* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Slide 1
Artificial gene synthesis wikipedia , lookup
Peptide synthesis wikipedia , lookup
Protein adsorption wikipedia , lookup
Bottromycin wikipedia , lookup
Cell-penetrating peptide wikipedia , lookup
Deoxyribozyme wikipedia , lookup
Metalloprotein wikipedia , lookup
Proteolysis wikipedia , lookup
Genetic code wikipedia , lookup
Fatty acid synthesis wikipedia , lookup
Amino acid synthesis wikipedia , lookup
Basal metabolic rate wikipedia , lookup
Expanded genetic code wikipedia , lookup
List of types of proteins wikipedia , lookup
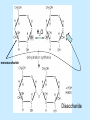
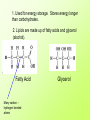
Aim: What are organic compounds? I. Organic compounds – substances that contain both carbon and hydrogen. Each carbon atom can form bonds with up to 4 different atoms. A. Organic compounds help to carry out metabolism 1. Metabolism - The chemical processes occurring within a living cell or organism that are necessary for the maintenance of life. In metabolism some substances are broken down to yield energy for vital processes while other substances, necessary for life, are synthesized. B. Polymers – a large molecule that is made up of many small molecules linked (covalent bonds) together. 1. Dehydration synthesis – when two molecules are bonded together by the removal of a water molecule. A + H2O B A B + H2O 2. Hydrolysis – when a larger molecule is broken down into smaller molecules by the addition of water molecules. H2O + A Demonstration # 1 B A + B Demonstration # 2 II. The four major organic compounds – carbohydrates, lipids, proteins, and nucleic acids. A. Carbohydrates 1. Composed of simple sugars (ex: glucose and fructose – C6H12O6) a. Monosaccharide – one simple sugar b. Disaccharide – 2 simple sugars bonded together 2. Starch – polymer made up of sugar subunits. Starch is also known as a polysaccharide. 3. Carbohydrates include the elements C, H, and O. a. In all carbohydrates, the number of hydrogen atoms is always twice the number of oxygen atoms. 4. Carbohydrates are used for energy storage and provide strong building materials for cells. Demonstration OSE ending!! H2O monosaccharide Disaccharide 1. Used for energy storage. Stores energy longer than carbohydrates. 2. Lipids are made up of fatty acids and glycerol (alcohol). Fatty Acid Many carbon – hydrogen bonded atoms Glycerol 3. One lipid molecule is composed of 3 fatty acid molecules bonded to one glycerol molecule 3 fatty Acids Glycerol 4. Saturated fats – all carbon atoms are single bonded to one another. Saturated fats are solid at room temperature. Ex: Butter, fats in meats, and cheese. 5. Unsaturated fats – there are one or more double bonds between the carbon atoms in the fatty acids. Unsaturated fats are liquids at room temperature. Ex: oils. C. Proteins – used for building materials, transporting substances, sending signals, providing defense, to control chemical activities (p. 106 of text). 1. Proteins are polymers that are made up of amino acids subunits. There are 20 different amino acids. Amino Group Carboxyl Group (NH2) (COOH) Side Chain 2. Amino acids are linked together by peptide bonds. 3. Dipeptide – 2 amino acids linked together by a peptide bond. 4. Polypeptide – many amino acids linked together. What are structures A, B, C, and D? D. Nucleic Acids – consist of DNA and RNA 1. DNA and RNA are responsible for storing genetic information that contains the directions for building every molecule in an organism. 2. DNA and RNA are polymers that are made up of subunits called nucleotides. 3. Nucleotides are made up of a phosphate group, a 5 Carbon sugar (DNA has deoxyribose and RNA has ribose), and a nitrogenous base. 4. Four types of nitrogenous bases: a. Adenine b. Thymine c. Cytosine d. Guanine e. Adenine always pairs up with Thymine (A-T) and Cytosine always pairs up with Guanine (C-G).