* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Drug Handling in kidney and liver disease 2005
Orphan drug wikipedia , lookup
Drug discovery wikipedia , lookup
Theralizumab wikipedia , lookup
Wilson's disease wikipedia , lookup
Drug design wikipedia , lookup
Prescription drug prices in the United States wikipedia , lookup
Pharmacognosy wikipedia , lookup
Pharmaceutical industry wikipedia , lookup
Psychopharmacology wikipedia , lookup
Pharmacogenomics wikipedia , lookup
Prescription costs wikipedia , lookup
Pharmacokinetics wikipedia , lookup
Neuropsychopharmacology wikipedia , lookup
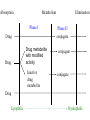
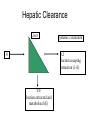
Drug Handling in kidney and liver disease Dr. Geoff Isbister Drug Action • Drugs tend to be small lipid-soluble molecules • Drugs must get access to sites of action • Drugs tend to bind to tissues, usually protein molecules • Drugs alter the actions of enzymes, ion channels and receptors Drug Action • ENZYME: example Angiotensin Converting enzyme inhibitors A I ----X---------->A II lowered A II -----> Reduced BP • ION CHANNELS: example Local Anesthetics Block Na channels--->Anesthesia • Receptor Binding – Receptors are specialised binding sites - often on cell surface- which have specificity for certain substances (incl drugs). Drugs may activate or block the receptor – Activation of the receptor changes the activity of the cell: eg adrenaline activates the beta 1 receptors in the heart and speeds up the heart – Drugs have selectivity for receptors: eg Histamine2 antagonists- reduce histamineinduced acid secretion and heal peptic ulcers Pharmacokinetics • The study of the action of the body on the drugs • Pharmacokinetics is the study of the time course of concentrations of drug in the body • The way the body handles drugs determines the dose, route and frequency of administration • The handling of drugs by the body can be split into absorption, distribution and elimination Pharmacokinetics • Rate of absorption determines the time to the peak concentration • The extent of absorption determines the height of the peak concentration and the AUC 30 25 20 15 10 5 0 0 1 5 9 13 Time after dosing Pharmacodynamics • The response of the tissue to the active free concentration of drug present at the site of action • May also be changed by disease processes Type of Disease • Renal disease – the nature of the disease doesn’t matter very much, the main determinant is the decline in GFR Routes of elimination - Kidney • Some drugs are water-soluble and are eliminated directly by the kidney – Molecules with MW below 20000 diffuse into glom filtrate. – examples: gentamicin, digoxin, atenolol – involves no chemical change to the drug – in most cases occurs by filtration (and depends on the GFR) – in a few cases (eg penicillin) some tubular secretion contributes to elimination • Highly lipid-soluble drugs are filtered into the tubules and then rapidly re-absorbed – High protein binding will reduce filtration Practical issues - treating real patients • Assessing kidney function is straightforward – serum creatinine reflects GFR – relationship between serum creatinine and GFR changes with age Effects of age on renal function • There is a steady and proportional decline in average GFR with increasing age • However the serum creatinine remains unchanged • Why is this? Effects of age on renal function (constant serum creatinine of 0.10 mmol/l) 100 90 80 70 60 50 40 30 20 10 0 20 40 60 80 Multiple Dosing - renally excreted drug Approx 5 half-lives to reach steady state 1 0.9 0.8 0.7 0.6 0.5 0.4 0.3 0.2 0.1 0 Elderly 0 12 24 36 48 60 72 84 96 Drug Types • Water soluble - excreted unchanged (by the kidney) • Lipid soluble - filtered but fully reabsorbed in the kidney - metabolised to polar products (filtered without reabsorption) A number of drugs are handled by tubular mechanisms • Two mechanisms – Active tubular secretion – important • Acidic drugs – frusemide, methotrexate, penicillins, salicylate, uric acid, probenecid • Bases – amiloride, morphine, quinine • Passive diffusion – After filtration lipid-soluble drugs will be re-absorbed passively. – Will depend on degree of ionization at certain pH levels Practical Examples of dosing in renal failure Gentamicin • Practice is changing - trend to once/daily dosing • The interval between doses may be >24 hours in the presence of renal failure and in the elderly • Toxicity relates to trough concentrations, particularly with prolonged therapy • Toxicity mainly affects the kidney and 8th cranial nerve Digoxin • In the presence of renal impairment the dose must be reduced • The dose is given once daily • Elderly people almost invariably have some renal impairment, so they usually require dose reduction - normally a halving of dose compared with young people Summary • Reduced elimination of drugs from the body in the elderly will lead to accumulation and toxicity • Disease and old age lead to reduced renal elimination of water-soluble drugs • Co-morbidity and concomitant drug therapy Hepatic Disease • Metabolism by the Liver : – role of metabolism – types of metabolism • Clearance – hepatic clearance • Liver disease Type of Disease • In liver disease the type of disease does matter: – Hepatitis – not much effect – Biliary obstruction – not much effect (initially) – Cirrhosis – has major effects on drug handling Assessing Function • Assessing liver function is hard - no single test of how well the liver metabolises drugs – Drug metabolism most likely to be impaired when the patient has cirrhosis, and has evidence of coagulation disturbances and low albumin Biotransformation • Majority produces metabolites that are : – less active – more polar and water soluble • Minority : – Pro-drugs that require metabolism to be active – active metabolites – more toxic (mutagenic, teratogenic etc.) Drugs with Active Metabolites DRUG ACTIVE METABOLITE allopurinol amitriptyline codeine diazepam procainamide prednisone primidone aspirin oxypurinol nortriptyline morphine oxazepam N-acetyl PA prednisolone phenobarbitone salicylate Types of Metabolism • Phase 1 Reactions – usually convert the parent drug into a more polar metabolite by introducing or unmasking a functional group (-OH, -NH2, -SH). Metabolite is usually inactive. • Phase 2 Reactions - Conjugation – an endogenous substrate (glucuronic acid, sulfuric acid, acetic acid, or amino acid) is attached to a functional group on the drug or phase I metabolite. Absorption Phase I Phase II conjugate Drug Drug Elimination Metabolism Drug metabolite with modified activity Inactive drug metabolite conjugate conjugate Drug Lipophilic Hydrophilic Phase I Reactions • Mixed Function Oxidase: – P450 enzyme system – induced and inhibited – hydroxylation and demethylation – family of isoenzymes • Monoamine Oxidase : catecholamines • Dehydrogenases :eg. Alcohol dehydrogenase Phase I - P450 System • • • • • FRAGILE High specificity Low volume Energy dependent First affected by liver disease Cytochrome P450 System • Not a single entity • Family of related isoenzymes (about 30) • Important for drug interactions : – Enzyme induction – Enzyme inhibition • Genetic polymorphism Phase II Reactions Conjugation • • • • • Glucuronidation Sulfation Acetylation Glutathione Glycine Phase II Reactions Conjugation • • • • • ROBUST High volume Low specificity Not energy dependent Less effected by liver disease Paracetamol toxicity – failure of Phase II Conjugation pathway saturates oxidation by P450 cytochrome pathway Formation of toxic metabolite NAPQI Initially detoxified by glutathione Glutathione depletion NAPQI accumulates and binds to tissue macromolecules - cell death Sites of Biotransformation • Liver • • • • Lung Kidney Large and small intestine Placenta Hepatic Clearance Liver Systemic circulation 0.2 fraction escaping extraction (1-E) 1.0 0.8 fraction extracted and metabolised (E) Extraction Ratio • High extraction ratio : – Effectively removed by the liver – Limited by hepatic blood flow – High first pass metabolism – Eg. Lignocaine, propranolol, diltiazem, morphine – Less effected by changes in intrinsic clearance, such as induction and inhibition Extraction Ratio • High Extraction ratio – Clearance approximates organ blood flow • Low Extraction ratio – Clearance proportional to free drug in the blood and intrinsic clearance of the liver Liver Disease • Severe disease before major effects on metabolism • Liver Disease : – Hepatocellular disease – Decrease liver perfusion • Type of metabolism : – Phase I – Phase II Disease Factors • Disease Type : – Acute hepatitis – little effect – Biliary Obstruction – little effect – Chronic Active Hepatitis – major effects – Cirrhosis – major effects • Indicators : – Established cirrhosis, varices, splenomegaly, jaundice, increased prothrombin time. Disease Factors • Poor perfursion • Cardiac failure : limits blood flow so effects those with high extraction ratios – Eg. Lignocaine – Combination with ischaemic liver injury • Other low perfusion states : – Other causes of shock Recent theories to account for impaired metabolism in cirrhosis • • • • Intact hepatocyte mass Sick cell theory Impaired drug uptake/shunting theory Oxygen limitation theory Type of Metabolism • Phase I, mainly P450 – Affected first • Phase II – Severe disease before any effect – Eg. Paracetamol poisoning. Other considerations • Renal function may be impaired in moderate to severe liver disease – Creatinine levels are not predictive • Pro-drug metabolism impairment – Eg ACE inhibitors • Pharmaco-dynamic disturbances – Tissues may be excessively sensitive to even low concentrations of the drug – eg morphone in the brain in the presence of severe liver disease