* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Physiology of cardiovascular system lecture 1, 2 Dr.Abdul
Survey
Document related concepts
Cardiac contractility modulation wikipedia , lookup
Heart failure wikipedia , lookup
Cardiovascular disease wikipedia , lookup
Management of acute coronary syndrome wikipedia , lookup
Quantium Medical Cardiac Output wikipedia , lookup
Electrocardiography wikipedia , lookup
Lutembacher's syndrome wikipedia , lookup
Antihypertensive drug wikipedia , lookup
Coronary artery disease wikipedia , lookup
Jatene procedure wikipedia , lookup
Arrhythmogenic right ventricular dysplasia wikipedia , lookup
Heart arrhythmia wikipedia , lookup
Dextro-Transposition of the great arteries wikipedia , lookup
Transcript
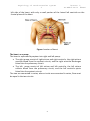
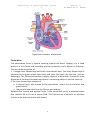
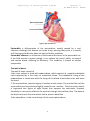
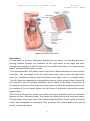
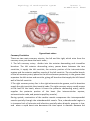
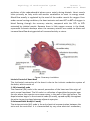
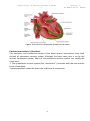
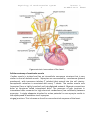
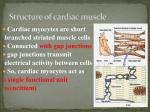
Physiology of cardiovascular system 2 lecture 1, Dr.Abdul-Aziz Ahmed The cardiovascular system The cardiovascular system(CVS) consists of the heart, blood vessels blood. Function The main function of the cardiovascular system: 1.Transport of substances: CVS delivers oxygen and nutrients to the tissues and carries waste products from cellular metabolism to the kidneys and other excretory organs for elimination. it also circulates electrolytes and hormones, immune cells, clotting proteins. 2. Thermoregulation (skin blood vessel) 3.Protection: blood clotting (protects against haemorrhage) and immune system against pathogens . An Overview of the Cardiovascular System The cardiovascular system perform its impressive work, depending on the heart together with a network of blood vessels. The network can be subdivided into two circuits: the pulmonary circuit, which carries carbon dioxide-rich blood from the heart to the gas exchange surfaces of the lungs and returns oxygen-rich blood to the heart the systemic circuit, which transports oxygen-rich blood from the heart to the rest of the body’s cells, returning carbon dioxide-rich blood back to the heart. The heart The heart, is hollow cone-shaped four-chambered muscular pump approximately the size of a fist. The heart rests on the diaphragm between the lungs in the mediastinal space of the intrathoracic cavity in a loose-fitting sac called the pericardium. It is suspended by the great vessels, with its broader side (i.e., base) facing upward and its tip (i.e., apex) pointing downward, forward, and to the left. It is located posterior to the sternum and anterior to the vertebral column. The heart is positioned obliquely, so that the right side of the heart is almost fully in front of the 1 Physiology of cardiovascular system 2 lecture 1, Dr.Abdul-Aziz Ahmed left side of the heart, with only a small portion of the lateral left ventricle on the frontal plane of the heart. Figure: location of heart. The heart as a pump The heart is separated by septum into right and left pump: The right pump consist of right atrium and right ventricle, the right atrium receives blood from the systemic circuit, and the right ventricle discharges blood into the pulmonary circuit. The left pump consist of left atrium and left ventricle, the left atrium collects blood from the pulmonary circuit, and the left ventricle ejects blood into the systemic circuit. The two are connected in series, when circuits are connected in series, flow must be equal in the two circuits. 2 Physiology of cardiovascular system 2 lecture 1, Dr.Abdul-Aziz Ahmed Figure:the chambers of the heart. Pericardium The pericardium forms a fibrous covering around the heart, holding it in a fixed position in the thorax and providing physical protection and a barrier to infection. The pericardium consists of: 1. a tough outer fibrous layer and a thin inner serous layer. The outer fibrous layer is attached to the great vessels that enter and leave the heart, the sternum, and the diaphragm. The fibrous pericardium is highly resistant to distention; it prevents acute dilatation of the heart chambers and exerts a restraining effect on the left ventricle. 2.The inner serous layer consists of a visceral layer, also known as the epicardium, covers the entire heart and great vessels the parietal layer that lines the fibrous pericardium. Between the visceral and parietal layers is the pericardial cavity, a potential space that contains 30 to 50 mL of serous fluid. This fluid acts as a lubricant to minimize friction as the heart contracts and relaxes. 3 Physiology of cardiovascular system 2 lecture 1, Dr.Abdul-Aziz Ahmed Figure:pericardium. Pericarditis is inflammation of the pericardium, usually caused by a viral infection. Although this disease can cause sharp, piercing chest pain, it is usually self-limiting and ordinarily does not lead to further problems. Pericardial effusion is a collection of fluid around the heart in the pericardial sac. If the fluid amount is great enough, it can reduce the heart’s ability to expand and receive blood, reducing its efficiency. This condition is known as cardiac tamponade. The wall of heart The wall of heart consist of 1.the inner surface is lined with endocardium, which consists of smooth endothelial cells supported by a thin layer of connective tissue. The endothelial lining of the endocardium is continuous with the lining of the blood vessels that enter and leave the heart. 2.The myocardium, consists largely of cardiac muscle tissue. The muscle fibers of the myocardium are branched and tightly joined to one another. The ventricular muscle is organised into figure of eight bands that squeeze the ventricular chamber forcefully in a way most effective for ejection through the outflow valve. The apex of the heart contracts first and relaxes last to prevent back flow . 3.the epicardium, is the visceral layer of the serous pericardium. 4 Physiology of cardiovascular system 2 lecture 1, Dr.Abdul-Aziz Ahmed Figure:the wall of the heart. Heart valves For the heart to function effectively, blood flow must occur in a one-way direction, moving forward through the chambers of the right heart to the lungs and then through the chambers of the left heart to the systemic circulation. This unidirectional flow is provided by the heart’s valves: 1.The atrioventricular (AV) valves control the flow of blood between the atria and the ventricles . The thin edges of the AV valves form cusps, two on the left side of the heart (i.e., bicuspid or mitral valve) and three on the right side (i.e., tricuspid valve). The AV valves are supported by the papillary muscles, which project from the wall of the ventricles, and the chordae tendineae, which attach to the valve. Contraction of the papillary muscles at the onset of systole ensures closure by producing tension on the leaflets of the AV valves before the full force of ventricular contraction pushes against them. 2.The aortic and pulmonic valves control the movement of blood out of the ventricles. Because of their halfmoon shape, they often are referred to as the semilunar valves. The semilunar valves have three little teacup-shaped leaflets. These cuplike structures collect the retrograde, or backward, flow of blood that occurs toward the end of systole, enhancing closure. 5 Physiology of cardiovascular system 2 lecture 1, Dr.Abdul-Aziz Ahmed Figure:Heart valves Coronary Circulation There are two main coronary arteries, the left and the right, which arise from the coronary sinus just above the aortic valve ; 1. The left coronary artery divides into the anterior descending and circumflex branches. The left anterior descending artery passes down between the two ventricles, it supply the left ventricle, the anterior portion of the interventricular septum and the anterior papillary muscle of the left ventricle. The circumflex branch of the left coronary artery passes to the left and moves posteriorly in the groove that separates the left atrium and ventricle, giving off branches that supply the left lateral wall of the left ventricle. 2.The right coronary artery lies in the right atrioventricular groove, and its branches supply the right ventricle,the sinoatrial node. The right coronary artery usually moves to the back of the heart, where it forms the posterior descending artery, which supplies the posterior portion of the heart (the interventricular septum, atrioventricular node, and posterior papillary muscle). During systole, contraction of the cardiac muscle compresses the intramyocardial vessels specially through the subendocardial vessels. Thus, subendocardial region is in increased risk of ischemia and infarction specially when diastolic pressure is low, and when a rapid heart rate decreases the time spent in diastole. Because the 6 Physiology of cardiovascular system 2 lecture 1, Dr.Abdul-Aziz Ahmed perfusion of the subendocardial plexus occur mainly during diastole. Heart muscle relies primarily on fatty acids and aerobic metabolism to meet its energy needs. Blood flow usually is regulated by the need of the cardiac muscle for oxygen. Even under normal resting conditions, the heart extracts and uses 60% to 80% of oxygen in blood flowing through the coronary arteries, compared with the 25% to 30% extracted by skeletal muscle. Because there is little oxygen reserve in the blood, myocardial ischemia develops when the coronary arteries are unable to dilate and increase blood flow during periods of increased activity or stress. Figure: Coronary circulation. Intrinsic Control of Heart beat The rhythmical contraction of the heart is due to the intrinsic conduction system of the heart, which consist of: 1. SA (sinoatrial) node The sinoatrial (SA) node is the normal pacemaker of the heart and the origin of each normal heartbeat. The SA node is a collection of specialized myocytes near the site where the superior vena cava enters in the wall of the right atrium.The depolarization begins in the sinoatrial node (SA node), spread rapidly throughout the atria via gap junctions between adjacent myocytes. 2.Atrioventricular Node(A-V node) The atrioventricular (AV) node is the only electrical communication between the atria and the ventricles. It is characterized by very slow electrical conduction, 7 Physiology of cardiovascular system 2 lecture 1, Dr.Abdul-Aziz Ahmed ensuring that atrial contraction is completed before the ventricles are activated. The AV node is continuous with the atrioventricular bundle (bundle of His). 3.Atrioventricular bundle (bundle of His) The AV bundle carry signals from atrium to the ventricles, in the ventricles the AV bundle divide into right and left bundle branch, these branches then divide into an extensive network of Purkinje fibers. 4. Purkinje fibers Specialized conducting fibers that transmit electrical signals very rapidly to all parts of the ventricular myocardium. Spread of electrical signals along the heart The SA node, other atrial centres, AV node, and bundle of His all have inherent pacemaker activity. The SA node has the highest rate of spontaneous depolarization and therefore suppresses the other pacemakers. In the denervated heart, the SA node discharges at a rate of approximately 100 times min21. Vagal tone leads to a lower heart rate in healthy subjects at rest. From the SA node, impulses spread throughout the atria to the AV node at a rate of 1 ms-1. The AV node is the only means of electrical connection between the atria and the ventricles. Conduction here is slow (approximately 0.05 ms-1). This means that the AV node will only transmit a maximum of 220 impulses min21, so protecting the ventricles from high rates of atrial depolarization. Depolarization spreads from the AV node to the bundle of His in the interventricular septum. The bundle splits into right and left bundle branches, supplying the respective ventricles. The left bundle itself divides into anterior and posterior divisions. From the bundle branches, impulses travel through the ventricular muscle via a network of Purkinje fibres, at a velocity of 1–4 ms-1. The conducting system is arranged so that the apices of the ventricles contract before the bases, propelling blood out of the chambers. 8 Physiology of cardiovascular system 2 lecture 1, Dr.Abdul-Aziz Ahmed Figure: the intrinsic conduction system of the heart. Extrinsic Innervations of the Heart The excitatory and conductive system of the heart receive innervations from both division of autonomic nervous system. Although the basic heart rate is set by the intrinsic conduction system, fibers of the autonomic nervous system can modify the heart beat : 1. The sympathetic nervous system (the “accelerator”) increases both the rate and the force of heartbeat. 2.parasympathetic slows the heart rate and force of contraction. 9 Physiology of cardiovascular system 2 lecture 1, Dr.Abdul-Aziz Ahmed Figure:extrinsic innervation of the heart. Cellular anatomy of ventricular muscle Cardiac muscle is striated and has an intracellular sarcomere structure that is very similar to that of skeletal muscle. Myocytes are surrounded by sarcolemma (plasma membrane), with transverse tubules (T tubules) that extend into the cell interior. Sarcoplasmic reticulum is associated with every T tubule. The cardiac muscle cells (myocytes) form a highly branched and interdigitated network. Myocyte connections occur at structures called intercalated disks. The presence of gap junctions in intercalated disks accounts for high electrical conductance (low resistance) between myocytes. A single adequate stimulus for action potential in one myocyte results in the rapid spread of excitation to all myocytes via gap junctions. This is known as the all-or-none electrical response of the heart. 10 Physiology of cardiovascular system 2 lecture 1, Dr.Abdul-Aziz Ahmed Figure: Cellular anatomy of ventricular muscle. Ventricular muscle action potential The cardiac action potential is very different to that seen in nerves. It has a prolonged plateau phase lasting around 300 ms compared with 1 ms in nerves. The cardiac action potential has five phases: Phase 0 is the initial rapid upstroke that occurs immediately after stimulation. During phase 0, membrane permeability to potassium decreases and fast sodium channels open, producing rapid depolarization from - 90 mV to + 30 mV Phase 1 During phase 1, there is partial repolarization, because of a decrease in sodium permeability. Phase 2, is the plateau phase of the cardiac action potential. Membrane permeability to calcium increases during this phase, maintaining depolarization and prolonging the action potential. Membrane permeability to calcium decreases somewhat towards the end of phase 2, and the plateau is partially maintained by an inward sodium current. Sodium flows into the cell through the sodium–calcium exchanger. The exchanger transfers three sodium ions into the cell in exchange for one calcium ion flowing out, and so produces a net inward positive current. Much of the calcium influx in the plateau phase occurs through L-type (long opening) calcium channels. Increased activation of L-type channels occurs with catecholamine exposure, whilst they are blocked by calcium channel antagonists such as verapamil. Phase 3 is the repolarization of membrane potential back to the resting value. As calcium channels inactivate towards the end of the plateau phase, an inward potassium current produces repolarization in phase 3. Phase 4 is the interval between action potentials when the ventricular muscles are at their stable resting membrane potential. The resting membrane potential in phase 4 is approximately -90 mV. This is produced mainly by the selective permeability of the cell membrane to potassium and the concentration gradient 11 Physiology of cardiovascular system 2 lecture 1, Dr.Abdul-Aziz Ahmed for potassium that exists across the cell membrane and is close to the Nernst equilibrium potential for potassium. The cardiac action potential lasts about 300 ms. For the vast majority of this time, the cell is absolutely refractory to further stimulation. In other words, a further action potential will not be generated until repolarization is virtually complete. This prevents tetany from occurring. If a supramaximal stimulus occurs during the relative refractory period, the resultant action potential has a slower rate of depolarization and is of smaller amplitude than normal, producing a much weaker contraction than normal. Fig. The cardiac action potential. Phase 0—depolarization because of the opening of fast sodium channels. Potassium flux also decreases. Phase 1—partial repolarization because of a rapid decrease in sodium ion passage as fast sodium channels close. Phase 2—plateau phase in which the movement of calcium ions out of the cell, maintains depolarization. Phase 3— repolarization, sodium, and calcium channels all close and membrane potential returns to baseline. Phase 4—resting membrane potential (-90 mV), resulting from the activity of the Na/K ATPase pump which creates a negative intracellular potential because of the exchange of three sodium ions for only two potassium ions. 12 Physiology of cardiovascular system 2 lecture 1, Dr.Abdul-Aziz Ahmed Figure: Long refractory period in a cardiac muscle prevents tetanus. Pacemaker Cells The Pacemaker Cells characterized by their unique ability to generate action potentials spontaneously in the absence of input from the nervous system (autorhythmicity).This ability results from their unstable membrane potential, which starts at –60 mV and slowly drifts upward toward threshold. This unstable membrane potential is called a pacemaker potential. The pacemaker potential is seen in cells in the SA and AV nodes., it differs from the action potential of cardiac myocytes in that phases 1 and 2 are absent. The pacemaker potential is produced by a decrease in membrane permeability to potassium, a slow inward current because of calcium influx via T-type (transient) calcium channels, and an increased sodium current because of sodium– calcium exchange. Once the threshold potential is reached, L-type calcium channels open, calcium ions enter the cell, and depolarization occurs. In contrast to the cardiac myocyte action potential, there is no inward movement of sodium ions during depolarization. Repolarization (phase 3 of the action potential) occurs because of an increase in potassium permeability. At the SA node, potassium permeability can be further enhanced by vagal stimulation. This has the effect of hyperpolarizing the cell and reducing the rate of firing. Sympathetic stimulation has the opposite effect. 13 Physiology of cardiovascular system 2 lecture 1, Dr.Abdul-Aziz Ahmed Fig. The pacemaker potential. Phases 1 and 2 do not occur. Phase 4— pacemaker potential. Because of slow inward current of sodium and a voltage gated increase in calcium conductance (via T channels) Phase 0— depolarization. As opposed to the ventricular muscle action potential, this occurs because of voltage gated calcium channels opening. Phase 3— repolarization. This is because of a decrease in potassium. Excitation-contraction coupling This is the process linking electrical excitation to contraction. Calcium has an essential role in this process; a raised intracellular calcium concentration is the trigger that activates contraction. The intracellular calcium ion concentration in the cardiac myocyte at rest is 0.0001 mM litre-1 and that in the extracellular fluid is 1.2 mM litre-1. During the plateau phase of the action potential, calcium ions flow down this steep concentration gradient and enter the myocyte. Most of this calcium enters through the L-type channels, located primarily at sarcolemmal/sarcoplasmic reticulum 14 Physiology of cardiovascular system 2 lecture 1, Dr.Abdul-Aziz Ahmed junctions. The influx of calcium triggers the release of further calcium from the sarcoplasmic reticulum via ryanodine receptors. This calcium-triggered calcium release is in contrast to skeletal muscle, where the action potential triggers calcium release directly. Free intracellular calcium interacts with the C subunit of troponin. This leads to a configuration change in the troponin/tropomyosin complex, allowing actin to interact with myosin. Cross bridge cycling occurs, leading to a shortening of the sarcomere and resultant muscular contraction. As intracellular calcium concentrations decrease during repolarization, calcium dissociates from troponin as intracellular calcium concentration decreases, resulting in relaxation. Diastolic relaxation is an active (ATP-dependent) process. Calcium transport out of the cytosol occurs via a sarcoplasmic reticulum Ca2-ATPase, through sarcolemmal Na/Ca2 exchange, via a sarcolemmal Ca2-ATPase, and finally by utilizing a mitochondrial Ca2 uniport. The strength of a contraction may be varied by increasing the amount of free intracellular calcium, by altering the sensitivity of the myofilaments to calcium, or both. The latter occurs during stretching of the myofilaments and is responsible for the Frank– Starling mechanism (discussed later). Myofilament calcium sensitivity is reduced by acidosis. High concentrations of phosphate and magnesium also impair cardiac function. Catecholamines activate beta-adrenergic receptors in the heart to produce a G-protein mediated increase in cAMP and enhanced activity of a cAMP-dependent protein kinase. This leads to the phosphorylation of calcium membrane channels, enhancing calcium entry into the cell. Phosphorylation of myosin also occurs, increasing the rate of cross bridge cycling. Catecholamines also increase the rate of re-uptake of calcium into the sarcoplasmic reticulum, thus aiding relaxation. Figure: Excitation-contraction coupling 15 Physiology of cardiovascular system 2 lecture 1, Dr.Abdul-Aziz Ahmed Digitalis Digitalis drugs that increase the availability of intracellular free Ca2+ cause an increase in myocardial contractility. Digitalis blocks the Na+/K+-ATPase on the myocyte of the cardiac cell membrane, yielding an increase in intracellular [Na+]. Increased intracellular [Na+] alters the electrochemical driving force for Na+/Ca2+ exchange and results in the reversal of the normal transport direction, bringing Ca2+ into the cell. Increased intracellular [Ca2+] causes more Ca2+ to be stored in the sarcoplasmic reticulum. Subsequent excitation releases more Ca2+, resulting in increased myocardial contraction (a positive inotropic effect). Metabolism and energetic of cardiac muscle The oxygen consumption of the beating heart is on average 9 ml per 100 g min-1 at rest. This increases during exercise. Oxygen extraction from blood in the coronary circulation is high; therefore, an increase in oxygen demand must be met by an increase in coronary blood flow. The heart is very versatile in its use of metabolic substrates. Carbohydrate utilization accounts for 35–40% of total oxygen consumption. Glucose and lactate are used in roughly equal proportions. A small amount of energy may be derived from ketones; however, 60% of the basal energy requirement is provided by fat. The proportion of substrates utilized may vary depending on the nutritional state of the person. After a large meal containing glucose, more pyruvate and lactate are used. During periods of starvation, more fat is utilized. Insulin enhances glucose uptake into cardiac myocytes, and in untreated diabetes proportionally more fat is utilized. Normally ,1% of the energy used by the myocardium is produced by anaerobic metabolism. This proportion increases during periods of hypoxaemia; however, lactic acidosis impairs myocardial function and can ultimately lead to myocardial cell death. 16