* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Dabigatran - Cardiology Update FK UNAND
Survey
Document related concepts
Transcript
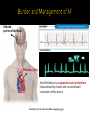
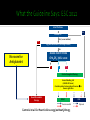
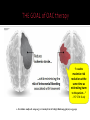
PUSAT JANTUNG Regional Dr. MUHAMMAD SYUKRI, Sp JP BAGIAN KARDIOLOGI DAN KEDOKTERAN VASKULAR FKUA/PUSAT JANTUNG RS. DR. M DJAMIL, PADANG Burden and Management of AF Challenges and limitation of ASA and VKA New Oral Anti Coagulants ( NOACs) Results of the studies with NOACs, RELY and RELY-ABLE Results of RELY among Asian population What the Guideline Says The goal of OAC therapy Summary Sinus Rhytm Affected portion of the brain Atrial Fibriillation Thrombus (clot) Atrial fibrillation is a supraventricular arrhythmia characterized by chaotic and uncoordinated contraction of the atrium Chowdhury P, et al. Cleve Clin J Med. 2009;76:543–550 David Bloom's silent killer. David Bloom was an American television journalist covering Iraq war who died suddenly in 2003 after a pulmonary embolism. The Stroke Association: www.stroke.org.uk. Base on: Office of National Statistics Health Statistics Quarterly, Winter 2001 "Stroke incidence and risk factors in a population based cohort study“. The Stroke Association estimate that 5,000 people per year have a stroke in Northern Ireland Scottish Stroke Care Audit 2005/2006. Prevention of complications, including thromboembolism (particularly ischaemic stroke) and heart failure Relief of symptoms Choice of antithrombotic therapy should be tailored to the patient based on: Risk of thromboembolism Risk of bleeding ESC guidelines: Camm J et al. Eur Heart J 2010;31:2369–429; ACCF/AHA/HRS Focused Update Guidelines: Fuster V et al. J Am Coll Cardiol 2011;57:e101–9 Superior Efficacy Profile of OAC vs ASA to Prevent Stroke in Patients With Non Valvuler AF Hart et al, Ann Intern Med 2007;146:857–867 Similar safety profile of OAC and ASA in intracranial bleeding and major bleeding Friberg, Rosenqvist & Lip Eur Heart J 2012 Camm AJ et al. Eur Heart J 2012;33:2719–47; Aspirin Tablets BP 300 mg: SmPC, 2013; Ansell J et al. Chest 2008;133;160S–198S; Nutescu EA et al. Cardiol Clin 2008;26:169–87; Umer Ushman MH et al. J Interv Card Electrophysiol 2008;22:129–37 Requirements ? At least as effective as warfarin ? Predictable response ? Wide therapeutic window ? Low incidence and severity of adverse effects ? Oral fixed dose ? No need for routine anticoagulation monitoring ? Low potential for food or drug interactions ? Fast onset and offset of action ? Guidelines ? Long term safety profile ? Lip GY et al. EHJ Suppl. 2005;7:E21–25 13 NOACs approved for prevention of systemic embolism or stroke in patients with non-valvular AF RRR 24% Dabigatran ® 150 mg twice daily is proven to provide superior Ischaemic Stroke prevention vs. Warfarin1 24% risk reduction in Ischaemic Stroke Haemorrhagic Stroke RRR 74% RRR 69% Both dosages of Dabigatran® dramatically reduced the risk of haemorrhagic stroke compared with warfarin:1 17 Life-Threatening Bleeding Rates RRR 20% RRR 33% Both doses of Dabigatran ® Significantly reduced the risk of life threatening bleeding compared with warfarin1 Intracranial Bleeding RRR 59% RRR 70% Both doses of Dabigatran ® substantially reduced the risk of intracranial bleeding compared with warfarin1 18 25 27 RE-LY® c Connolly S et al NEJM 2009; Patel M et al NEJM 2011; Granger C et al NEJM 2011 Not head-to-head comparison – for illustrative purposes only ROCKET-AFARISTOTLE Rate (%/year) Dabigatran Warfarin 150mg bid 110mg bid Stroke or SEE Asia Non-Asia Ischemic stroke Asia Non-Asia Hemorrhagic stroke Asia Non-Asia Myocardial infarction Asia Non-Asia Death from any cause Asia Non-Asia Dabigatran 150mg bid vs. Warfarin Dabigatran 110mg bid vs. Warfarin HR (95%CI) HR (95%CI) Interaction p value Interaction p value 1.39 1.06 2.50 1.37 3.06 1.48 0.0853 0.5597 1.12 0.81 2.05 1.14 2.02 0.98 0.1977 0.5959 0.17 0.09 0.11 0.12 0.75 0.32 0.7590 0.2729 0.50 0.86 0.51 0.88 0.58 0.65 0.3782 0.3761 4.01 3.57 5.01 3.53 5.09 3.96 0.4244 0.5929 0 1.0 2.0 Dabigatran better Warfarin better RE-LY® Asia 0 1.0 2.0 Dabigatran better Warfarin better Rate (%/year) Dabigatran Warfarin 150mg bid 110mg bid Major bleeding Asia Non-Asia GI major bleeding Asia Non-Asia Life threatening bleeding Asia Non-Asia Intracranial bleeding Asia Non-Asia Minor bleeding Asia Non-Asia Major or minor bleeding Asia Non-Asia Dabigatran 150mg bid vs. Warfarin Dabigatran 110mg bid vs. Warfarin HR (95%CI) HR (95%CI) Interaction p value Interaction p value 2.17 3.52 2.22 2.99 3.82 3.53 0.0079 0.0705 0.96 1.69 1.15 1.14 1.41 1.01 0.0089 0.3379 1.28 1.52 0.91 1.29 2.20 1.79 0.1749 0.0738 0.45 0.29 0.23 0.23 1.10 0.71 0.9509 0.4561 12.43 15.27 10.12 13.69 19.66 15.81 <0.0001 <0.0001 13.99 17.02 11.72 15.27 22.03 17.74 <0.0001 <0.0001 0 1.0 2.0 0 1.0 Dabigatran better Warfarin better Dabigatran better RE-LY® Asia 2.0 Warfarin better The RELY-ABLE® study: Long-term multicentre extension of dabigatran treatment in patients with atrial fibrillation Study design 20 OBJECTIVE: Evaluate long-term safety of dabigatran etexilate (two doses) in patients with AF RE-LY® AF and ≥1 additional risk factor for stroke Absence of contraindications R Warfarin (INR 2.0–3.0) n=6022 Dabigatran etexilate 110 mg BID n=6015 Dabigatran etexilate 150 mg BID n=6076 Dabigatran etexilate 110 mg BID N=2914 Dabigatran etexilate 150 mg BID N=2937 RELY-ABLE® BID = twice daily Goals To describe the long-term efficacy and safety of ongoing dabigatran therapy following RE-LY® Methods Patients eligible at completion of RE-LY® study if: ▪ Alive and still receiving study dabigatran ▪ Being followed at centre participating in RELY-ABLE® Dabigatran blinded dose continued in RELY-ABLE® for 2.3 years Analysis Two follow-up periods described ▪ RELY-ABLE® (post-RE-LY®) ▪ RE-LY® + RELY-ABLE® (beginning of RE-LY® to end of RELY-ABLE®) In patients who continued treatment on dabigatran after RELY®, the rates of stroke and major bleeding remain low There were no new safety signal observed during this extended follow up period The results from RELY-ABLE® are highly consistent with those observed in RE-LY® Atrial fibrillation Yes Valvular AF* No (i.e. nonvalvular) Yes <65 years and lone AF (including females) No Assess risk of stroke CHA2DS2-VASc score No room for Antiplatelet 0 1 ≥2 Oral anticoagulant therapy Assess bleeding risk (HAS-BLED score) Consider patient values and preferences to choose right dose No antithrombotic therapy NOAC Recommended Camm AJ et al. Eur Heart J doi:10.1093/eurheartj/ehs253 VKA Optional Camm AJ et al. Eur Heart J doi:10.1093/eurheartj/ehs253 Update strongly recommends a practice shift towards identification of ‘truly low risk’ patients with AF (i.e. age <65 years and lone AF) who do not need antithrombotic therapy CHADS2 does not reliably identify ‘truly low risk’ patients CHA2DS2-VASc: inclusive of the most common stroke risk factors validated in multiple cohorts better than CHADS2 at identifying ‘truly low risk’ patients As good as CHADS2 in identifying patients who develop stroke and thromboembolism HAS-BLED score: • allows clinicians to make informed assessment of bleeding risk • makes clinicians think of the correctable risk factors for bleeding • has been validated in several independent cohorts • correlates well with ICH risk High HAS-BLED score per se should not be used to exclude patients from OAC therapy Camm AJ et al. Eur Heart J doi:10.1093/eurheartj/ehs253 2012 28 Recommendation Class Level In patients with CHA2DS2-VASc score ≥2, OAC therapy with: •a dose-adjusted VKA (INR 2–3); or •a direct thrombin inhibitor (dabigatran); or •an oral Factor Xa inhibitor (e.g. rivaroxaban, apixaban*) … is recommended unless contraindicated I A In patients with CHA2DS2-VASc score 1, OAC therapy with: •a dose-adjusted VKA (INR 2–3); or •a direct thrombin inhibitor (dabigatran); or •an oral Factor Xa inhibitor (e.g. rivaroxaban, apixaban*) … should be considered, based upon an assessment of the risk of bleeding complications and patient preferences IIa A 29 *Pending approval; INR = international normalized ratio; OAC = oral anticoagulation; VKA = vitamin K antagonist Camm AJ et al. Eur Heart J doi:10.1093/eurheartj/ehs253 “I need to maximize risk reduction at the same time as minimizing harm to the patient… “ - PCP CPA Study 1. Circulation. 2008; 118 : 2029-2037. 2. Connoly SJ et al. N Engl J Med 2009; 361(12): 1139-1151 Requirements At least as effective as warfarin Dabigatran SUPERIOR Predictable response YES Wide therapeutic window YES Low incidence and severity of adverse effects YES Oral fixed dose YES No need for routine anticoagulation monitoring YES Low potential for food or drug interactions YES Fast onset and offset of action YES Guidelines Long term safety profile ACCP, ESC, AHA/ASA, NICE, CCS, PERDOSSI RELY-ABLE, PMS (FDA and EMA) 34 Lip GY et al. EHJ Suppl. 2005;7:E21–25 AF confers an increased risk of stroke, which is dependant upon the presence of various stroke risk factors All NVAF patient with ≥ 1 risk of stroke should receive anticoagulation - ASA is not an alternative, availability of NOACs has led to revisions in treatment guidelines The net clinical benefit balancing ischaemic stroke vs intracranial bleeding favors Dabigatran from RE-LY® Net clinical benefit was consistently in favor of DE for both doses compared with warfarin, in both Asians and nonAsians Give right dose for the right patient (150mg or 110mg): Age, HASBLED, renal function and drug interactions Dabigatran provides long-term safety data in this setting (RELY-ABLE, PMS EMA and FDA)