* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download An Update on Reversal Agents for NOACs: Where Are
Survey
Document related concepts
Transcript
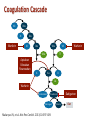
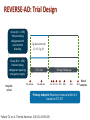
An Update on Reversal Agents for NOACs: Where Are We Now? Moderator Christian T. Ruff, MD, MPH Assistant Professor of Medicine TIMI Study Group Brigham and Women's Hospital Harvard Medical School Boston, Massachusetts Panelists Joshua N. Goldstein, MD, PhD Arash Afshinnik, MD Associate Professor of Surgery Harvard Medical School Director Center for Neurologic Emergencies Massachusetts General Hospital Boston, Massachusetts Director of Neurocritical Care Assistant Clinical Professor Department of Neurologic Surgery Division of Neurology University of California, San Francisco Fresno, California Carmelo Graffagnino, MD Medical Director Duke Comprehensive Stroke Center Professor and Chief Division of Vascular Neurology, Stroke and Neurocritical Care Department of Neurology Duke University Medical Center Durham, North Carolina This program will include discussion of investigational drugs not approved by the FDA for use in the United States. Overview • NOACs are at least as effective as warfarin in the prevention of stroke in patients with atrial fibrillation and in VTE treatment and prevention • Easier to administer • Safer with respect to bleeding, particularly serious bleeding – Overall, an approximate 50% reduction in fatal and life-threatening bleeding (particularly ICH) events across trials • NOACs were developed without a reversal agent • Many eligible patients are still not receiving NOACs partially because of perceived fear of bleeding, lack of antidote Ruff CT, et al. Lancet. 2014;383:955-962. Better to Prevent Than Treat • Most bleeding events can be effectively managed with supportive care and are not associated with any bad long-term outcomes • Serious bleeding is a lot less common with NOACs • Avoid concomitant use of antiplatelet agents, NSAIDs • Monitor renal function and dose appropriately X • Due to short half-lives of NOACs, temporarily stopping them may be all that is required Rivaroxaban[a] Apixaban[b] Dabigatran[c] Edoxaban[d] Half-life, h 5-9 ~12 12-17 10-14 Renal clearance, % 33* 27 80 50 Characteristic *33% renally cleared; 33% excreted unchanged in urine. a. Xarelto® PI 2016; b. Eliquis® PI 2015; c. Pradaxa® PI 2015; d. Savaysa™ PI 2015. Coagulation Cascade XII XIIa XI Warfarin XIa IX IXa VIIa VIIIa Apixaban Edoxaban Rivaroxaban X Warfarin VII TF Xa X Va Warfarin Prothrombin II Thrombin IIa Fibrinogen I Makaryus JN, et al. Nat Rev Cardiol. 2013;10:397-409. Fibrin Ia Dabigatran Clot Is Testing Helpful? • Dabigatran – APTT, dilute TT, ECA, ECT can determine if drug levels are present – Normal TT likely excludes relevant dabigatran level • Factor Xa inhibitors – Anti-Xa can detect drug levels – PT may detect if drug is present Cuker A, et al. J Am Coll Cardiol. 2014;64:1128-1139. Reversal Agents Idarucizumab Humanized Fab fragment Andexanet alfa Recombinant factor Xa variant Ciraparantag Synthetic small molecule Dabigatran Factor Xa inhibitors UFH, LMWH, NOACs (not warfarin) Binds dabigatran with high affinity Competes with factor Xa for inhibitor binding Noncovalent hydrogen bond (exact mechanism unclear) Reconstitution None Add 10 mL sterile water None Administration 2 × 2.5 g/50 mL IV bolus, may require repeat dose 400 mg IV bolus plus 2-hour infusion* Single 100 mg IV dose (dose under investigation) FDA-Approved 10/16/2015 Not yet approved Not yet approved Structure Target Mechanism Status *Dose being studied in ANNEXA-4; dosing will depend on which FXa inhibitor was taken, and when it was taken. Hu TY, et al. Vasc Health Risk Manag. 2016;12:35-44. REVERSE-AD: Trial Design Group A (n = 298) Patients taking dabigatran with uncontrolled bleeding 5 g idarucizumab (2 ×2.5 g IV) Group B (n = 196) Patients taking dabigatran requiring emergency surgery Hospital arrival 0-15 min Pre-1st vial Pre-2nd vial 90 days follow-up 1 h 2 h 4 h 12 h 24 h 30 d 90 d Primary endpoint: Maximum reversal within 4 h based on dTT, ECT Pollack CV, et al. Thromb Haemost. 2015;114:198-205. Blood samples REVERSE-AD: Mortality • Patients had life-threatening conditions • Deaths: – Group A (bleeding, n = 298): 12.3% (30 days); 18.7% (90 days) – Group B (surgery, n = 196): 12.4% (30 days); 18.5% (90 days) Idarucizumab • Given two 2.5 g loading doses, 15 minutes apart = 5 g total • Rapidly eliminates dabigatran from circulation – Allows the body to establish hemostasis Pollack CV. ESC 2016. ANNEXA-4: Andexanet Alfa • Recombinant modified human factor Xa decoy protein • Multicenter, prospective, open-label, single-group study • N = 67 (interim analysis) • Patients with acute major bleeding ≤18 hours after the administration of a fXa inhibitor – Bolus of andexanet followed by a 2-hour infusion • Patients were evaluated for changes in measures of anti-fXa activity and clinical hemostatic efficacy during a 12-hour period • All the patients were followed for 30 days Connolly SJ, et al. N Engl J Med. 2016;375:1131-1141. ANNEXA-4: Mortality Interim Analysis • Safety group, n = 67 – All patients who received andexanet – Death = 15% – Patients with ICH: 6/28 died = 21% • Efficacy group, n = 47 – Patients with baseline anti-fXa activity ≥ 75 ng/mL (or ≥ 0.5 IU/mL for enoxaparin) – Death = 15% Connolly SJ, et al. N Engl J Med. 2016;375:1131-1141. The Bleeding Has Stopped -What's Next? • Reversal agents rapidly and safely remove the anticoagulant from the body • When to restart anticoagulation is patient specific, but should be as early as possible due to high risk of thrombosis • Reversal agents are not associated with immune reactions • Reversal agents remove the anticoagulant rapidly without serious adverse events Ciraparantag (PER977) • Universal reversal agent • Synthetic molecule binds: – Direct Xa inhibitors (apixaban, rivaroxaban, and edoxaban) – Direct thrombin inhibitors (dabigatran) – UFH, LMWH Hu TY, et al. Vasc Health Risk Manag. 2016;12:35-44. Supply and Distribution • How will reversal agents be incorporated into protocols? • Where will the drugs be kept? – Emergency department? – Pharmacy? – Blood bank? • How are they stored? – Need for refrigeration? • Need for reconstitution? • Who gives approval for use? • Where are they needed? – ICU, ED Summary • Reversal agents will provide reassurance and hopefully improve use of anticoagulation in at-risk patients – NCDR PINNACLE Registry demonstrated that approximately half of eligible patients with AF do not receive anticoagulation[a] • Patients on NOACs are less likely to have serious bleeding events than patients on warfarin • Idarucizumab is available across the United States[b] • Approval of andexanet alfa for fXa inhibitors expected soon • Hospitals should be developing protocols to enable rapid and effective use of reversal agents a. Hsu JC, et al. JAMA Cardiol. 2016;1:55-62. b. Praxbind (idarucizumab) website. Thank you for participating in this activity. To proceed to the online CME test, click on the Earn CME Credit link on this page.