* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Polymerization
Survey
Document related concepts
Transcript
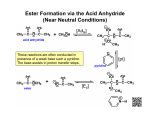
Polymerization 1. Polymerization is the formation of long carbon chains that occurs through a kind of chain reaction. It is a kind of addition reaction is one in which two or more molecules join together to give a single product. 2. Vocab a. Polymer – a large molecule consisting of repeating units. b. Repeating unit – recurring unit in a polymer c. Monomer – smallest molecule from which the polymer is made i. Naming Polymers: put poly- in front of the name of the monomer 3. There are many polymerization mechanisms. You need to know: a. Addition of alkenes: i. Polyethylene ii. polyvinylchloride b. Condensation of carboxylic acids and amines: i. nylons (amides), ii. peptides (amides) c. Condensation of carboxylic acids and alcohols i. polyesters Example 1: Addition of Alkenes 1. 3 steps in addition of alkenes d. initiation i. need a radical to start the reaction: Ra ii. radical: atom or compound w/ unpaired electron. It’s very reactive b/c it does not have the octet rule satisfied. iii. To form the radical heat is applied to a molecule with a O-O bond like peroxide or oxygen gas. 1. breaking the O-O bond will create two radicals e. Propagation i. Reaction of monomer unit, hundreds of times f. Termination i. Two chains come together so that radicals are consumed 2. Polyethylene a. Two kinds of polyethylene ii. HDPE = gallon milk cartons (more rigid) iii. LDPE = plastic bags, squeeze bottles (more flexible) b. Reaction: nCH2=CH2 [-CH2-CH2-]n c. Monomer: ethylene, CH2=CH2 d. Conditions: iv. Temp: 200 C v. Pressure: 2000 atm vi. Initiator: O2 Initiation Ra + 2 CH2=CH2 2 Ra-CH2-CH2 Propagation Ra-CH2-CH2 + H2C=CH2 Ra-CH2-CH2-CH2-CH2 This step keeps repeating, making longer chains: Ra(CH 2-CH2)n-CH2-CH2 Termination 2 Ra(CH2-CH2)n-CH2-CH2 + CH2-CH2 -n(CH2-CH2)Ra Ra(CH2-CH2)n-CH2-CH2-Ra H H H H H H n n H H H H H H 3. Polyvinylchloride a. Vinyl is a common name for ethylene b. Reactions: nCH2=CHCl [-CH2-CHCl-]n c. Monomer: vinyl chloride or chloroethylene, CH2=CHCl Cl H Cl H Cl H n n H H H H H H Example 2: Condensation of carboxylic acid and amine 1. The basic condensation reaction occurs as below: OH + N + HN O O H2O O CH3 C O CH3 O H + N CH3 H CH3 O- C + H + N H CH3 CH3 H2O CH3 O CH3 C N CH3 1. Polymerization occurs when a molecule has two functional groups a. Polymers formed are copolymers because they are made of two different monomers. b. Polymers are formed in a step-growth method rather than a chain-growth method. i. In other words, molecules with 2 functional groups can grow from both ends instead of just one end as in polyethylene. c. Formation of nylon – i. Hexanedioic acid (2 carboxylic acid groups) and 1,6 diaminohexane (2 amine groups) O HO OH + NH2 H2N O O NH + NH 2n H2O O n d. Formation of polypeptide i. Polypeptide is a chain of amino acids, each amino acid has one carboxylic acid and one amine group ii. Note that the polymerization here occurs because there are two different groups on the same molecule iii. Polypeptides are not, technically, polymers since they don’t have repeating units (R group is different) iv. Peptide bond between alanine and cysteine: O OH + HO O O HS NH OH NH2 O NH2 NH2 + H2O + H2O SH polypeptide: O R OH + HO O O HO NH2 R R O NH R NH2 NH2 adding another amino acid to COOH group O R OH O HN O R NH R NH2 Example 3: Condensation of carboxylic acid and alcohol 1. Polyester is a step-growth polymer like nylon – can grow from both ends 2. Formation of polyester a. Benzene-1,4-dicarboxylic acid (2 carboxylic acid group) and ethane-1,2-diol (2 amine groups) O O OH + HO OH HO O O O O + O n 2n H2O 3. Basic Reaction mechanism for formation of ester from carboxylic acid and alcohol 1. 2. O + R H O H Cl + OH R 3. OH OH R O + H O H + R H OH O R O + H H O + H H + O H R O OH 5. R HO 4. OH R R + + H