* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Campbell - Microelectronics Research and Communications
Electrical substation wikipedia , lookup
Opto-isolator wikipedia , lookup
Electrical ballast wikipedia , lookup
Immunity-aware programming wikipedia , lookup
Three-phase electric power wikipedia , lookup
Switched-mode power supply wikipedia , lookup
Buck converter wikipedia , lookup
Voltage optimisation wikipedia , lookup
Stray voltage wikipedia , lookup
Mains electricity wikipedia , lookup
Rectiverter wikipedia , lookup
Alternating current wikipedia , lookup
Resistive opto-isolator wikipedia , lookup
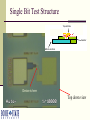
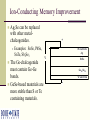
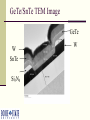
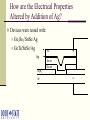
Multi-Layer Phase-Change Electronic Memory Devices Kris Campbell Associate Professor Dept. of Electrical and Computer Engineering & Dept. of Materials Science and Engineering Boise State University University of Idaho ECE Research Colloquium March 8, 2007 Introduction memories – why do we need a new memory technology? Types of chalcogenide resistive memories – ion conducting and phase-change Chalcogenide memory stack structures Tuning the phase-change memory operating parameters Chalcogenide-based With materials Electrically Summary What is a Chalcogenide Material? A Chalcogenide material contains one of the Group VI elements S, Se, or Te (O is usually omitted). Some examples of chalcogenides: GeS – germanium sulfide SnSe – tin selenide ZnTe – zinc telluride Uses of Chalcogenide Materials Energy generation (solar cells) Memory (CD’s, electronic) Chalcogenide materials are key to many new technology developments Environmental pollutant detection Photodetectors Energy storage (batteries) Why Are New Memory Technologies Under Development? Could replace both DRAM and Flash memory types DRAM has reached a size scaling limitation and is volatile Flash is prone to radiation damage, is high power, and has a short cycling lifetime Radiation resistant Scalable Low power operation Reconfigurable electronics applications Potential for multiple resistance states (means multiple data states in a single bit) How Does a Chalcogenide Material Act as a Memory? Chalcogenide materials can be used as resistance variable memory cells: Logic ‘0’ state: Rcell> 200 kΩ Logic ‘1’ state: Rcell= 200 Ω to 100 kΩ The resistance ranges vary quite a bit depending upon the material used. V V Write, Vw 10 kΩ 1 MΩ Erase, Ve ‘0’ OFF ‘1’ ON ON and OFF State Distributions Resistance values in the ON and OFF states have a distribution of values; 1.0 Distribution 0.8 0.6 ON OFF 1k to 200k 1M to 1G 0.4 0.2 Resistance Threshold voltages or programming currents for ON and OFF states also have a distribution of possible values. Single Bit Test Structure Top electrode Memory cell Metalchalcogenide Insulator Bottom electrode Device is here Top down view Types of Chalcogenide Resistive Memory Ion-Conducting Ions (e.g. Ag+ and Cu+) are added to a chalcogenide glass Application of electric field causes formation of a conductive channel through glass (Kozicki, M.N. et al., Microelectronic Engineering 63, 485 (2002)) Thermally Induced Phase Change Crystalline to amorphous phase change; low R to high R shift High current heats material to cause phase change (S.R. Ovshinsky, Phys. Rev. Lett. 21, 1450 (1968)) Ion-Conducting Memories Resistance variable memory based on Ag+ mobility in a chalcogenide glass; Ag is photodoped into a GexSe100-x based chalcogenide glass (x<33). Visible light Ag Ge30Se70 (Ge40Se60)33 (Ag2Se)67 Developed by Axon Technologies (http://www.axontc.com) Ion-Conducting Memories - Operation A positive potential applied to the Ag electrode writes the bit to a low resistance state; +Ag electrode V (Ge2Se3)33(Ag2Se)67 A negative potential applied to the Ag-containing electrode erases the bit to a high resistance state. -+ Ion-Conducting Chalcogenide-Based Memories Example material: Ge30Se70 photodoped with Ag Ag V (Ge30Se70)67Ag33 W From Kozicki, et al. NVMTS, Nov. 2004. Why is Glass Stoichiometry Important For Photodoping? Glasses in region I Mitkova, M.; et al., Phys. Rev. Lett. 83 (1999) 3848-3851. phase separate and form Ag2Se. Glasses in region II will not phase separate Ag2Se but will put Ag on the glass backbone. Photodoped Ge30Se70 will form 32% Ge40Se60 and 68% Ag2Se. Traditional Ion-Conducting Structure vs Stack Structure Ag Ge30Se70 Bottom electrode Traditional Ion-Conducting Memory Structure Top electrode Ag2+xSe Ge40Se60 Bottom electrode Stacked Layer Ion-Conducting Memory Structure Ag2Se-Based Ion-Conducting Memory (Instead of Photodoping with Ag) 20 ‘1’ Low R Current (microamps) 15 + W electrode 10 V Ag2+Se 5 Ge40Se60 0 -5 -10 -0.4 W electrode ‘0’ High R -0.2 Ve 0.0 Voltage - Vw 0.2 0.4 Ion-Conducting Memory Improvement Ag2Se can be replaced with other metalchalcogenides. Examples: SnSe, PbSe, SnTe, Sb2Se3 The Ge-chalcogenide must contain Ge-Ge bonds. GeSe-based materials are more stable than S or Te containing materials. + W electrode Ag V SnSe Ge40Se60 W electrode - Ion-Conducting Memory Improvement Eliminate Ag photodoping Use a metal-chalcogenide layer above a GexSe100-x glass with carefully selected stoichiometry 20 + ‘1’ Low R W electrode Ag V SnSe Metal Chalcogenide Ge40Se60 W electrode - Current (microamps) 15 10 5 0 -5 -10 -0.4 ‘0’ High R -0.2 Ve 0.0 Voltage Vw 0.2 0.4 Ion-Conducting Memory Research Projects Investigate operational mechanism: Influence of metal in the Metal-Se layer. Role of redox potential Glass – rigid or floppy Type of mobile ion (e.g. Ag or Cu) Effects of these on memory properties: switching speed power data retention resistance distribution thermal tolerance What Are Phase-Change Materials? Materials that change their electrical resistance when they are switched between crystalline and glassy (disordered) structures. A well-studied example is Ge2Sb2Te5 (referred to as GST). Figure modified from Zallen, R. “The Physics of Amorphous Solids” JohnWiley and Sons, New York, (1983) 12. Low Resistance High Resistance Thermally Induced Phase Change Creates High R State Creates Low R State Phase Change Memory IV Curve One programming voltage polarity. Current requirement can be high. Voltage Polycrystalline application must go beyond VT before switching will occur. Traditional Phase Change Structure Compared to a Stack Structure Top electrode Top electrode SnTe Ge2Sb2Te5 Bottom electrode Traditional Phase Change Memory Structure GeTe Bottom electrode Stacked Phase Change Memory Structure Phase-Change Memory Multi-Layer Stack Structures Tested Devices consist of a core Ge-chalcogenide (Ge-Ch) layer and a metal chalcogenide layer (MCh). Properties wanted: Flexible operational properties; tunable via materials selection or operating method Multiple resistance states Low power Large cycling lifetime Device Dimensions: 0.25 um via Initial Devices Tested Initial devices tested consisted of the stacks: (1) GeTe/SnTe (2) Ge2Se3/SnTe (3) Ge2Se3/SnSe It was found that the material layers used had a significant effect on device operation.* *Campbell, K.A.; Anderson, C.M. Microelectronics Journal, 38 (2007) 52-59. GeTe/SnTe TEM Image GeTe W SnTe Si3N4 W Electrical Characterization Methodology Perform a current sweep with the top electrode potential either at a +V or a -V. Perform limited cycling endurance measurements on single bit structures. Initial Electrical Characterization GeTe/SnTe Structure, +V +V is on the electrode nearest the SnTe Layer (top electrode) -4 10 -5 10 -6 Current (A) 10 -7 10 -8 10 -9 10 -10 10 -11 10 0.4 0.6 1.0 0.8 Voltage 1.2 1.4 Initial Electrical Characterization GeTe/SnTe Structure, -V -V is on the electrode nearest the SnTe layer (top electrode) -4 10 -5 10 -6 Current (A) 10 -7 10 -8 10 -9 10 -10 10 -11 10 0.0 0.5 1.0 1.5 Voltage (V) 2.0 2.5 Snap back at a higher V and higher I than the +V case. Initial Electrical Characterization Ge2Se3/SnTe Structure -4 10 -5 10 -6 Current (A) 10 -7 10 -8 10 +V -V -9 10 -10 10 -11 10 0.2 0.4 0.6 0.8 Voltage 1.0 1.2 1.4 Initial Electrical Characterization Ge2Se3/SnSe Structure -4 10 -5 10 -6 Current (A) 10 -7 10 -8 10 +V -V -9 10 No switching! -10 10 -11 10 0 2 4 6 Voltage 8 10 Initial Electrical Characterization Ge2Se3/SnSe Structure A 30nA pre-condition (+V), Followed by -V -7 -5 10 -8 Current (A) Current (A) 10 10 -9 10 -10 10 -11 -7 10 -9 10 Switching! -11 10 10 0.0 1.0 2.0 Voltage (V) (a) 3.0 0.0 1.0 2.0 Voltage (V) (b) 3.0 Movement of Sn Ions into Ge2Se3 Activates Operation +V drives Sn2+ or Sn4+ ions into the lower glass layer, thus allowing it to phase change. -V will not produce phase change since Sn ions do not move into lower glass. An activation (pre-conditioning) step of +V at very low current (nA) will alter the Ge2Se3 material, thus allowing phase change operation to occur with –V. Initial Results Summary GeTe/SnTe – phase change switching, +/-V Ge2Se3/SnTe – phase change switching, +/-V Ge2Se3/SnSe – phase change switching, +V; -V switching only possible after +V, low current conditioning. Sn ions were moved into the Ge-Ch layer during +V operation. Te ions were moved into Ge-Ch layer during -V operation. Tuning the Switching Properties By selection of stack structure, we can create a device with selective operation (on only when activated). Operational mode depends on the voltage polarity used with the device. Can we tune the switching properties by altering the metal used in the metal chalcogenide layer or the electrode materials? Tuning Operating Parameters with Materials Ge-Ch stoichiometry: Ge-Ge bonds provide a thermodynamically favorable pathway for ion incorporation. Metal-Ch: The redox potential, ionic radii, oxidation state, and coordination environment properties of the metal will impact the ability of the metal ion to migrate into and incorporate into the Ge-Ch material. Addition of other metal ions: What happens upon the addition of small amounts of Cu or Ag? Testing the Lower Glass and Metal Ion Influence We have subsequently tested the following stacks: (1) GeTe/ZnTe – metal ion influence (2) GeTe/SnSe – lower glass influence (3) Ge2Se3/SnSe/Ag – metal ion (4) GeTe/SnSe/Ag – metal ion and lower glass (5) Ge2Sb2Te5 (GST)/SnTe – lower glass Resistance switching is observed in all stacks – but switching properties are different. Current-Voltage Curves of Stack Structures -4 +V 10 applied -5 10 -6 Current (A) 10 -7 10 -8 10 Ge2Se3/SnTe Ge2Se3/SnSe GeTe/SnTe GST/SnTe GST -9 10 -10 10 -11 10 0.0 0.5 1.0 1.5 2.0 Voltage 2.5 3.0 3.5 Effects of M-Ch Layer on Switching +V applied -4 10 -5 10 -6 Current (A) 10 -7 10 GeTe/ZnTe GeTe/SnTe -8 10 -9 10 -10 10 -11 10 0.5 1.0 1.5 2.0 Voltage 2.5 3.0 3.5 How are the Electrical Properties Altered by Addition of Ag? Devices were tested with: Ge2Se3/SnSe/Ag GeTe/SnSe/Ag W + Ag Sn-ch Ge-ch Si3N4 W _ Ge2Se3/SnSe/Ag Device – Multistate Resistance Behavior 100 700 Current (A) 80 60 1K 40 20 2K 5K 5K 0 0.00 0.02 0.04 0.06 0.08 0.10 Voltage (V) 0.12 0.14 GeTe/SnSe/Ag Device – Some Multistate Behavior 100 Current (A) 80 60 1k 40 3k 20 0 0.00 0.05 0.10 0.15 0.20 Voltage (V) 0.25 0.30 0.35 Metal Ion Effects Summary The metal ion influences the possible multiple resistance states. Metal ion allows phase change switching in cases where the Ge-Ch normally does not switch. We can use the metal ion to alter the voltage needed to initiate ‘snap back’ for phase change operation or alter the switching currents. Under investigation: Switching speed and cycle lifetime Temperature dependence Resistance state retention Resistance stability of multistate behavior. Electrical Characterization – Lifetime Cycling Single bit testing is not ideal, however it does provide insight into how the material stack might perform over many cycles. Agilent 33250A Arbitrary Waveform Generator Agilent Oscilloscope Micromanipulator PCRAM Device Micromanipulator Rload Rload is typically 10 kΩ to 1 kΩ depending on the material under study. Electrical Characterization – Lifetime Cycling – GeTe/SnTe GeTe/SnTe – initial tests show bits cycle > 2 million times. Input (red) and V across load resistor (black) Electrical Characterization – Lifetime Cycling – Ge2Se3/SnTe Ge2Se3/SnTe – initial tests show more consistent cycling than GeTe/SnTe structures. Input (red) and V across load resistor (black) Current through device (calculated by Vload/Rload) Electrical Characterization – Lifetime Cycling –Ge2Se3/SnSe > 1e6 Erase 6 Amplitude (V) cycles Operation up to 135 °C. 8 Vout Vin 4 Write 2 Read Read 0 0 1 2 Time (ms) 3 Ge2Se3/SnSe/Ag Device Cycling T = 135°C; Rload = 1kΩ 1.5 Write 1.0 0.5 Voltage (V) Input Response after given number of cycles: 1 10 2 10 3 10 4 10 5 10 6 10 Read 0.0 Read -0.5 -1.0 Erase -1.5 0 100 200 Time (s) 300 400 GeTe/SnSe/Ag Device Cycling T = 30°C; Rload = 1.5kΩ 1.5 Write 1.0 Voltage (V) Input Response after given number of cycles: 1 10 2 10 3 10 4 10 5 10 6 10 0.5 Read 0.0 Read -0.5 -1.0 Erase -1.5 0 100 200 300 400 Time (s) 500 600 Materials Questions We Need To Ask How are switching parameters altered by the materials and stack structure? Influence of Ge-Ch structure on switching? Properties of the M-Ch work function? Metal ion properties? How well does it ‘fit’ into the glass structure? How mobile is the ion and what energy is required to cause it to move? Adhesion to electrodes? Knowing these answers will allow optimization for device electrical property tuning. Tuning Operating Parameters Electrically Can we find electrical probing techniques that will: Enable well separated resistance states? Improve data retention and temperature dependence? Create a wide dynamic range of allowed resistance values in a programmed state? What are the operating limitations in order to avoid losing the resistance state while in use in a circuit? Multiple Resistance States – Challenges Resistance range can vary as a function of: Programming current Temperature Programming pulse parameters Retention time of the resistance value can also vary as a function of these parameters. How well does the resistance state get retained during operation as a ‘resistor’ in a circuit? Quite often, due to the nature of the amorphous materials, the resistance values have a large spread. This overlap prevents reliable use of multistate programming with these materials. Can we use electrical techniques to help? Example of Poor Programming Resistance Distributions: GeTe/SnSe + potential Programming Current 100uA 1mA - potential 5 10 8 7 6 5 2 Resistance (Ohms) Resistance (Ohms) 6 10 6 4 2 5 10 6 4 4 3 2 4 10 8 7 6 5 2 4 10 0 2 4 6 8 Device Number 0 2 4 6 8 Device Number Electrical Control: Reverse Potential Programming Provides Multiple Resistance States 9 10 8 Resistance (Ohms) 10 100A max +V Reverse potential 1mA max -V OFF 7 10 6 10 5 10 4 10 0 2 4 6 Device Number 8 Electrical Control Summary Multistate resistance programming possible by programming with negative and positive potentials in the Ge-Ch/M-Ch stack structure. Electrically controlled activation of stack structure allows a device to be ‘turned on’ when it is needed. Summary Using Stacked Layers, we have more device operational flexibility… We can control and tune operational parameters: Threshold voltage, programming current, speed, retention, endurance Value of resistance states Number of possible resistance states We can electrically control device function Electrically activated devices Larger dynamic range between resistance states Acknowledgements Collaborators: Prof. Jeff Peloquin, Boise State University – synthesis of materials. Mike Violette, Micron Technology – equipment loan and use of analytical facilities for thin film characterization (SEM, ICP, TEM). Prof. Santosh Kurinec, Rochester Institute of Technology – characterization of thin film stacks using XRD, RBS, Raman; development of CMOS-based test array for materials stacks. Students: Morgan Davis, Becky Munoz, Chris Anderson, Daren Wolverton. Funding: This research was partially supported by a NASA Idaho EPSCoR grant, NASA grant NCC5-577. Phase-Change Memory Radiation Resistance Phase-Change Memory ON state: OFF state: Complete crystallization is not induced by SEE or TID. Localized crystallization can occur.* Metal 2 Even if some regions in the crystalline material are disturbed by SEE or TID, the crystallinity in the rest of the cell will keep R low. Metal 2 Rc1 Rc2 Rc1 Chalcogenide Rc2 Crystalline Ra1 Crystalline Ra2 Ra1 Amorphous Metal 1 * El-Sayed, S.M. Nuclear Instruments and Methods in Physics Research B 225 (2004) 535-543. Chalcogenide Ra2 Amorphous Metal 1 Ion-Conducting Memory Radiation Resistance Ion-Conducting Memory OFF State: Material is ON State: Ag filling the disordered, SEE or TID will not affect it. conductive channel would have to be completely displaced from contact with either electrode. + + Ag electrode Ag electrode V (Ge2Se3)33(Ag2Se)67 V (Ge2Se3)33(Ag2Se)67 - -































































![Electricity and Magnetism [2]](http://s1.studyres.com/store/data/007809428_1-2a02c27dae15966177eeec747c9ebd14-150x150.png)