* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download SAS Clinical Programming In 18 Easy Steps
Drug interaction wikipedia , lookup
Polysubstance dependence wikipedia , lookup
Drug design wikipedia , lookup
Pharmaceutical marketing wikipedia , lookup
Pharmacognosy wikipedia , lookup
Neuropsychopharmacology wikipedia , lookup
Pharmacokinetics wikipedia , lookup
Prescription drug prices in the United States wikipedia , lookup
Pharmacogenomics wikipedia , lookup
Drug discovery wikipedia , lookup
Prescription costs wikipedia , lookup
Pharmaceutical industry wikipedia , lookup
Clinical trial wikipedia , lookup
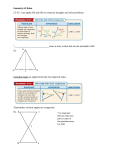
SAS Clinical Programming In 18 Easy Steps FOR M.Pharm / M.Sc (Life Sciences) /M.sc (Stats)/ M.C.A/M.Tech / B.Pharm /Bio-Tech/B.Tech. SAS Clinical Programming In 18 Easy Steps FOR M.Pharm / M.Sc (Life Sciences) /M.sc (Stats)/ M.C.A/M.Tech / B.Pharm /Bio-Tech/B.Tech. Mr. Y. Lakshmi Prasad Notion Press 5 Muthu Kalathy Street, Triplicane, Chennai - 600 005 First Published by Notion Press 2014 Copyright © Mr. Y. Lakshmi Prasad 2014 All Rights Reserved. ISBN: 978-93-84381-63-9 This book has been published in good faith that the work of the author is original. All efforts have been taken to make the material error-free. However, the author and the publisher disclaim the responsibility. No part of this book may be used, reproduced in any manner whatsoever without written permission from the author, except in the case of brief quotations embodied in critical articles and reviews. This book is dedicated to P. V. Narasimha Rao (Patriotic statesman who believed that the nation is bigger than the political system) Preface Finally, An Indian author created an indispensable guide focuses on SAS Clinical Programming, in an instructive and conversational tone which helps them who want to make their career in SAS Clinical Programming and entry level programmers for their day to day tasks with practical, real world examples, detailed description, work flows, Issues, Resolutions, key techniques and many more. This book is like your personal trainer, explains the art of SAS clinical Programming in 18 easy steps which covers from basics to ADS, TLF Creation, as well as CDISC SDTM, ADaM. Many statistical Concepts are explained in an easy way so that you feel confident while using Statistical Procedures. If you are already working as a SAS Clinical Programmer, still you need this book to sharpen your skills. This book will be an asset to you and your career by making you a better SAS clinical Programmer. Authors’ Note One interesting thing in SAS Clinical Programming, it is the career Option for people with various study backgrounds. I have seen SAS Programmers with different qualifications like Pharmacy, Statistics, M.C.A, M. Tech, M.Sc Life sciences, Bio Tech, Mathematics and many more. It is wonderful to see people with different backgrounds working on the same project, but how can we expect Clinical Research and Biostatistics knowledge from a person with technical qualification. Every person might be strong in their own subject but SAS Clinical Programmer needs to know more than one subject (Programming (Tech), Clinical Research (Pharma) and Biostatistics (Stats)). This might be the reason I thought it would be beneficial to have a resource that brings together all these aspects in one volume so that it would help everybody who wants to make SAS Programming as their career Option. This book was written to assist learners in getting started, while at the same time providing techniques that I have found to be useful to Entry level SAS Clinical programmers. This book is aimed more at the SAS Clinical programmer who is responsible for producing submission ready Reports. This book assumes that the reader has no prior knowledge of clinical research and SAS programming. Each one of us has our own style of approach to an issue; it is likely that others will find alternate solutions for many of the issues discussed in this book. The clinical trial data that appears in a number of examples throughout this book was just an imaginary phase 2 Diabetic study, any resemblance was simply accidental. This book was organized in 18 easy steps from introduction to creating final reports, which resembles the SAS Clinical programmer’s work environment. viii Author’s Note The solutions to some of the questions are not written fully but only some steps of hints are mentioned. It is just for the sake of recalling the memory involving important facts in common practice. I shall ever be thankful, grateful and eager to accept the insightful suggestions from my colleagues make this work more worthy. Y L Prasad Acknoweldgements A great deal of information was received from the numerous people who offered their time. I would like to thank each and every person who helped me in creating this book. I heartily express my gratitude to all of my peers, colleagues, friends and students whose sincere response geared up to meet the exigent way of expressing the contents. I am very much grateful to our Press, editors and designers whose scrupulous assistance completed this work to reach your hands. Finally, I am personally indebted to my wonderful partner Prajwala, and my kid Prakhyath, for their support, enthusiasm, and tolerance, without which this book would have never been completed. Y.L.Prasad Contents Prefacevi Authors’ Note vii Acknoweldgements ix Step 1: INTRODUCTION TO CLINCAL RESEARCH 1 Drug Development Process Clinical Research Clinical Trials Clinical Research Process Industry Regulations and Standards for Clinical Trials Departments and Roles in a CRO Process Flow in a Typical Clinical Trial 1 2 3 3 6 8 12 Step 2: INTRODUCTION TO SAS 15 Basic SAS Program Requirements Rules for User-Supplied SAS Names SAS WINDOWS Reading Data into a SAS Data Set Introduction to SAS/Library 16 17 18 19 20 Step 3: INTRODUCTION TO OUR STUDY 23 Diabetes Mellitus 23 Types of Diabetes Mellitus 23 Pathophysiology24 Diagnosis 24 WHO Diabetes Diagnostic Criteria 25 Study Details 25 Study Design 26 Efficacy Measures 27 Analysis Populations 28 Treatment Compliance 29 Contents xi Baseline Definition Efficacy Evaluation Safety Evaluation 29 30 30 Step 4: STUDY DATASETS 32 List Input Method Double Trailing AT Method (@@) Reading Non Standard Data (Format Input Method) Column Input, Column Pointer Method Column Pointer At Method 33 34 36 40 45 Step 5: INTRODUCTION TO PROC STEP 56 Proc Print (PRINT Procedure) Proc Contents (CONTENTS Procedure) Proc Sort (SORT Procedure) Proc Format (FORMAT Procedure) 57 65 67 72 Step 6: COMBINING DATASETS 81 Concatenating Datasets Appending Datasets Interleaving Datasets Merging Datasets 82 83 87 88 Step 7: SAS FUNCTIONS 98 Arithmetic Functions Character Functions DATE/TIME Functions 99 101 122 Step 8: UNDERSTANDING DATA STEP PROCESS AND PDV 128 Flow of Action in a Datastep Processing a DATA Step, a Walkthrough Types of Errors in SAS Common Automatic Variables 128 130 131 132 Step 9: MODIFYING SAS DATASETS 133 Conditional Processing Using UNTIL and WHILE Clauses 133 149 xii Contents Debugging154 Creating User Generated Log Messages 154 RETAIN Statement 155 Array158 Step 10: STATISTICAL CONCEPTS 165 Common Statistical Terms Types of Variables Types of Data Types of Clinical Studies Selection of an Appropriate Statistical Test Proc Freq (FREQ Procedure) CHI-Square Test Fisher’s Exact Test Mantel-Heanszel Chi-Square Test Cochran-Mantel-Heanszel Chi-Square Test (CMH Test) Proc Means (MEANS Procedure) Proc Univariate (UNIVARIATE Procedure) Proc Univariate in Data Cleaning ODS (Output Delivery System) Proc Ttest (TTEST Procedure) Two Sample Ttest Proc Npar1way (NPAR1WAY Procedure) Wilcoxon Signed-rank Test Wilcoxon Rank Sum Test Proc ANOVA Kruskal-Wallis Test CORR Procedure Regression Analysis Multiple Linear Regressions LOGISTIC Regression 165 166 167 172 174 175 179 182 183 184 184 188 191 192 199 201 203 204 205 206 208 209 213 218 219 Step 11: SAS MACROS 222 Macro Terminology Macro Variables %LET Statement Creating Macro Variables from Data Step 222 223 225 228 Contents xiii Symget and Symgetn Functions Building a Better Macro Macro Parameters Steps in Creating Macro Macro Functions Macro Debugging Storing Macros 230 233 235 236 237 242 244 Step 12: PROC SQL 248 SQL Terminology SELECT Clause INTO Clause Joining Tables Performing Inner Join 248 249 256 257 259 Step 13: CDISC SDTM 264 CDISC Standards SDTM Domains DM Domain CM Domain AE Domain LB Domain 264 266 274 281 287 294 Step 14: CDISC ADaM 301 Structure of Analysis Dataset Specifications ADSL Specification ADAE Specifications ADCM Specifications ADLB Specifications Data Definition Tables (Define.pdf) 301 303 315 320 326 332 Step 15: CREATING ANALYSIS DATASETS 337 Study Populations / Analysis Populations Study Day Variable Change from Baseline Visit Windowing Basic Lab Pre-Processing 339 339 343 344 346 xiv Contents Dealing with Partial Dates 348 Time to Event Analysis 349 Duration of Response 350 Determining Value for VISFWDID in Adverse Events 351 Proc Transpose (Transposing Datasets) 351 Flagging Concomitant Medications 354 TEAE Flag 356 LOCF358 Duration Calculation 362 TABLE Shells 365 Step 16: CREATING REPORTS 376 TABULATE Procedure Proc Report (REPORT Procedure) Using the Column Statement Using the Define Statement Using the Break /Rbreak Statements Using Compute Block 376 384 386 388 392 392 Step 17: SAS/GRAPH397 Proc Gchart (GCHART Procedure) GPLOT Procedure 397 400 Step 18: DOCUMENTATION & STANDARD OPERATING PROCEDURES 405 Different Documents we use while Performing SAS Clinical Programming 405 Standard Operating Procedures 412 Validation and QC Guidelines 420 Messages to be Checked in the Log Window 421 Proc Compare 422 Check Your Understanding 426 Index432 We all have dreams. But in order to make dreams come into reality, it takes an awful lot of determination, dedication, self-discipline, and effort. ---Jesse Owens Step 1 INTRODUCTION TO CLINCAL RESEARCH The Process from discovering a new drug to registering it for marketing is very complex and lengthy. There are several people involved in the Process of drug discovery and development includes Scientists, Clinicians, Statisticians as well as Data managers and SAS Programmers. It is estimated that, on average, a drug takes 10-12 years from initial research to reach the market. Drug discovery and development are mainly carried out by pharmaceutical companies and research agencies. The pharmaceutical market is very competitive. It is imperative that pharmaceutical companies discover and develop drugs efficiently and within the shortest time span to remain competitive. Drug Development Process The Drug development Process has different stages: Drug Discovery Drug Development Clinical Trials Manufacturing Marketing Approval The Drug Discovery Process involves in finding out the target that causes the disease, chemical or biological compounds are screened and tested against these targets to find leading drug candidates for further development. The Drug Development Process involves Tests performed on the lead compounds in test tubes (laboratory, in vitro) and on animals (in vivo) to check how they affect the biological systems. 2 SAS Clinical Programming The development Process includes pharmacological studies of the lead compound and its effects on toxicity, carcinogenicity, mutagenicity and reproductive development. These data are important for determining the safety and effectiveness of the lead compound as a potential drug. Typically, tens of thousands of compounds are screened and tested, and only a handful makes it into the market as drug products. The statistics are such that, of 5000 compounds that show initial promise, five will go into human clinical trials, and only one will become an approved drug. Clinical Research Clinical research is a branch of healthcare science that determines the safety and effectiveness of medications, devices, diagnostic products and treatment regimens intended for human use. Clinical research includes: Medical and behavioral research involving volunteer participants Investigations that are carefully developed and conducted with clinical outcomes recorded Identification of better ways to prevent, diagnoses, treats, and understands human disease Trials that test new treatments, clinical management and clinical outcomes, and long–term studies Strict scientific guidelines There are ethical and regulatory constraints for the design and conduct of a clinical trial that have to be considered. Ethical principles are to protect participants before a drug is put forward for a clinical trial. The United States National Institutes of Health (NIH) has stipulated seven ethical requirements: 1. Social value 2. Scientific validity 3. Fair subject selection 4. Informed consent 5. Favorable risk-benefit ratio Y Lakshmi Prasad 3 6. Independent review 7. Respect for human subjects Clinical Trials: Clinical trials are prospective studies on human subjects that are designed to answer specific questions about drugs, treatments, devices or new ways of using known interventions, generating safety and efficacy data. Clinical Research is a study that tests how well an intervention works in a group of people, Tests for new methods of screening, prevention, diagnosis, or therapy. During a trial, additional information is learned about an intervention, its Risks, and its effectiveness. Clinical Research Process Pre-clinical testing Investigational New Drug Application (IND) Phase I (assess safety) Phase II (test for effectiveness) Phase III (large-scale testing) Licensing (approval to use) Approval (available for prescription) Post-marketing studies (special studies and long-term effectiveness/ use) Pre-Clinical Testing Pre-clinical testing is required before testing humans. Pre-clinical testing is often conducted on animals many pre-clinical studies use a review Committee to determine if the use of animals is warranted. The review Committee also checks to see if the research can be improved by reducing or replacing animals. Laboratory and animal studies are conducted to find out if there is a potential benefit of the drug, vaccine, or other product and to explore general safety concerns. If a vaccine, Drug has a potential benefit, it is prepared for human testing. Pre-clinical testing takes approximately three to four years. 4 SAS Clinical Programming Investigational New Drug Application (IND): For studies that involve a new vaccine, drug, after completing Preclinical testing, an investigational new drug application (IND) must be filed describing the results of pre-clinical testing and how future studies will be conducted. The U.S. Food and Drug Administration (FDA) have 30 days to review the IND. If the FDA approves the IND within 30 days, the test drug can precede to a phase I trial. Phase I (Assess Drug Safety): The Phase I clinical trial is the first experiment in which a drug is tested on the human body. The primary aim of the trial is to assess the safety of the new drug. Phase I trials are usually conducted with open label, i.e. the subjects are aware of the drugs that they are being given. For the first time, the Test drug is introduced to humans. Testing occurs in a small number of healthy volunteers (20 to 100). This initial phase of testing usually lasts several months to 1 year. The goals of phase I clinical trials are Assess safety for humans as well as select the dose to be used in future studies. During phase I, the study is designed to determine how the human body reacts and what side effects occur as dosage levels is increased. Open-label Study: A trial in which subjects and investigators know which product each subject is receiving. Phase II (Test for Safety and Effectiveness):A phase II study provides comparative information about relative safety and effectiveness and efficacy. Most phase II studies are randomized trials. This means One group receives the experimental Test Drug other Control group receives the current standard treatment or Placebo. Some phase II studies are blinded, This means participants and researchers do not know who receives the experimental test drug, this testing may last from several months to 2 years it may involve from 100-300 participants. Phase III (Large-Scale Testing):The objective of Phase III is to confirm the efficacy of the drug in a large patient group. This trial is normally conducted in several hospitals Y Lakshmi Prasad 5 in different demographic locations, to determine the influence of ethnic responses, so this trial is also known as a multicentre trial. This largescale testing (1,000-3,000 participants/volunteers) provides a better understanding of efficacy. Most phase III studies are randomized and blinded trials with specific entry criteria. Phase III studies typically last several years. After a phase III study is successfully completed, a company can request NDA from the FDA. Randomization: The Process of assigning trial subjects to treatment or control groups using an element of chance to determine the assignments in order to reduce bias. Blinded study: A study in which the subject, the investigator, or anyone assessing the outcome is unaware of the treatment assignment(s). Control group: The group of subjects in a controlled study that receives no treatment, a standard treatment or a placebo. Placebo: A pharmaceutical preparation that does not contain the investigational agent. Multicenter trial: Clinical trial conducted according to a single protocol but at more than one site and therefore, carried out by more than one investigator. Food and Drug Administration (FDA): The United States regulatory authority charged with, among other responsibilities, granting IND and NDA approvals. New Drug Application (NDA): An application to FDA for a license to market a new drug in the United States. Licensing (Approval to Use): After all three clinical trial phases are complete and, if the research demonstrates that the test drug is safe and effective, a New Drug Application (NDA)/ Biologics License Application (BLA) is filed with the FDA. This NDA/BLA must contain all scientific information compiled over the course of the trials. The FDA is allowed at least 6 months to review the NDA/BLA. However, this review Process can sometimes take up to 2 years, depending on specific country requirements. 6 SAS Clinical Programming Approval (Available for Prescription): Health care providers are able to prescribe. Even after approval, reviews continue to ensure safety over time. For example, all cases of adverse events must be reported, and quality control standards must be met (Sometimes studies to evaluate long-term effects are also required). The accelerated approval Process for serious diseases is designed to help development of treatments and to fill an unmet medical need to get important new treatments to patients faster. Post-Marketing Studies: Post-marketing studies (special studies and long-term effectiveness/ use) are also called Phase IV studies. These studies are often performed in special populations not previously studied (for example, children or the elderly) the studies are designed to monitor Long-term effectiveness and/or efficacy and the impact on a person’s quality of life. Some studies help determine the cost -effectiveness of a therapy compared to other traditional and new therapies. Industry Regulations and Standards for Clinical Trials Every trial has to be approved and carried out under regulatory compliance to comply with GCP requirements. Different countries have different requirements for clinical trials. Two organizations that carry significant regulatory weight are the combined forces of the US Food and Drug Administration (FDA) and International Conference on Harmonization of Technical Requirements for Registration of Pharmaceuticals for Human Use (ICH). It is the role of public regulatory authorities to ensure that pharmaceutical companies comply with regulations. There is legislation that requires drugs to be developed, tested, trialed and manufactured in accordance to guidelines so that they are safe and patient’s well being is protected. Regulatory authorities perform the watchdog role to ensure that animal studies comply with Good Laboratory Practice (GLP), clinical trials are performed in accordance with Good Clinical Practice (GCP) and drugs are manufactured under current Good Manufacturing Practice (cGMP) conditions. ICH stands for “International Conference on Harmonization of Technical Requirements for Registration of Pharmaceuticals for Y Lakshmi Prasad 7 Human Use”. ICH is a joint initiative involving both regulators and research-based industry representatives of the EU, Japan and the US in scientific and technical discussions of the testing Procedures required to assess and ensure the safety, quality and efficacy of medicines. The objective of ICH is to increase international harmonization of technical requirements to ensure that safe, effective and high quality medicines are developed and registered in the most efficient and cost effective manner. The ICH Topics are divided into four major categories: Quality (Q): Those relating to chemical and pharmaceutical Quality Assurance. Safety (S): Those relating to in vitro and in vivo pre clinical studies. Efficacy (E): Those relating to clinical studies in human subject. Multidisciplinary topics (M): Topics which do not fit uniquely into one of the above categories. Efficacy Topics: E3: Structure and Content of Clinical Study Reports E4: Dose-Response Information to Support Drug Registration E6: Good Clinical Practice: Consolidated Guideline E8: General Considerations for Clinical Trials E9: Statistical Principles for Clinical Trials E10: Choice of Control Group and Related Issues in Clinical Trials ICH’s mission is to make recommendations towards achieving greater harmonization in the interpretation and application of technical Guidelines and requirements for pharmaceutical product registration. The ICH Steering Committee and its sub-committee are comprised of representatives from six parties that represent the regulatory bodies and research-based industry in the European Union, Japan and the USA. 8 SAS Clinical Programming Region Japan Europe USA Regulatory Body MHLW - Ministry of Health, Labour and Welfare EU - European Union FDA - Food and Drug Administration Research Based Industry JPMA-Japan Pharmaceutical Manufacturers Association EFPIA - European Federation of Pharmaceutical Industries and Associations PhRMA - Pharmaceutical Research and Manufacturers of America U.S.FDA: Branch of the United States Department of Health and Human Services and regulates all aspects of pharmaceutical industry. 21 CFR Part 11 Compliance: In your research the computer systems used to collect and analyze data must be validated to meet the FDA requirements for electronic records and signatures. Title 21 of the Code of Federal Regulations (CFR): Parts applicable to clinical research: Part 11 - Electronic Records and Signatures Part 50 - Protection of Human Subjects Part 54 - Financial Disclosure by Clinical Investigators Part 56 - Institutional Review Boards Part 312 - Investigational New Drug Application Part 314 - Applications for FDA Approval to Market a New Drug or an Antibiotic Drug Part 600 - Biological Products Part 812 - Medical Devices Departments and Roles in a CRO Contract Research Organization: CRO is an organization that provides support to the Pharmaceutical, biotechnology, and medical device industries in the form of research services outsourced on a contract basis. A CRO may provide such services as Pre Clinical Research, Clinical Research, Clinical Trials Management, Clinical Data Management, Bio statistics and SAS Programming. Y Lakshmi Prasad 9 A Typical Clinical Research Organization may contain the following departments: Clinical Operations Data Management Medical writing Pharmacovigilance Biostatistics/ SAS Programming Regulatory affairs Key Functions in Clinical Operations: Managing and coordination of study conduct Monitoring and tracking of project milestones to ensure that the project runs within timelines. Participation as appropriate to expedite the feasibility and conduct of global trials Ensuring that the regulatory submission are of acceptable quality Support Investigator as and when required (e.g. Finalization of Investigator agreements and contracts, Finalization of Protocol and CRF) Key Functions in Data Management: Data Entry Database creation, Updating, Validation and Database lock Data Quality Check Data Clarification Form generation Coordination with Operations team to resolve queries 10 SAS Clinical Programming Protocol: Clinical research is conducted according to a plan (a protocol) or action plan. The plan describes what will be done in the study, how it will be conducted, and why each part of the study is necessary. The protocol or plan is carefully designed to safeguard the participant’s health and answer specific research questions. Case Report Form (CRF). A printed, optical, or electronic document designed to record all of the protocol-required information to be reported to the sponsor for each trial subject. Key Functions in Quality Assurance: Facilitate audits which are conducted by clients locally within the country Ensure that all staff within the country has a complete and current training record Facilitate the auditing of suppliers and vendors used by company within the country Ensure that all GCP compliance issues with sites or elsewhere are raised to the Director of Quality Assurance and the Director of Medical Affairs Maintaining version control of SOPs to ensure that all staff are following the correct and up to date SOPs Key Functions in Pharmacovigilance: Collect, follow-up, transmit all local adverse events, and pregnancy cases, to Global Pharmacovigilance. Process cases in accordance Pharmacovigilance Procedures. with Global and Local Answer ADR and ADR case processing questions from local Regulatory Authorities and Health Care Professionals. Submit the report able ADRs, to the local Regulatory Authorities according to the national regulations and answer any subsequent questions in collaboration with the Global Pharmacovigilance. Y Lakshmi Prasad 11 Key Functions in Medical Writing: Clinical Study Protocol Writing Documentation for Regulatory Submission Technical Documentation for Clinical Trials Writing Medical Cases Managing SAEs during clinical trials Closely associated with regulatory department in preparing narratives for submission Standard Operating Procedure (SOP): Detailed, written instructions to achieve uniformity of the performance of a specific function. Adverse Drug Reaction (ADR): Any noxious and unintended response associated with the use of a drug in humans. Serious Adverse Event (SAE ): Any untoward medical occurrence that at any dose: results in death, is life threatening, requires inpatient hospitalization or prolongation of existing hospitalization, results in persistent or significant disability/incapacity, or is a congenital anomaly / birth defect. 12 SAS Clinical Programming Process Flow in a Typical Clinical Trial Protocol Development Blank CRF SITE CLINICAL OPERATIONS CRF designing Database Designing Filled CRF DCF Edit check Programming Data Entry Discrepancy DATA MANAGEMENT Data Validation Database Lock Data Extract CDISC Conversion BIO STATISTICS ADS Creation TLF Creation Study Submission Process Flow in a Typical Clinical Trial: Every clinical investigation begins with the development of a clinical protocol. The protocol is a document that describes how a clinical trial will be conducted and ensures the safety of the trial subjects and integrity of the data collected. After preparing the protocol we go with CRF designing, The Case Report Form is a data-reporting document used in a clinical study. CRF Collects relevant data in a specific format in accordance with the protocol and compliance with regulatory requirements which SAS Clinical Programming In 18 Easy Steps FOR M.Pharm / M.Sc (Life Sciences) /M.sc (Stats)/ M.C.A/M.Tech / B.Pharm /Bio-Tech/B.Tech.