* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Chapter 2 Basic Chemistry
Resistive opto-isolator wikipedia , lookup
Electric machine wikipedia , lookup
Current source wikipedia , lookup
Power engineering wikipedia , lookup
Flexible electronics wikipedia , lookup
Stray voltage wikipedia , lookup
Electrification wikipedia , lookup
History of electromagnetic theory wikipedia , lookup
History of electric power transmission wikipedia , lookup
Mains electricity wikipedia , lookup
Mercury-arc valve wikipedia , lookup
Electric battery wikipedia , lookup
Opto-isolator wikipedia , lookup
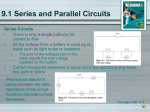
Chapter 17 Electricity Electric Charge and Force • Electric charge: an electrical property of matter that creates electric and magnetic forces • An object has a negative charge, a positive charge, or no charge at all • Like charges repel, and opposite charges attract Electric Charge and Force • Rub a balloon back and forth across your hair – The balloon will be attracted to your hair and vice-versa • Two balloons will repel each other Electric Charge and Force Electric Charge and Force Electric Charge and Force Electric Charge and Force Electric charge • Electric charge depends on an imbalance of protons and electrons – The difference in the number of protons and electrons determines an object’s electric charge • Negatively charged objects have more electrons than protons • Positively charged objects have fewer electrons than protons Electric Charge and Force • The SI unit of electric charge is the coulomb (C) – Proton has a charge of: +1.6 x 10-19C – Electron has a charge of: -1.6 x 10-19C • An object with a charge of -1.0 has 6.25 1018 excess electrons – 1.6 – 1.0 = 0.6 x 1019 = 6 x 1018 electrons Electric Charge and Force • Transfer of charge: – When materials are rubbed together, electrons can be transferred from one material to other • Conductors allow charges to flow: – Copper, aluminum • Insulators do not allow charges to flow: – Paper, cardboard, glass, plastic Electric Charge and Force • Charges can move within uncharged objects without changing the overall charge of the material – Induction: redistribution of charge without contact – Contact: redistribution of charge with contact – Friction redistribution of charge with rubbing – Surface charge can be created on an insulator • Charged comb and paper / water a charged balloon Electric Charge and Force Electric Charge and Force Electric Charge and Force Electric Charge and Force Electric Charge and Force Electric Charge and Force Electric Charge and Force • Electric field lines never cross one another Current • Just as a ball will roll downhill, a negative charge will move away from another negative charge • Electrical potential energy is the ability to move an electric charge from one point to another Current • Potential difference is measured in volts – 1 volt (V) is equivalent to 1 joule per coulomb (1 J/C) • J = Nxm / W = F x d Current • There is voltage across the terminals of a battery: potential difference between the positive and negative terminals – Example: 1.5V small battery / 12V car battery – Most batteries are electrochemical cells • Convert chemical energy into electrical energy • Contains an electrolyte (solution that conducts electricity) and two electrodes (each a different conducting material: lead/lead alloy/zinc) – Dry cell: paste – Wet cell: liquid Current Current • The chemical reactions in the battery causes a build up of electrons at the anode. This results in an electrical difference between the anode and the cathode. You can think of this difference as an unstable build-up of the electrons. The electrons wants to rearrange themselves to get rid of this difference. But they do this in a certain way. Electrons repel each other and try to go to a place with fewer electrons. Current • In a battery, the only place to go is to the cathode. But, the electrolyte keeps the electrons from going straight from the anode to the cathode within the battery. When the circuit is closed (a wire connects the cathode and the anode) the electrons will be able to get to the cathode. In the picture above, the electrons go through the wire, lighting the light bulb along the way. This is one way of describing how electrical potential causes electrons to flow through the circuit. Current • However, these electrochemical processes change the chemicals in anode and cathode to make them stop supplying electrons. So there is a limited amount of power available in a battery. Current • When you recharge a battery, you change the direction of the flow of electrons using another power source, such as solar panels. The electrochemical processes happen in reverse, and the anode and cathode are restored to their original state and can again provide full power. Current Current • Lead–acid storage battery, 2 V. (usually 6 in series to give 12V supply). • The electrodes are initially hard lead–antimony alloy plates coated in a paste of lead(II) sulphate encased in dilute sulphuric acid. During the first charging some of the lead(II) sulphate is reduced lead(0) on one of the electrodes (this will acts as the (–) anode in discharging). Simultaneously in charging, lead(II) sulphate is oxidised to lead(IV) oxide on the other electrode which acts as the cathode (+) in discharging. • Pb(s)|PbSO4(s)|H+(aq),HSO4–(aq)||PbO2(s)|PbSO4(s)|Pb(s) – Note the standard conventions in common use • the | notation indicates a phase boundary, solutes in the same phase are separated by a comma – the || notation 'divides' the two half cells and » the oxidation state increases 'towards' it Current • (–) anode discharging reaction (i) Pb(s) + HSO4–(aq) ==> PbSO4(s) + H+(aq) + 2e– • (+) cathode discharging reaction (ii) PbO2(s) + 3H+(aq) + HSO4–(aq) + 2e– ==> PbSO4(s) + 2H2O(l) • working cell reaction (iii) PbO2(s) + 2H+(aq) + 2HSO4– (aq) + Pb(s) ==> 2PbSO4(s) + 2H2O(l) • oxidation state changes: (i) oxidation Pb(0) ==> Pb(II) : (ii) reduction Pb(IV) ==> Pb(II) • The charging reactions will be the opposite of (i) and (ii) Current • Advantages: Inexpensive, high power density (can car starter motor as well as lights), long shelf life, readily recharges, so has a long working life of many years. • Disadvantages: Lead needs to be recycled to avoid environmental contamination, sometimes generates hydrogen gas at the cathode when charging (explosive in air + spark) – though batteries seem to be made of a high standard these days in completely sealed units that last many years. • Uses: Car batteries. Current • - The cell has one plate made of lead and another plate made of lead dioxide, with a strong sulfuric acid electrolyte in which the plates are immersed. - Lead combines with SO4 (sulfate) to create PbSO4 (lead sulfate), plus one electron. - Lead dioxide, hydrogen ions and SO4 ions, plus electrons from the lead plate, create PbSO4 and water on the lead dioxide plate. - As the battery discharges, both plates build up PbSO4 and water builds up in the acid. The characteristic voltage is about 2 volts per cell, so by combining six cells you get a 12-volt battery. Current • A lead-acid battery has a nice feature — the reaction is completely reversible. If you apply current to the battery at the right voltage, lead and lead dioxide form again on the plates so you can reuse the battery over and over. Current • Most car batteries constitute six galvanic cells laid out in series. Each cell delivers 2.1 volts of electromotive force that, when combined, produce a 12. volt battery (commonly advertised as 12 volt) – Series: one dies: battery dies Current • A galvanic cell, or voltaic cell, named after Luigi Galvani, or Alessandro Volta respectively, is an electrochemical cell that derives electrical energy from spontaneous redox reactions taking place within the cell. It generally consists of two different metals connected by a salt bridge, or individual half-cells separated by a porous membrane. • Volta was the inventor of the voltaic pile, the first electrical battery. In common usage, the word "battery" has come to include a single galvanic cell, but a battery properly consists of multiple cells. Current Current • A salt bridge, in electrochemistry, is a laboratory device used to connect the oxidation and reduction half-cells of a galvanic cell (voltaic cell), a type of electrochemical cell. It maintains electrical neutrality within the , preventing the cell rapidly running its reaction to equilibrium. If no salt bridge were present, the solution in one half cell would accumulate negative charge and the solution in other half cell would accumulate positive charge as the reaction proceeded, quickly preventing further reaction, and hence production of electricity.[1] • Salt bridges usually come in two types: glass tube and filter paper. Current Current • Each galvanic cell consists of a series of lead (Pb) and lead dioxide (PbO2) plates which are submerged in an electrolyte solution-a mix of sulfuric acid (35%) and water (65%). This acidic bath triggers a reaction with the lead dioxide plate (the positive electrode) which produces ions and lead sulfate. Current • These sulfuric ions then react with adjacent lead plates (the negative electrode) to produce hydrogen ions and lead sulfate. This chemical reaction, in turn, generates electrons that can flow out via conductive terminals as electricity to power essential functions. Current Current • So that is the reaction that is occurring in each cell of the battery. For each atom of lead converted to PbSO4 two electrons are produced. These are absorbed by one piece of PbO2 becoming PbSO4. Current • How Batteries Work: The Reverse Reaction: Recharging the Battery • To obtain the reverse reactions all we need to do is turn the arrows to face the opposite direction. If we do this we can see that the PbSO4 is converted back into Pb on one plate and PbO2 on the other. This is a forced reaction that can only occur if extra electrons are pushed into the battery. • The reaction is not permanent, with lead sulfate reforming into lead dioxide and lead when the battery is recharged. Overtime, the battery’s efficiency will deplete, with most working optimally for about 4 years Current Current • Resistance is caused by internal friction, which slows the movement of charges through a conducting material • The resistance of the filament of a light bulb determines how bright the bulb will be. – The filament of a 40 W light bulb has a higher resistance than the filament of a 100 W light bulb Current • A 40 Watt light's filament (for example) operates at much the same temperature as the filament in a 100 Watt light. The difference between them is that the 100 Watt light has a thicker filament and uses more current. But their filament temperatures are much the same, so their rate of evaporation of their filaments is similar. Current Current • Ohm’s Law • Resistance = voltage / current • R=V/I • Cover the variable that you require and perform the resulting calculation in the triangle Current • The headlights of a typical car are powered by a 12 volt battery. What is the resistance of the headlights if they draw 3.0 A of current when they are turned on? • R = V / I = 12 V / 3 A = 4.0 ohm Current • Conductors have low resistance • Insulators have high resistance • Semiconductors conduct under certain conditions – Electrical properties between those of an insulator and a conductor – Naturally insulator but when specific materials are added their ability to conduct increases Current Current • The atoms in a semiconductor are materials from either group IV of the periodic table, or from a combination of group III and group V (called III-V semiconductors), or of combinations from group II and group VI (called II-VI semiconductors). Because different semiconductors are made up of elements from different groups in the periodic table, properties vary between semiconductors. Silicon, which is a group IV, is the most commonly used semiconductor material as it forms the basis for integrated circuit (IC) chips and is the most mature technology and most solar cells are also silicon based. A full periodic table is given in the page Periodic Table. Several of the material properties of silicon are given in the page Silicon Material Parameters Current • Some materials can become superconductors – Some metals and compounds have zero resistance when their temperature falls below a critical temperature and pressure • Temperature/pressure depends on the material – Usually very high or low temperatures – Usually ambient (pressure of surrounding material) or high pressure – The search continues for superconductors at room temperature Current Circuits • Circuit: a closed-loop path for electrons to follow – Because charges are moving, there is a current in the circuit • The conducting path produced when a load, such as a string of light bulbs, is connected across a source of voltage (outlet) is called a closed circuit Circuits • Switches interrupt the flow of charges in a circuit Circuits • Schematic diagrams are used to represent circuits Circuits Circuits Circuits Circuits • A resistor is a passive two-terminal electrical component that implements electrical resistance as a circuit element. Resistors act to reduce current flow, and, at the same time, act to lower voltage levels within circuits. Resistors may have fixed resistances or variable resistances, such as those found in thermistors, varistors, trimmers, photoresistors, humistors, piezoresistors and potentiometers. • The current through a resistor is in direct proportion to the voltage across the resistor's terminals. This relationship is represented by Ohm's law: – • where I is the current through the conductor in units of amperes, V is the potential difference measured across the conductor in units of volts, and R is the resistance of the conductor in units of ohms (symbol: Ω). Circuits Circuits Circuits • Series circuits have a single path for current – Voltage is divided among the devices – Current in each device is the same but the resistance can be different • Therefore, the voltage across each device can be different – If one element along the path is removed, the circuit will not work Circuits Circuits Circuits • Parallel circuits have multiple paths for current – Voltage across each device is the same – The current does not have to be the same • The sum of the currents in all of the devices equals the total current • If one bulb is removed the charges would still move through the other loop Circuits Circuits Circuits Circuits • When a charge moves in a circuit, the charge loses energy. Some of this energy is transformed into useful work, such as the turning of a motor, and some is lost as heat. • The rate at which electrical energy is changed to other forms of energy is called electric power • Power = current x voltage • P = IV • SI unit for power is the watt (W) – 1 W = 1J./s – 1kW = 1,000W Circuits • Electric companies measure energy in kilowatt-hours – Charge for energy, not power used in the home – One kilowatt-hour (kW x h) is the energy delivered in 1 h at the rate of 1kW • 1 kW x h = 3.6 x 106J • Price could be 5-20 cents per kilowatt-hour Circuits Circuits Circuits Circuits • When a hair dryer is plugged into a 120 V outlet, the hair dryer has a 9.1 A current in it. What is the hair dryer’s power rating? • Power = current x voltage • P = IV • P = 9.1A x 120V • P = 1.1 x 103W Circuits • High currents in overloaded circuits can cause fires • Large power overloads may potentially destroy electrical equipment, or in more serious cases, cause a fire. A fuse and circuit breaker both serve to protect an overloaded electrical circuit by interrupting the continuity, or the flow of electricity. How they interrupt the flow of electricity is very different, however. A fuse is made up of a piece of metal that melts when overheated; a circuit breaker has an internal switch mechanism that is tripped by an unsafe surge of electricity. Fuses tend to be quicker to interrupt the flow of power, but must be replaced after they melt, while circuit breakers can usually simply be reset. Circuits