* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Nuclear Chemistry
Nuclear fission product wikipedia , lookup
Atomic theory wikipedia , lookup
Nuclear fission wikipedia , lookup
Nuclear and radiation accidents and incidents wikipedia , lookup
Nuclear binding energy wikipedia , lookup
Valley of stability wikipedia , lookup
Nuclear transmutation wikipedia , lookup
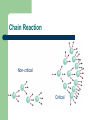
Nuclear Chemistry Chapter 23 23.1-23.6 Nuclear Chemistry Nuclear Chemistry- the study of reactions involving changes in atomic nuclei. Importance Disadvantages Nuclear Reactions Except for Hydrogen, all nuclei contain particles called protons and neutrons. Nuclei can be stable or unstable. Unstable Nuclei emit particles and/or electromagnetic radiation spontaneously. Phenomenon is called Radioactivity. Nuclear Transmutation- results from the bombardment of nuclei by neutrons, protons or other nuclei. Nuclear Reactions Atomic number (Z) = number of protons in nucleus Mass number (A) = number of protons + number of neutrons = atomic number (Z) + number of neutrons Mass Number Atomic Number A ZX Element Symbol Nuclear Reactions neutron 1n 0 proton 1p 1H or 1 1 positron 0 0 +1e or +1 b electron 0b 0e or -1 -1 a particle 4He 4a or 2 2 Balancing Nuclear Equations 1. Conserve mass number (A). The sum of protons plus neutrons in the products must equal the sum of protons plus neutrons in the reactants. 235 92 U + 10n 138 55 Cs + 96 37 Rb 235 + 1 = 138 + 96 + 2x1 + 2 10n Balancing Nuclear Equations 2. Conserve atomic number (Z) or nuclear charge. The sum of nuclear charges in the products must equal the sum of nuclear charges in the reactants. 235 92 U + 10n 138 55 Cs + 96 37 Rb 92 + 0 = 55 + 37 + 2x0 + 2 10n Balancing Nuclear Equations 212Po decays by alpha emission. Write the balanced nuclear equation for the decay of 212Po. 4 alpha particle - 42He or 2a 212Po 84 4He 2 212 = 4 + A A = 208 Z = 82 84 = 2 + Z 212Po 84 + AZX 4He 2 + 208 82Pb Chemical Reactions vs. Nuclear Reactions Nuclear Stability Nucleus is very small Contributes most of weight of atom Extremely high density Even higher # of particles Nuclear Stability Particles repel/attract each other neutron-to-proton ratio Predicting stability: – – – – Magic numbers: 2,8,20,50,82,126 Even numbers of neutrons and protons vs. odd numbers All isotopes of elements with atomic numbers higher than 83 are radioactive. All isotopes of Tc and Pm are radioactive. n/p too large beta decay X Y n/p too small positron decay or electron captur Nuclear Stability Beta decay +-10b + n 14C 6 14N 7 40K 19 40Ca 20 + -10b + n 1n 0 Decrease # of neutrons by 1 Increase # of protons by 1 1p 1 + -10b + n 23.2 Nuclear Stability Positron decay ++10b + n 11C 6 11B 5 38K 19 38Ar 18 Increase # of neutrons by 1 ++10b + n 1p 1 Decrease # of protons by 1 1n 0 ++10b + n n and n have A = 0 and Z = 0 Nuclear Stability Electron capture decay +n 37Ar 18 + -10e 37Cl 17 55Fe 26 + -10e 55Mn 25 1p 1 +n + -10e Increase # of neutrons by 1 Decrease # of protons by 1 1n 0 +n Nuclear Stability Alpha decay 212Po 84 4He 2 + 208 82Pb Decrease # of neutrons by 2 Decrease # of protons by 2 Nuclear Binding Energy Nuclear Binding Energy- the energy required to break up a nucleus into its component protons and neutrons. Necessity? Mass Defect Einstein’s Theory of Relativity E = mc2 Nuclear Binding Energy BE + 199F 911p + 1010n E = mc2 BE = 9 x (p mass) + 10 x (n mass) – 19F mass BE (amu) = [(9 x 1.007825) + (10 x 1.008665)] – 18.9984 BE = 0.1587 amu BE = 2.37 x 10-11J 1 amu = 1.49 x 10-10 J Nuclear Binding Energy binding energy binding energy per nucleon = number of nucleons 2.37 x 10-11 J = 19 nucleons = 1.25 x 10-12 J Nuclear Binding Energy Natural Radioactivity Outside the belt of stability, nuclei are radioactive. Radioactive nuclei spontaneously emit radiation. – α particles, β particles, γ rays, etc. Disintegration of radioactive nucleus leads to a decay series. Radioactive Decay--Dating Uranium decay After time, half of parent exsists Equal amounts of parent and daughter Age? Nuclear Transmutations Rutherford, 1919 Artificial Radioactivity Nitrogen bombarded by α particles 14N 7 + 24a 17O 8 + 11p Nuclear Transmutation Notation for reactions First Isotope (bombarding particle, ejected particle) Final Isotope Notation for Nitrogen-14 bombarded with a particle. Transuranium Elements Synthetic elements Atomic Numbers greater than 92 Particle Accelerator necessary for preparation Particle Accelerator Cyclotron Particle Accelerator Nuclear Fission Nuclear Fission- the process in which a heavy nucleus (mass number > 200) divides to form smaller nuclei of intermediate mass and one or more neutrons. Energy is released. Uranium-235 was the first element discovered to go through nuclear fission. Nuclear Fission 235U 92 + 01n 90Sr 38 1n + Energy + 143 Xe + 3 0 54 Nuclear Fission Nuclear chain reaction is a self-sustaining sequence of nuclear fission reactions. The minimum mass of fissionable material required to generate a self-sustaining nuclear chain reaction is the critical mass. Chain Reaction Non-critical Critical The Atomic Bomb Nuclear Reactors Peaceful application of nuclear fission Generates electricity from chain reactions Provides 20% of electricity in U.S. Light water reactors; Heavy water reactors; Breeder reactors Light Water Reactors Most U.S. nuclear reactors are light water Light Hydrogen Use Uranium-235 under controlled conditions Releases large quantities of steam Steam drives electric generators Needs large amounts of coolant Plants built by lakes and rivers Large amounts of thermal pollutant Light Water Reactors Heavy Water Reactors Uses Deuterium D2O D absorbs neutrons less efficiently than H Does not require U-235 Neutrons leak out of reactor Expensive to prepare D2O Environmentally friendly Breeder Reactors Breeder Reactor- uses uranium fuel, but unlike a conventional nuclear reactor, it produces more fissionable materials than it uses. Converts uranium-238 to plutonium-239 in a 3 step process. Plutonium-239 undergoes fission Reactor produces 1 mole of p-239 for every 1 mole used. Takes 7-10 years for complete regeneration. Hazards of Nuclear Energy Production of radioactive isotopes with long half-lives (24,400 years) Radioactive and toxic substances Three-mile Island Reactor- radiation escaped Chernobyl Nuclear Plant- fire and explosion Accidents Waste Disposal Nuclear Fusion Nuclear Fusion- the combining of small nuclei into larger ones. Two small nuclei can combine and release large amounts of energy To occur, the nuclei must be in an environment with high temperature. Thermonuclear Reactions Nuclear fusion occurs constantly on the Sun. Nuclear Fusion How do we get it to occur? Container? The Hydrogen Bomb Thermonuclear Bomb All power and no control Fusion reaction then fission reaction Fusion reaction creates high temp. for fission reaction Bombs usually contain Co-59 and upon explosion convert to Co-60