* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download 15.1 Amines
Survey
Document related concepts
Transcript
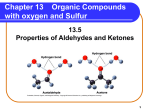
Chapters 16-19 (short version) • AMINES, ALDEHYDES, KETONES, CARBOXYLIC ACIDS, ESTERS, ANHYDRIDES 16-1 AMINES Ch 16……what to know 16-2 Structure and Classification Amines are classified as 1°, 2°, or 3° depending on the number of carbon groups bonded to nitrogen. CH3 -NH2 Methylamin e (a 1° amine) H CH3 -N-CH3 D imeth ylamine (a 2° amine) CH3 CH3 -N-CH3 Trimeth ylamine (a 3° amine) Aliphatic amine: All carbons bonded to nitrogen are derived from alkyl groups. See the three above. Aromatic amine: One or more of the groups bonded to nitrogen are aryl groups. NH2 Anilin e (a 1° aromatic amine) H N-CH3 N-Methylan iline (a 2° aromatic amine) CH3 CH2 -N-CH3 Ben zyldimethylamine (a 3° aliph atic amin e) 16-3 Structure and Classification • Heterocyclic amine: An amine in which the nitrogen atom is part of a ring. • Heterocyclic aliphatic amine: A heterocyclic amine in which the ring is saturated (has no C=C bonds). • Heterocyclic aromatic amine: The amine nitrogen is part of an aromatic ring. N N H N H Pyrrolidin e Pip eridin e (h eterocyclic aliph atic amin es) N N N N N N N N H H Pyridin e Pyrimidin e Imid azole Purine (heterocyclic aromatic amines ) *DNA bases are classified as Purines and Pyrimidines (only 2 specific ones you have to know for test) 16-4 Nomenclature IUPAC names • We derive IUPAC names for aliphatic amines just as we did for alcohols. • Drop the final -e of the parent alkane and replace it by -amine. • Use a number to locate the amino group on the parent chain. NH2 CH3 CHCH3 2-Propanamine NH2 H2 N Cyclohexanamine NH2 1,6-Hexan ediamine 16-5 Nomenclature • IUPAC names (cont’d) • IUPAC nomenclature retains the common name aniline for C6H5NH2, the simplest aromatic amine. • Name simple derivatives of aniline by using numbers to locate substituents or, alternatively, use the prefixes ortho (o), meta (m), and para (p). • Several derivatives of aniline have common names that are still widely used; among them is toluidine: NH2 NH2 NH2 CH3 A niline NO2 4-N itroaniline (p-N itroan iline) 3-Methylaniline (m-Tolu idine) 16-6 Nomenclature IUPAC names (cont’d) • Name unsymmetrical secondary and tertiary amines as N-substituted primary amines. • Take the largest group bonded to nitrogen as the parent amine. • Name the smaller group(s) bonded to nitrogen, and show their location on nitrogen by using the prefix N(indicating that they are bonded to nitrogen). CH3 NHCH3 N-Methylanilin e N CH3 N,N-Dimethylcyclopentan amine 16-7 Nomenclature Common names • For most aliphatic amines, list the groups bonded to nitrogen in alphabetical order in one word ending in the suffix -amine. NH2 Propylamine NH2 NH2 N sec-Butylamin e D ieth ylmethylamin e Cyclohexylamine CW- Name the above by IUPAC 16-8 Physical Properties Like ammonia, low-molecular-weight amines have very sharp, penetrating odors. • Trimethylamine, for example, is the pungent principle in the smell of rotting fish. • Two other particularly pungent amines are 1,4butanediamine (putrescine) and 1,5-pentanediamine (cadaverine). H 2N N H2 1,4-Butanediamine (Putrescine) H 2N N H2 1,5-Pentanediamine (Cadaverine) 16-9 Physical Properties Amines are polar compounds: • Both 1° and 2° amines have N-H bonds, and can form hydrogen bonds with one another. • 3° Amines have no N-H bond and cannot form hydrogen bonds with one another. 16-10 Physical Properties • An N-H---N hydrogen bond is weaker than an O-H---O hydrogen bond, because the difference in electronegativity between N and H (3.0 - 2.1 = 0.9) is less than that between O and H (3.5 - 2.1 = 1.4). • We see the effect of hydrogen bonding between molecules of comparable molecular weight by comparing the boiling points of ethane, methanamine, and methanol. MW (amu) bp (°C) CH3 CH3 CH3 NH2 CH3 OH 30.1 -88.6 31.1 -6.3 32.0 65.0 16-11 Physical Properties • All classes of amines form hydrogen bonds with water and are more soluble in water than are hydrocarbons of comparable molecular weight. • Most low-molecular-weight amines are completely soluble in water. • Higher-molecular-weight amines are only moderately soluble in water or are insoluble. 16-12 Basicity of Amines Like ammonia, amines are weak bases, and aqueous solutions of amines are basic. • The acid-base reaction between an amine and water involves transfer of a proton from water to the amine. H + CH3 -N-H :O-H H Meth ylammoniu m hydroxide : : : : H CH3 -N: + H-O-H H Methylamin e (a bas e) (*Conjugate acid) *lone pr of e- accepts H+ 16-13 Basicity of Amines • Given the basicities of amines, we can determine which form of an amine exists in body fluids, say blood. • In a normal, healthy person, the pH of blood is approximately 7.40, which is slightly basic. • If dissolved in blood, it is present predominantly as its protonated (conjugated acid) form. HO NH2 HO D opamine HO NH3 + HO Con jugate acid of d op amine (the major form p res ent in b lood p lasma) 16-14 Reactions of Amines The most important chemical property of amines is their basicity. • Amines, whether soluble or insoluble in water, react quantitatively with strong acids to form water-soluble salts. HO H HO HO NH2 + HCl HO (R)-N orepinep hrin e (on ly s ligh tly solub le in w ater) H2 O HO H + - NH3 Cl HO (R)-N orep ineph rine h yd roch loride (a w ater-soluble s alt) HW- 1-What is norepinephrine?; 2-What is epinephrine?; 3- Explain their functions in the body 16-15 Amines Another connection to Biology/Biochemistry: How are amines applicable to proteins? 16-16 Summary: Amines •Amines –NH2. There are primary, secondary and tertiary amines. •Nomenclature: IUPAC-Name amines like alcohols; Common- end with “amine” •EX-CH3NH2-IUPAC: methanamine; Common: Methyl amine (both are primary) •Aliphatic amines- Have alkyl groups (methyl amine) •Aromatic amines- one or more of the groups bonded to nitrogen are aryl groups Ex-aniline (aminobenzene); •Heterocyclic- N is part of the ring (ex-purines, pyrimidines) •Amino acids-monomers of proteins. Have both amino group and Carboxyl group •Reactions: Amines are weak bases. The lone electron pair attracts H+ (Remember-acids donate H+ and bases accept H+) •Physical properties- smaller ones are polar, but weaker than alcohols (O eneg is 3.5; N is 3.0) 16-17 ALDEHYDES & KETONES CH 17 16-18 Structure The functional group of an aldehyde is a carbonyl group bonded to a hydrogen atom. • In methanal, the simplest aldehyde (formaldehyde), the carbonyl group is bonded to two hydrogens. • In other aldehydes, it is bonded to one hydrogen and one carbon group. The functional group of a ketone is a carbonyl group bonded to two carbon groups. O O O HCH CH3 CH CH3 CCH3 Meth anal Ethanal Prop anone (Formaldehyde) (Acetaldeh yd e) (Aceton e) 17-19 Nomenclature IUPAC names for aldehydes • To name an aldehyde, change the suffix -e of the parent alkane to -al. • Because the carbonyl group of an aldehyde can only be at the end of a parent chain and numbering must start with it as carbon-1, there is no need to use a number to locate the aldehyde group. • For unsaturated aldehydes, indicate the presence of a carbon-carbon double bond by changing the ending of the parent alkane from -ane to -enal. Numbering the carbon chain begins with the aldehyde carbonyl carbon. Show the location of the carbon-carbon double bond by the number of its first carbon. 17-20 Nomenclature • The IUPAC system retains common names for some aldehydes, including these three. O CHO CHO H OCH3 t rans-3-Phenyl-2-prop enal (Cinn amald ehyd e; in oil of cin namon) Ben zaldehyde (in almond s) OH Van illin (from van illa bean s) 17-21 Nomenclature IUPAC names for ketones. • The parent alkane is the longest chain that contains the carbonyl group. • Indicate the presence of the carbonyl group by changing the -ane of the parent alkane -one. • Number the parent chain from the direction that gives the carbonyl carbon the smaller number. • The IUPAC retains the common name acetone for 2propanone. O O 1 Acetone O 2 3 4 5 1 6 5-Meth yl-3-h exanone 2 2-Methylcycloh exanone 17-22 Nomenclature To name an aldehyde or ketone that also contains an -OH (hydroxyl) or -NH2 (amino) group: • Number the parent chain to give the carbonyl carbon the lower number. • Indicate an -OH substituent by hydroxy-, and an -NH2 substituent by amino-. • Hydroxyl and amino substituents are numbered and alphabetized along with other substituents. O OH O 5 4 3 1 H 3-Hydroxy-4-meth ylp entanal 6 4 3 2 1 NH2 3-Amino-4-ethyl-2-h exanone 17-23 Nomenclature Common names The common name for an aldehyde is derived from the common name of the corresponding carboxylic acid. • Drop the word "acid" and change the suffix -ic or -oic to -aldehyde. • Name each alkyl or aryl group bonded to the carbonyl carbon as a separate word, followed by the word "ketone”. Alkyl or aryl groups are generally listed in order of increasing molecular weight. O O O CH3 CH CH3 COH Acetaldehyde Acetic acid O Methyl ethyl ketone Ethyl isopropyl ketone 17-24 Physical Properties A C=O bond is polar, with oxygen bearing a partial negative charge and carbon bearing a partial positive charge. • Therefore, aldehydes and ketones are polar molecules. • Figure 17.1 The polarity of a carbonyl group. 17-25 Physical Properties • In liquid aldehydes and ketones, there are weak intermolecular attractions between the partial positive charge on the carbonyl carbon of one molecule and the partial negative charge on the carbonyl oxygen of another molecule. • No hydrogen bonding is possible between aldehyde or ketone molecules. • Aldehydes and ketones have lower boiling points than alcohols and carboxylic acids, compounds in which there is hydrogen bonding between molecules. See the table on the next screen. 17-26 Physical Properties Name diethyl ether pentane butanal 2-butanone 1-butanol propanoic acid bp Molecular Structural Formula Weight (amu) (°C) CH3 CH2 OCH 2 CH3 34 74 CH3 CH2 CH2 CH2 CH 3 72 36 CH3 CH2 CH2 CHO 72 76 72 80 CH3 CH2 COCH3 74 117 CH3 CH2 CH2 CH2 OH 74 141 CH3 CH2 COOH • Formaldehyde, acetaldehyde, and acetone are infinitely soluble in water. • Aldehydes and ketones become less soluble in water as the hydrocarbon portion increases in size. • Flammable • Simple ones are toxic (ex- formaldehyde/methanal) • FYI- aldehydes found in some flavorings (cinnamon,17-27 vanilla); dyes; perfumes Properties • Reduction (hydrogenation) of aldehydes yields primary alcohol; reduction of ketone makes a secondary alcohol • Ketones cannot be further oxidized • Simple ketones are excellent solvents. Dissolve in both polar and non polar substances. Simple ones low in toxicity • FYI- Some ketones found in some perfumes, camphor 17-28 Oxidation • Aldehydes are oxidized to carboxylic acids by a variety of oxidizing agents, including potassium dichromate. O O H Hexanal K2 Cr2 O 7 H2 SO 4 OH Hexanoic acid • Liquid aldehydes are so sensitive to oxidation by O2 in the air that they must be protected from contact with air during storage. O C 2 H Benzaldehyde O C + O2 2 OH Benzoic acid 17-29 Oxidation • Ketones resist oxidation by most oxidizing agents, including potassium dichromate and molecular oxygen. • **Not on test: Tollens’ reagent is specific for the oxidation of aldehydes. If done properly, silver deposits on the walls of the container as a silver O mirror. R-C-H + 2 Ag( NH3 ) 2 + + 3 OHA ldehyde Tollens' reagen t O R-C-O- + 2 Ag + 4 NH3 + 2 H2 O Carboxylic Silver an ion mirror 17-30 Reduction • The carbonyl group of an aldehyde or ketone is reduced to an -CHOH group by hydrogen in the presence of a transition-metal catalyst. • Reduction of an aldehyde gives a primary alcohol. • Reduction a ketone gives a secondary alcohol. O H + H2 Pentanal O + H2 Cyclopen tan on e transition metal catalyst tran sition metal catalyst OH 1-Pentanol OH Cyclopen tanol 17-31 Reduction *specifics of slides 32-34 NOT on THIS test. You need to correct your copy! The most common laboratory reagent for the reduction of an aldehyde or ketone is sodium borohydride, NaBH4. • This reagent contains hydrogen in the form of hydride ion, H:-. • In a hydride ion, hydrogen has two valence electrons and bears a negative charge. • In a reduction by sodium borohydride, hydride ion adds to the partially positive carbonyl carbon which leaves a negative charge on the carbonyl oxygen. • Reaction of this intermediate with aqueous acid gives the alcohol. 17-32 Reduction - H: + C O H C O - H3 O + H C O-H Hydride ion O O- NaBH4 H H3 O+ O-H H • Reduction by NaBH4 does not affect a carbon-carbon double bond or an aromatic ring. O C H 1 . NaBH4 CH2 OH 2 . H2 O Cin namaldehyde Cinnamyl alcoh ol 17-33 Reduction • In biological systems, the agent for the reduction of aldehydes and ketones is the reduced form of nicotinamide adenine dinucleotide, abbreviated NADH (Section 27.3B) • This reducing agent, like NaBH4, delivers a hydride ion to the carbonyl carbon of the aldehyde or ketone. • Reduction of pyruvate, the end product of glycolysis, by NADH gives lactate (anaerobic respiration) O CH3 -C-COO Pyruvate NADH OCH3 -C-COO H + H3 O O-H CH3 -C-COO H Lactate 17-34 Addition of Alcohols Addition of a molecule of alcohol to the carbonyl group of an aldehyde or ketone forms a hemiacetal (a half-acetal). • The functional group of a hemiacetal is a carbon bonded to one -OH group and one -OR group. • In forming a hemiacetal, -H of the alcohol adds to the carbonyl oxygen and -OR adds to the carbonyl carbon. O H C + O-CH2 CH3 H Benzaldehyde Ethanol O-H C OCH2 CH3 H A hemiacetal *For this test, just know that Aldehyde + Alcohol Hemiacetal *Hemiacetal product contains –OH and ether 17-35 Addition of Alcohols • Hemiacetals are generally unstable and are only minor components of an equilibrium mixture except in one very important type of molecule. • When a hydroxyl group is part of the same molecule that contains the carbonyl group and a five- or sixmembered ring can form, the compound exists almost entirely in a cyclic hemiacetal form. O 5 4 3 2 1 H O-H 4-Hyd roxypentanal redraw to show the -OH an d -CHO clos e to each oth er 3 2 1 4 5 O H C H O H O-H O A cyclic hemiacetal • -H of the alcohol adds to the carbonyl oxygen and –O (from –OH) adds to the carbonyl carbon, closing the ring. *This is how glucose becomes cyclic in our bodies! (Next unit) 17-36 Carboxylic Acids • Ch 18 17-37 Carboxylic Acids • Carboxylic acids: another class of organic compounds containing the carbonyl group. • The functional group of a carboxylic acid is a carboxyl group, which can be represented in any one of three ways. O C-OH COOH CO2 H 18-38 Nomenclature IUPAC names • For an acyclic carboxylic acid, take the longest carbon chain that contains the carboxyl group as the parent alkane. • Drop the final -e from the name of the parent alkane and replace it by -oic acid. • Number the chain beginning with the carbon of the carboxyl group. • Because the carboxyl carbon is understood to be carbon 1, there is no need to give it a number. 18-39 Nomenclature • In these examples, the common name is given in parentheses (do not have to know for test). O 6 O 1 3 OH Hexanoic acid (Caproic acid) 1 OH 3-Methylbutanoic acid (Isovaleric acid) • An -OH substituent is indicated by the prefix hydroxy-; an -NH2 substituent by the prefix amino-. OH 5 O 1 OH 5-Hydroxyhexan oic acid H2 N COOH 4-A min ob enzoic acid 18-40 Nomenclature • To name a dicarboxylic acid, add the suffix -dioic acid to the name of the parent alkane that contains both carboxyl groups; thus, -ane becomes -anedioic acid. • The numbers of the carboxyl carbons are not indicated because they can be only at the ends of the chain. O HO 2 O 1 3 OH HO O 1 OH O Ethan edioic acid Prop aned ioic acid (Malonic acid ) (Oxalic acid ) O HO 4 O 5 1 OH O Butaned ioic acid (Succinic acid) HO O O 1 OH Pen tanedioic acid (Glutaric acid) HO 6 1 OH O Hexan edioic acid (Ad ipic acid) 18-41 Name these 18-42 Nomenclature Structure HCOOH CH3 COOH CH3 CH2 COOH CH3 (CH2 ) 2 COOH CH3 (CH2 ) 3 COOH CH3 (CH2 ) 4 COOH CH3 (CH2 ) 6 COOH CH3 (CH2 ) 8 COOH CH3 (CH2 ) 1 0 COOH CH3 (CH2 ) 1 2 COOH CH3 (CH2 ) 1 4 COOH CH3 (CH2 ) 1 6 COOH CH3 (CH2 ) 1 8 COOH IU PAC N ame (acid) methanoic ethan oic propanoic bu tanoic pen tanoic hexan oic octanoic decanoic dodecanoic tetradecan oic hexad ecanoic octadecanoic eicosan oic Common N ame D erivation formic acetic propionic bu tyric valeric cap roic cap rylic cap ric lauric myristic palmitic stearic arachid ic Latin : formica, ant Latin : acet um, vinegar Greek: propion, firs t fat Latin : buty rum, b utter Latin : valere, to be s trong Latin : caper, goat Latin : caper, goat Latin : caper, goat Latin : laurus , laurel Greek: my ris tikos, fragrant Latin : palma, palm tree Greek: st ear, solid fat Greek: arachis, p eanut 18-43 Physical Properties The carboxyl group contains three polar covalent bonds; C=O, C-O, and O-H. • The polarity of these bonds determines the major physical properties of carboxylic acids. 18-44 Physical Properties • Carboxylic acids have significantly higher boiling points than other types of organic compounds of comparable molecular weight. • Their higher boiling points are a result of their polarity and the fact that hydrogen bonding between two carboxyl groups creates a dimer that behaves as a higher-molecular-weight compound. hydrogen bondin g betw een tw o molecules H3 C O + H O C C O H + CH3 O - 18-45 Physical Properties Carboxylic acids are more soluble in water than are alcohols, ethers, aldehydes, and ketones of comparable molecular weight. Boilin g Solubility Molecular Poin t Weigh t (°C) (g/100 mL H 2O) Structu re N ame CH3 COOH CH3 CH2 CH2 OH CH3 CH2 CHO acetic acid 60.5 1-prop anol prop anal CH3 (CH2 ) 2 COOH butan oic acid CH3 (CH2 ) 3 CH2 OH 1-pentan ol pentan al CH3 (CH2 ) 3 CHO 60.1 58.1 118 97 48 infinite infinite 16 88.1 88.1 86.1 163 137 103 infinite 2.3 slight 18-46 Fatty Acids Fatty acids: Long chain carboxylic acids derived from animal fats, vegetable oils, or phospholipids of biological membranes. • More than 500 have been isolated from various cells and tissues. • Most have between 12 and 20 carbons in an unbranched chain. • In most unsaturated fatty acids, the cis isomer predominates; trans isomers are rare. 18-47 Fatty Acids Table 18.3 The Most Abundant Fatty Acids in Animal Fats, Vegetable Oils, and Biological Membranes. Carbon Atoms: Double Bonds * Structure Saturated Fatty Acids 12:0 CH 3 ( CH2 ) 1 0 COOH Common Name Melting Point (°C) lauric acid 44 14:0 CH 3 ( CH2 ) 1 2 COOH myristic acid 58 16:0 CH 3 ( CH2 ) 1 4 COOH palmitic acid 63 18:0 CH 3 ( CH2 ) 1 6 COOH stearic acid 70 arachidic acid 77 CH 3 ( CH2 ) 1 8 COOH 20:0 Uns aturated Fatty Acids 16:1 CH3 ( CH2 ) 5 CH= CH( CH2 ) 7 COOH 18:2 palmitoleic acid CH3 ( CH2 ) 7 CH= CH( CH2 ) 7 COOH oleic acid CH3 ( CH2 ) 4 ( CH= CHCH2 ) 2 ( CH 2 ) 6 COOH linoleic acid 18:3 CH3 CH2 ( CH= CHCH 2 ) 3 ( CH 2 ) 6 COOH 20:4 CH3 ( CH2 ) 4 ( CH= CHCH2 ) 4 ( CH 2 ) 2 COOH arachidonic acid 18:1 linolenic acid * The first number is the number of carbons in the fatty acid; the s econd is the number of carbon-carbon double bonds in its hydrocarbon chain. 1 16 -5 -11 -49 18-48 Fatty Acids Unsaturated fatty acids generally have lower melting points than their saturated counterparts. COOH Stearic acid (18:0) (mp 70°C) COOH Oleic acid (18;1) (mp 16°C) COOH Linoleic acid (18:2) (mp-5°C) COOH Linolenic acid (18:3) (mp -11°C) 18-49 Fatty Acids Saturated fatty acids are solids at room temperature. • The regular nature of their hydrocarbon chains allows them to pack together in such a way as to maximize interactions (by London dispersion forces) between their chains. COOH COOH COOH COOH COOH 18-50 Fatty Acids In contrast, all unsaturated fatty acids are liquids at room temperature because the cis double bonds interrupt the regular packing of their hydrocarbon chains. COOH COOH COOH COOH COOH 18-51 Link to Biology • Amino Acids (monomers of proteins) have carboxyl groups, as well as amino groups (see amine section of PPt) 18-52 Soaps • Natural soaps are sodium or potassium salts of fatty acids. • They are prepared from a blend of tallow and palm oils (triglycerides). • Triglycerides are triesters of glycerol. (Esters will be covered shortly) • The solid fats are melted with steam and the water insoluble triglyceride layer that forms on the top is removed. 18-53 Soaps Preparation of soaps begins by boiling the triglycerides with NaOH. The reaction that takes place is called saponification (Latin: saponem, “soap”). Boiling with KOH gives a potassium soap. O O CH2 OCR saponification + 3 N aOH RCOCH O CH2 OCR A triglyceride ( a triester of glycerol) CH2 OH CHOH + O + 3 RCO N a CH2 OH 1,2,3-Propanetriol Sodium soaps (Glycerol; glycerin) What triglyceride did you use in your lab? 18-54 Soaps In water, soap molecules spontaneously cluster into micelles, a spherical arrangement of molecules such that their hydrophobic parts are shielded from the aqueous environment, and their hydrophilic parts are in contact with the aqueous environment. 18-55 Soaps When soaps and dirt, such as grease, oil, and fat stains are mixed in water, the nonpolar hydrocarbon inner parts of the soap micelles “dissolve” the nonpolar substances. 18-56 Acidity of Carboxylic Acids Carboxylic acids are relatively weak acids, however, substituents of high electronegativity, especially -OH, Cl, and -NH3+, near the carboxyl group increase the acidity of carboxylic acids. Formula: CH3 COOH N ame: pK a: Acetic acid 4.76 ClCH2 COOH Cl2 CHCOOH Cl3 CCOOH Chloroacetic D ichloroacetic Trich loroacetic acid acid acid 2.86 1.48 0.70 In creasing acid strength 18-57 Ch 19 • Esters, Anhydrides, Amides 18-58 Carboxyl Derivatives Three classes of compounds derived from carboxylic acids: anhydrides, esters, and amides. • Each is related to a carboxyl group by loss of H2O (DEHYDRATION SYNTHESIS). O RCOH A carboxylic acid O O RCOCR' An anhydride -H2 O O O RC- OH H- OCR' O RCOR' An ester -H2 O O RCN H2 An amide -H2 O O O RC- OH H- OR' RC- OH H- NH 2 19-59 Fischer Esterification (Ch 19) Fischer esterification is one of the most commonly used methods for the preparation of esters. • In Fischer esterification, a carboxylic acid is reacted with an alcohol in the presence of an acid catalyst, most commonly concentrated sulfuric acid. O O H2 SO4 CH3 C-OH + H-OCH2 CH3 Ethanoic acid Ethanol (Acetic acid) (Ethyl alcohol) CH3 COCH2 CH3 + H2 O Ethyl ethanoate (Ethyl acetate) *-H is removed from alcohol and –OH from carboxylic acid ALCOHOL + ACID ESTER + WATER 18-60 Triglycerides • Glycerol (1,2,3 –propantriol) + 3 Fatty acidsTriglyceride +H2O • Triglycerides are esters 18-61 Esters Ch 19 • Alcohol + Acid Ester + Water • Many have pleasant, fruity odors • General formula: RCOOR • Naming: 2 words. First, name the alkyl group (next to the O), then name the parent (from the acid) and change the –ic ending to –ate • Ex- draw methyl ethanoate (ethanoate’s common name is acetate) 18-62 Anhydrides The functional group of an anhydride is two carbonyl groups bonded to the same oxygen. • The anhydride may be symmetrical (from two identical acyl groups), or mixed (from two different acyl groups). • To name an anhydride, drop the word "acid" from the name of the carboxylic acid from which the anhydride is derived and add the word "anhydride”. O O CH3 C-O-CCH3 Acetic anhydride *We will use anhydrides to synthesize aspirin in lab 19-63 Amides (will not test, just FYI) The functional group of an amide is a carbonyl group bonded to a nitrogen atom. • To name an amide, drop the suffix -oic acid from the IUPAC name of the parent acid, or -ic acid from its common name, and add -amide. • If the amide nitrogen is also bonded to an alkyl or aryl group, name the group and show its location on nitrogen by N- ; two alkyl or aryl groups by N,N-di-. O O CH3 CNH2 CH3 CNHCH3 Acetamide N -Methylacetamide (a 1° amide) (a 2° amide) O HCN(CH3 ) 2 N ,N-D imethylformamide (a 3° amide) 19-64 Hydrolysis The “reverse” of dehydration synthesis.Decomposition by the addition of water • Esters: hydrolyze very slowly, even in boiling water. • Hydrolysis becomes considerably more rapid, however, when the ester is heated in aqueous acid or base. • Hydrolysis of esters in aqueous acid is the reverse of Fischer esterification. O CH3 COCH2 CH3 Ethyl acetate + H2 O H + O CH3 COH + CH3 CH2 OH Acetic acid Ethanol 19-65 Hydrolysis of Esters (for lab only) • We can also hydrolyze an ester using a hot aqueous base, such as aqueous NaOH. • This reaction is often called saponification, a reference to its use in the manufacture of soaps. • The carboxylic acid formed in the hydrolysis reacts with hydroxide ion to form a carboxylic acid anion. • Each mole of ester hydrolyzed requires one mole of base. O H2 O CH3 COCH2 CH3+ N aOH heat Ethyl acetate Sodium hydroxide O CH3 CO - N a++ CH3 CH2 OH Sodium Ethanol acetate 19-66 Hydrolysis of Anhydrides • Carboxylic anhydrides, particularly the low-molecularweight ones, react readily with water (hydrolyze) to give two carboxylic acids. O O CH3 COCCH3 + H2 O Acetic anhydride O O CH3 COH + HOCCH3 Acetic acid Acetic acid 19-67 Hydrolysis of Amides (not on test) Amides require more vigorous conditions for hydrolysis in both acid and base than do esters. • Hydrolysis in hot aqueous acid gives a carboxylic acid and an ammonium ion. • Hydrolysis is driven to completion by the acid-base reaction between ammonia or the amine and the acid to form an ammonium ion. • Each mole of amide hydrolyzed requires one mole of acid. O CH3 CH2 CH2 CNH2 + H2 O + HCl Butanamide H2 O heat O + CH3 CH2 CH2 COH + NH4 Cl Butanoic acid 19-68 Hydrolysis of Amides • Hydrolysis of an amide in aqueous base gives a carboxylic acid salt and ammonia or an amine. • Hydrolysis is driven to completion by the acid-base reaction between the carboxylic acid and base to form a salt. • Each mole of amide hydrolyzed requires one mole of base. O CH3 CNH Acetanilide + NaOH H2 O heat O CH3 CO-Na+ + H2 N Sodium acetate Aniline 19-69 Reaction with Alcohols Anhydrides react with alcohols and phenols to give an ester and a carboxylic acid. O O CH3 COCCH3 + HOCH2 CH3 Acetic anhydride Ethanol O O CH3 COCH 2 CH 3 + HOCCH3 Ethyl acetate Acetic acid Aspirin is prepared by the reaction of salicylic acid with acetic anhydride. COOH OH Salicylic acid + O O CH3 C- O-CCH3 Acetic anhydride COOH OCCH 3 O Acetyls alicylic acid (Aspirin) + O CH3 C- OH Acetic acid 19-70 Reaction with Amines Anhydrides react with ammonia and with 1° and 2° amines to form amides. • Two moles of amine are required; one to form the amide and one to neutralize the carboxylic acid byproduct. O O CH3 C- O-CCH3 + N H3 O CH3 C- OH + N H3 O O CH3 C- O-CCH3 + 2 N H3 Acetic anhydride O O CH3 CN H2 + CH3 C- OH O CH3 CO - N H4 + O O CH3 CN H2 + CH3 CO - N H4 + Acetamide Ammonium acetate 19-71 Reaction with Amines • Esters react with ammonia and with 1° and 2° amines to form amides. O O OCH2 CH3 + N H3 Ethyl 2-phenyl acetate N H2 + CH3 CH2 OH 2-Phenylacetamide • Thus, an amide can be prepared from a carboxylic acid by first converting the carboxylic acid to an ester by Fischer esterification and then reaction of the ester with an amine. • Amides do not react with ammonia or with amines 19-72 Phosphoric Anhydrides The functional group of a phosphoric anhydride is two phosphoryl (P=O) groups bonded to the same oxygen atom. O O HO-P-O-P-OH OH OH O O O-P-O-P-O O- O- D iphosph oric acid (Pyroph os phoric acid) D iphosph ate ion (Pyroph os phate ion) O O O HO-P-O-P-O-P-OH OH OH OH Triphosp horic acid O O O O-P-O-P-O-P-O O O O Triph os phate ion 19-73 Phosphoric Esters • Phosphoric acid forms mono-, di-, and triphosphoric esters. • In more complex phosphoric esters, it is common to name the organic molecule and then indicate the presence of the phosphoric ester by either the word "phosphate" or the prefix phospho-. • Dihydroxyacetone phosphate and pyridoxal phosphate are shown as they are ionized at pH 7.4, the pH of blood plasma. O CH3 O-P-OH OCH3 D imethyl ph os phate CH2 OH CO O CH2 -O-P-O O Dih yd roxyacetone p hosphate O CHO HO CH2 O-P-O O H3 C N Pyridoxal ph os phate 19-74 Step-Growth Polymerization Step-growth polymers are formed by reaction between two molecules, each of which contains two functional groups. Each new bond is created in a separate step. • in this section, we discuss three types of step-growth polymers; polyamides, polyesters, and polycarbonates. 19-75 Polyamides Nylon-66 was the first purely synthetic fiber. • It is synthesized from two six-carbon monomers. remove H 2O H N O H N H OH + O H Hexanedioic acid 1,6-Hexanediamine (Adipic acid) (Hexamethylenediamine) HO heat - H2 O O O N H Nylon-66 (a polyamide) H N n 19-76 Polyamides The polyaromatic amide known as Kevlar is made from an aromatic dicarboxylic acid and an aromatic diamine. remove H 2O O nHOC O COH H H heat + N N -H2 O H H 1,4-Benzen edicarboxylic 1,4-Benzen ediamine acid (p-Phen ylened iamin e) (Terep hthalic acid) O C O CNH NH n Kevlar (a p olyaromatic amide) 19-77 Polyesters The first polyester involved polymerization of this diester and diol. remove CH 3 OH O OCH3 HO heat + CH3 O O Dimethyl terephthalate -CH3 OH OH 1,2-Ethaned iol (Ethylene glycol) O O O O n Poly(eth ylene tereph thalate) (D acron , Mylar) 19-78 Polycarbonates Lexan, the most familiar polycarbonate, is formed by reaction between the disodium salt of bisphenol A and phosgene. + remove N a Cl O CH3 + - Na O - O Na + CH3 D isodium salt of Bis phenol A + Cl Cl -NaCl Phosgen e O CH3 O CH3 Lexan (a p olycarbonate) O n 19-79 Hydrolysis • The “reverse” of dehydration synthesis • ESTERS: Ex 19-80