* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Dioxygen Activation and Alkane Hydroxylation By The
Survey
Document related concepts
Expanded genetic code wikipedia , lookup
Nucleic acid analogue wikipedia , lookup
Cell-penetrating peptide wikipedia , lookup
Citric acid cycle wikipedia , lookup
List of types of proteins wikipedia , lookup
Proteolysis wikipedia , lookup
Butyric acid wikipedia , lookup
Biosynthesis wikipedia , lookup
Fatty acid metabolism wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
Metalloprotein wikipedia , lookup
Fatty acid synthesis wikipedia , lookup
Biochemistry wikipedia , lookup
Transcript
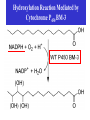
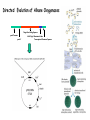
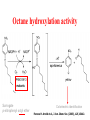
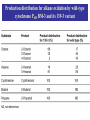
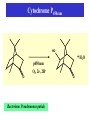
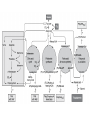
Direct Evolution of P450 Enzyme to Achieve the Controlled Oxidation of Aliphatic Compounds or Methane Steve S.-F. Yu Institute of Chemistry, Academia Sinica 2009/04/15 Methane is a green-house gas. It is an abundant fossil fuel. It is excreted from the earth’s mantle via vents in the ocean floor. There is considerable methane trapped as methane cathrates in the ocean floor at the high pressures that exist there. There is sufficient methane in the earth to serve as a source of fuel for 200 years at the present rate of energy consumption, if we know how to harness the gas. One of the Holy Grails of Organic Chemistry R1R2R3CH + “O” R1R2R3OH Energetics of C-H Activation Calculated bond dissociation energies for the reaction R-H R + H R DR-H/KJ mol-1 (kcal mol-1) CH3 CF3 CHMe2 CMe3 CH=CH2 = 435 (104.0) 444 (106.0) 394 (94.2) 387 (92.5) 453 (108.3) 500 (119.5) 363 (86.7) CCH HC=O C-H activation by Metal Complexes in Organometallic Chemistry Organometallic catalyzed C-H activation usually involves the activation of the C-H bond first, then reacts with dioxygen to form the alcohol. R M + RH [M RH] M H [O] R M + O H C-H activation by Metal Complexes in Biology In the enzymatic pathway, the dioxygen is usually activated at the metal site first, and then followed by oxo-transfer chemistry with the substrate, either by a two step radical process (hydrogen abstraction followed by rebound chemistry), or by concerted "oxene" insertion. R R Concerted Radical M M [O] [O] MO + RH MO + RH ] [MO M + O H . . R M OH H R M + O H The Enzymes that Involve in C-H Activation R H C H C H H R R [O] H C C H H OH R H R C H C R 1.Alkane Monooxygenase:Soluble Methane Monooxygenase, C1-C8 alkanes or aromatic compounds; Particulate Methane Monooxygenase, C1-C5 alkanes, Iron Heme Cyt. P-450, fatty acids or bridge compounds, i.e. camphor; Butane Monooxygenase from Pseudomonas butanovora (ATCC 43655);Alkane monooxygenase from Pseudomonas oleovorans (ATCC 29347). 2. Fatty Acid Desaturases. Soluble Methane Monooxygenase from Methylococcus capsulatus (Bath) cytoplasm periplasm w-Hydroxylase from Pseudomonas oleovorans Cytochrome P-450cam from Pseudomonas putida Cellular metabolism In all forms of life, many important and difficult chemical transformations are catalyzed by enzymes. Many of these enzymes are involved in the metabolism of the cell, particularly, the biosynthesis of key metabolites. Nature has adapted modular design to allow the evolution of these enzymes to accommodate different substrates. Enzymes belonging to a given type of chemical transformation are designed to consist of a “module” that is specific to a given type of chemistry, for example, the transfer of “O” to the C-H bond of an organic substrate, and a different module that could be tuned to accommodate specific substrates and the specific C-H bond to be oxidized. The modular design makes sense if the same chemistry is to be utilized for different substrates. The substrate module could be tuned to control the regio-specificity and stereoselectivity of the chemical transformation. In this manner, the wheel is not reinvented every time a new substrate comes on to the scene. Instead, Nature responds to a specific need by taking advantage of what is already there and adapting the design of the second module to the accommodate the new substrate. For many years, the paradigm of the enzymes that mediate controlled biological oxidations was Cytochrome P450. • A family of closely similar enzymes. • Several hundred members of this protein family are known, each with a different substrate specificity. Cytochrome P450 • In the adrenal cortex, a specific cytochrome P450 participates in the hydroxylation of steroids to yield the adrenocortical hormones. • Mitochondrial cytochrome P450 converts cholesterol to the sex hormones progesterone, testosterone and estradiol in the human reproductive glands (gonads and placenta). • Eukaryotic microsomal cytochrome P450 is also important in the hydroxylation of many different drugs, such as barbiturates and other xenobiotics (substances that are foreign to the body, e.g., the carcinogen benzo[a]pyrene (found in cigarette smoke)). Typically, the substrates are hydrophobic, so many of these enzymes are membrane proteins. There are a few exceptions, for example cytochrome P450cam from the bacterium Pseudomonas putida, and the fatty acid monooxygenase from Bacillus megaterium. Hydroxylation Reaction Mediated by Cytochrome P450 BM-3 Directed Evolution of Alkane Oxygenases Target Gene Coding Sequence pmo1 Promoter pmo1 C 6xHis Tag & Termination Codon Transcriptional Terminator Sequence Octane hydroxylation activity mutants Surrogate p-nitrophenyl octyl ether Colorimetric identification Frances H. Arnold et al., J. Am. Chem. Soc. (2003), 125, 13442. Experimental Procedures Cytochrome P450 BM-3 CH3CH2CH2 CH3 + “O” CH3CH2CH(OH) CH3 Fatty acid monooxygenase from Bacillus megaterium Glieder, A.; Farinas, E. T.; Arnold, F. H. Nat. Biotech. 2002, 20, 1135. Glieder, A.; Farinas, E. T.; Arnold, F. H. Nat. Biotech. 2002, 20, 1135. Fasan, R.; Meharenna, Y. T.; Snow, C. D.; Poulos,T. L. and Arnold, F. H. J. Mol. Biol. 2008, 383, 1069 Comparison of the specific activities of various monooxygenases toward their substrates Comparison of the specific activities of the wild type cytochrome P450 BM-3 and its 139-3 variant for various alkane substrates Production distribution for alkane oxidation by wild-type cytochrome P450 BM-3 and its 139-3 variant Cytochrome P450cam HO + H2O O p450cam O2, 2e-, 2H+ Bacterium: Pseudomonas putida O Cytochrome P450cam Schlichting I, Jung C, Schulze H. FEBS Lett. 1997, 415, 253. Production of alkane oxidation by wild-type cytochrome P450cam and its mutants F87W Y96F V247L S. G. Bell, J.-A. Stevenson, H. D. Boyd, S. Campbell, A. D. Riddle, E. L. Orton, L.-L. Wong, Chem. Commun. 2002, 490. F. Xu, S. G. Bell, J. Lednik, A. Insley, Z. Rao, L. L. Wong, Angew. Chem. Int. Edit. 2005, 44, 4029. The above analysis should pertain to the hydroxylation of any hydrocarbon that show a reasonable binding affinity for the active site of cytochrome P450. What about small alkanes, e.g., methane, ethane, propane, and so on, which have larger C-H bond energies (and large barriers for C-H activation), but which are expected to bind to the hydrophobic pocket of cytochrome P450 with only a very low sticking coefficient. Production of synthetically useful alcohols by enzymatic hydroxylation. Sites of enzymatic hydroxylation of (a) Trimegestrone ® and (b) codeine are indicated by open arrows. (c) The conversion of a-pinene to the mint flavour ingredient verbenol. (d) The production of (S)-Nbenzyl-3-hydroxypyrrolidine by the hydroxylation of Nbenzylpyrrolidine. (e) The prodution of diastereomerically define vicinal diol derivatives of fatty acids by the hydroxylation of hydroxymyristic acids using cytochrome (cyt P450-BM3). Taxol Biosynthetic Pathway Addition of Radicals to Alkenes: Polymers • A polymer is a very large molecule consisting of repeating units of simpler molecules, formed by polymerization • Alkenes react with radical catalysts to undergo radical polymerization • Ethylene is polymerized to poyethylene, for example Free Radical Polymerization of Alkenes • Alkenes combine many times to give polymer – Reactivity induced by formation of free radicals Free Radical Polymerization: Initiation • Initiation - a few radicals are generated by the reaction of a molecule that readily forms radicals from a nonradical molecule • A bond is broken homolytically Polymerization: Propagation • Radical from intiation adds to alkene to generate alkene derived radical • This radical adds to another alkene, and so on many times Polymerization: Termination • Chain propagation ends when two radical chains combine • Not controlled specifically but affected by reactivity and concentration Other Polymers • Other alkenes give other common polymers Polymer Synthesized by Microorganism • • • • PHA Poly(hydroxyalkanoic acid) ACP Acyl-carrier protein PhaA b-Ketothiolase PhaB NAD(P)H-dependent acetoacetyl-CoAreductase • PhaC PHA synthase References: Polyhydroxylbutyrate, Wikipedia Alexander Steinbüchel and Bernd Füchtenbusch, TIBTECH OCTOBER 1998 (VOL 16), 419. Glossary 3HA 3-Hydroxyalkanoic acid 3HB 3-Hydroxybutyric acid 3HD 3-Hydroxydecanoic acid 3HDD 3-Hydroxydodecanoic acid 3HHx 3-Hydroxyhexanoic acid 3HO 3-Hydroxyoctanoic acid 3HTD 3-Hydroxytetradecanoic acid 3HV 3-Hydroxyvaleric acid 4HB 4-Hydroxybutyric acid 4HV 4-Hydroxyvaleric acid Polyhydroxybutyrate (PHB) • Polyhydroxybutyrate (PHB) is a polyhydroxyalkanoate (PHA), a polymer belonging to the polyesters class that was first isolated and characterized in 1925 by French microbiologist Maurice Lemoigne. PHB is produced by micro-organisms (like Alcaligenes eutrophus or Bacillus megaterium) apparently in response to conditions of physiological stress. • Microbial biosynthesis of PHB starts with the condensation of two molecules of acetyl-CoA to give acetoacetyl-CoA which is subsequently reduced to hydroxybutyryl-CoA. • This latter compound is then used as a monomer to polymerize PHB. • Water insoluble and relatively resistant to hydrolytic degradation. This differentiates PHB from most other currently available biodegradable plastics, which are either water soluble or moisture sensitive. • Good oxygen permeability. • Good ultra-violet resistance but poor resistance to acids and bases. • Soluble in chloroform and other chlorinated hydrocarbons. • Biocompatible and hence is suitable for medical applications. •Melting point 175°C., and glass transition temperature 15°C. •Tensile strength 40 MPa, close to that of polypropylene. •Sinks in water (while polypropylene floats), facilitating its anaerobic biodegradation in sediments. •Nontoxic. • PHB is everywhere. Trace amounts in short chains of only about 150 units have been found in the cells of yeast, carrots, spinach, sheep, pigs, cattle and even in humans. It exists in the cells of a staggering variety of different organisms. In fact, it seems that you can find PHB in any cell that you care to choose, if you look hard enough. And nobody knows what it's there for. Surely, for something to be so ubiquitous, it must have some function. It's inconceivable that it's just an accident that PHB is present in so many places. Some scientists have even claimed that PHB could be as important as proteins and that HB units (hydroxy butyrate) might have been present in the primordial soup on earth, before amino acids and proteins. These claims may be extravagant, but whatever the real story is, watch this space; PHB must do something significant in cells. White PHB blobs inside a cressleaf The PHB formed 14% of the dry weight of the leaves. The future for PHA • Owing to a number of novel features of poly(3HB) and poly(3HB-co-3HV), these PHAs were initially used mainly in the manufacture of bottles, films and fibres for biodegradable packaging materials and as mulch films for agriculture. • A latex of PHAs may be applied to paper or cardboard to form a waterresistant layer and to produce a completely biodegradable compound material that requires relatively low amounts of the currently expensive PHAs; • PHAs can also be applied as a matrix in retardant materials for the slow release of drugs, hormones, herbicides, insecticides, flavours and fragrances in medicine, pharmacy, agriculture and the food industry. • In addition to the production of PHA by microbial fermentation for special biotechnological applications, the production of some PHAs as commodity chemicals in transgenic plants will most probably be feasible in the future.