169m2 (excluding the unheated basement). Superinsulated and airtight construction incorporating passive solar design. Environmentally friendly materials used throughout. Recycled materials used during the construction. Rainwater is collected from the roof and is stored in the basement. Clivius Multrum composting toilet. Produces an odourless fertilizer which is used in the garden. 2.2kW grid-connected PV system. Examples The Beddington Zero Energy Development (BedZed), Sutton, London [Source: BedZed, 2002] Design objective is to achieve zero net CO2 emissions by avoiding the use of fossil fuels. A mixed development containing 82 dwellings which will be a mixture of flats, maisonettes and town houses and approximately 2500m2 of workspace/office and community accommodation. Superinsulated and airtight construction. Wood fuelled CHP plant. Rainwater collected, stored and recycled. Large areas of PV’s installed on buildings. Where possible, materials and equipment sourced from within a 35 mile radius. References AN INCONVENIRENT TRUTH (2006) What is Global Warming? Available from <http://wwww.aninconvenienttruth.co.uk [Accessed 29th September 2006]. BEDZED (2002) Beddington Zero Energy Development. Available from:http://www.bedzed.org.uk [Accessed 3rd October, 2002]. CENTRE FOR ATMOSPHERIC SCIENCE (2006) The Ozone Hole Tour Part III: The Science of the Ozone Hole. Available from: <http://www.atm.ch.cam.ac.uk/tour/part3.html> [Accessed October 5 th, 2006]. CLIMATIC RESEARCH UNIT (2009) Global Temperature Record. Available from: <http://www.cru.uea.ac.uk/cru/info/warming/> [Accessed January 13 tht, 2009]. DEFRA (2009) e-Digest Statistics about: Climate Change. [Internet]. Available from: <http://www.defra.gov.uk/environment/statistics/globatmos/index.htm> [Accessed January 13 th, 2009]. EARTH SYSTEM RESEARCH LABORATORY (2007) Atmospheric Carbon Dioxide – Mauna Loa. [Internet], Washington DC, USA, National Oceanic and Atmospheric Administration. Available from: <http://www.esrl.noaa.gov/gmd/ccgg/trends/co2_data_mlo.html> [Accessed January 13 th, 2009]. FAHEY, D. W. (2002) Twenty Questions and Answers About the Ozone Layer. US Environmental protection Agency. HOUGHTON, J. T. JENKINS, G. J. and EPHRAUMS, J. J. (eds.) (1990) Climate Change: The IPCC Scientific Assessment. Cambridge, Cambridge University Press. HULME, M. JENKINS, G. J. LU, X. TURNPENNY, J. R. MITCHELL, T. D. JONES, R. G. LOWE, J. MURPHY, J. M. HASSELL, D. BOORMAN, P. McDONALD, R. and HILL, S. (2002) Climate Change Scenarios for the United Kingdom: The UKCIP02 Scientific Report. Tyndall Centre for Climate Change Research, School of Environmental Sciences, Norwich, UK, University of East Anglia. References (continued) IPCC (2001) Climate Change 2001: The Scientific Basis. Technical Summary. Geneva, Intergovernmental Panel on Climate Change. IPCC (2007) Summary for Policy Makers. In: Climate Change 2007: The Physical Science Basis. Contribution of Working Group 1 to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change. [Solomon, S. QUIN, D. MANNING, M. CHEN, Z. MARQUIS, M. AVERYT, K. B. TIGNOR, T.MILLER, H. S. (eds.). Cambridge, United Kingdom, Cambridge University Press. KRAUSE, F. BACH, W. and KOOMEY, J. (1990) Energy Policy in the Greenhouse: From Warming Fate to Warming Limit. London, Earthscan Publications. MET OFFICE (2009a) Clearer: Make a Difference About the Facts of Climate Change. [Internet] Available from: <http://www.metoffice.gov.uk/corporate/pressoffice/myths/bigpicture.pdf> [Accessed January 13 th, 2009]. MET OFFICE (2009b) Climate Change Projections. [Internet] Available from: <http://www.metoffice.gov.uk/climatechange/science/projections/> [Accessed January 13th, 2009]. NASA (2007) Ozone Hole Monitoring. [Internet] Available from: <http://jwocky.gsfc.nasa.gov/eptoms/dataqual/ozone.html> [Accessed November 2nd, 2007]. ROYAL COMMISSION ON ENVIRONMENTAL POLLUTION (2000) Twenty Second Report. Energy - The Changing Climate. Royal Commission on Environmental Pollution. London, HMSO. US ENVIRONMENTAL PROTECTION AGENCY (2006) What is Acid Rain? [Internet] Available from: <http://www.epa.gov/airmarkt/acidrain/what/index.html> [Accessed October 13th, 2006].