* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Enzyme Kinetics and Mechanisms
Multi-state modeling of biomolecules wikipedia , lookup
Citric acid cycle wikipedia , lookup
Metabolic network modelling wikipedia , lookup
NADH:ubiquinone oxidoreductase (H+-translocating) wikipedia , lookup
Basal metabolic rate wikipedia , lookup
Amino acid synthesis wikipedia , lookup
Nucleic acid analogue wikipedia , lookup
Oxidative phosphorylation wikipedia , lookup
Biochemistry wikipedia , lookup
Deoxyribozyme wikipedia , lookup
Enzyme inhibitor wikipedia , lookup
Metalloprotein wikipedia , lookup
Biosynthesis wikipedia , lookup
Photosynthetic reaction centre wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
King Saud University
College of Science
Department of Biochemistry
Disclaimer
The texts, tables and images contained in this course presentation
are not my own, they can be found on:
References supplied
Atlases or
The web
Mechanism of Enzyme Action
BCH 321
Professor A. S. Alhomida
King Saud University
College of Science
Department of Biochemistry
Disclaimer
The texts, tables and images contained in this course presentation
(BCH 320) are not my own, they can be found on:
References supplied
Atlases or
The web
Mechanism of Enzyme Action
Enzyme Kinetic and Mechanism
Professor A. S. Alhomida
Enzyme Catalysis
Catalysts
Chemical
catalysts
Biological
catalysts
Acids, bases,
metals
Proteins
Enzymes
Nonallostrics
Nonproteins
Ribuzymes
Allostrics
Abnzymes
‡,
Catalyst only lower DG but not
effect on the EQM positions
1.
2.
3.
4.
Stabilization the transition state
Destabilizing substrate bound at the
binding site
Destabilizing ES complex
Forming an intermediate
Types of Enzymatic Catalysis
1.
Approximation (Proximity) and
Orientation (Entropy
Contribution)
(a) Intermolecular catalysis
(b) Intramolecular catalysis
(c) Effective morality (concentration)
Types of Enzymatic Catalysis
2. Preferential Binding of Transition
Sstate (TS)
(a) Oxyanion hole
(b) Strain or distortion
(c) Transition analongs
Types of Enzymatic Catalysis
3. Electrostatic Catalysis
4. General Acid-base Catalysis
(a) General acid
(b) General base
(c) Concerted acid/base
Types of Enzymatic Catalysis
5. Nucleophilic-Electrophilic
Catalysis
(a) Covalent catalysis
(b) Schiff base catalysis
(c) Electron sink (electron flow) catalysis
Types of Enzymatic Catalysis
6. Metal ion Catalysis
(a) Metalloenzymes
Contain tightly bound metal cofactors such as
Fe2+, Fe3+, Cu2+, Zn2+, Mn2+, Co2+
(b) Metal Activated Enzymes
Only loosely bind the metal ions.
The ions are usually Na+, K+, Mg2+, or Ca2+
Enzyme Catalysis
Enzymes endow cells with the remarkable
capacity to exert kinetic control over
thermodynamic potentiality
Enzymes are the agents of metabolic
function
Enzyme Catalysis
Enzyme Catalysis
1. Enzyme works simply by lowering the
energy barrier of a reaction. By doing so,
the enzyme increases the fraction of
molecules that have enough energy to
attain the transition state, thus making
the reaction go faster in both directions
Enzyme Catalysis
2. The position of the equilibrium (the amount of
product versus reactant) is unchanged by an
enzyme.
3. Even though K1, K-1 many be greatly changed
from their values in the absence of an enzyme,
each one changes by the same factor and the
equilibrium constant, K, is unchanged, because
K = k1/k-1
Catalytic Power
Enzymes can accelerate reactions as
much as 1016 over uncatalyzed rates!
Urease is a good example:
Catalyzed rate: 3x104/sec
Uncatalyzed rate: 3x10 -10/sec
Ratio is 1x1014 !
Catalytic Power
Specificity
Enzymes selectively recognize proper
substrates over other molecules
Enzymes produce products in very high
yields - often much greater than 95%
Specificity is controlled by structure - the
unique fit of substrate with enzyme
controls the selectivity for substrate and
the product yield
Enzyme Kinetics
Several terms to know!
rate or velocity
rate constant
rate law
order of a reaction
molecularity of a reaction
The Transition State
Understand the difference between DG
and DG‡
The overall free energy change for a
reaction is related to the equilibrium
constant
The free energy of activation for a reaction
is related to the rate constant
It is extremely important to appreciate this
distinction!
What Enzymes Do....
Enzymes accelerate reactions by lowering
the free energy of activation
Enzymes do this by binding the transition
state of the reaction better than the
substrate
Much more of this in Chapter 16!
The Michaelis-Menten Equation
You should be able to derive this!
Louis Michaelis and Maude Menten's theory
It assumes the formation of an enzymesubstrate complex
It assumes that the ES complex is in rapid
equilibrium with free enzyme
Breakdown of ES to form products is assumed
to be slower than 1) formation of ES and 2)
breakdown of ES to re-form E and S
Understanding Km
The "kinetic activator constant"
Km is a constant
Km is a constant derived from rate constants
Km is, under true Michaelis-Menten
conditions, an estimate of the dissociation
constant of E from S
Small Km means tight binding; high Km
means weak binding
Understanding Vmax
The theoretical maximal velocity
Vmax is a constant
Vmax is the theoretical maximal rate of the
reaction - but it is NEVER achieved in reality
To reach Vmax would require that ALL enzyme
molecules are tightly bound with substrate
Vmax is asymptotically approached as
substrate is increased
The dual nature of the
Michaelis-Menten equation
Combination of 0-order and 1st-order kinetics
When S is low, the equation for rate is 1st
order in S
When S is high, the equation for rate is 0order in S
The Michaelis-Menten equation describes a
rectangular hyperbolic dependence of v on
S!
The turnover number
A measure of catalytic activity
kcat, the turnover number, is the number of
substrate molecules converted to product
per enzyme molecule per unit of time,
when E is saturated with substrate.
If the M-M model fits, k2 = kcat = Vmax/Et
Values of kcat range from less than 1/sec
to many millions per sec
The catalytic efficiency
Name for kcat/Km
An estimate of "how perfect" the enzyme is
kcat/Km is an apparent second-order rate
constant
It measures how the enzyme performs
when S is low
The upper limit for kcat/Km is the diffusion
limit - the rate at which E and S diffuse
together
Linear Plots of the MichaelisMenten Equation
Be able to derive these equations!
Lineweaver-Burk
Hanes-Woolf
Hanes-Woolf is best - why?
Smaller and more consistent errors across
the plot
Inhibitions
Reversibles
Compatitives
Umcompatitives
Km
Increase
Decrease
Vmax
No Change
Decrease
Irreversibles
Noncompatitives
No Change
Decrease
Enzyme Inhibitors
Reversible versus Irreversible
Reversible inhibitors interact with an
enzyme via noncovalent associations
Irreversible inhibitors interact with an
enzyme via covalent associations
Classes of Inhibition
Two real, one hypothetical
Competitive inhibition - inhibitor (I) binds
only to E, not to ES
Noncompetitive inhibition - inhibitor (I) binds
either to E and/or to ES
Uncompetitive inhibition - inhibitor (I) binds
only to ES, not to E. This is a hypothetical
case that has never been documented for a
real enzyme, but which makes a useful
contrast to competitive inhibition
Ribozymes and Abzymes
Relatively new discoveries
Ribozymes - segments of RNA that display
enzyme activity in the absence of protein
Examples: RNase P and peptidyl transferase
Abzymes - antibodies raised to bind the
transition state of a reaction of interest
For a great recent review, see Science, Vol. 269,
pages 1835-1842 (1995)
We'll say more about transition states in Ch 16
Mechanisms of
Enzyme Action
Mechanisms of Enzyme Action
Stabilization of the Transition State
Enormous Rate Accelerations
Binding Energy of ES
Entropy Loss and Destabilization of ES
Transition States Bind Tightly
Types of Enzyme Catalysis
Serine
Proteases
Aspartic Proteases
Lysozyme
Enzyme Catalysis
Reactions in solution that are not
catalyzed are slow since charge
development and separation occurs in the
transition state.
When bonds are made or broken, charged
intermediates are often formed which are
higher in energy than the reactants.
Enzyme Catalysis
Since the intermediate is higher in energy
than the reactants, the transition state
would be even higher in energy, and
hence more closely resemble the charged
intermediate.
Enzyme Catalysis
Anything that can stabilize the charges on the
intermediate and hence the developing charges
in the transition states will lower the energy of
the transition state and catalyze the reaction.
In this section will investigate the mechanism
underlying the catalysis by small molecules of
chemical reactions.
Enzyme Catalysis
Presumably, biological macromolecular
catalyst (like protein enzymes) will use
similar mechanisms in their catalytic
effects (which will be discussed in the next
section).
Approximation Catalysis
Enzyme serves as a template to bind
the substrates so that they are close
to each other in the reaction center.
Bring substrate into contact with catalytic
groups or other substrates.
Correct orientation for bond formation.
Freeze translational and rotational motion.
Catalysis by Approximation
The classic way that an enzyme increases
the rate of a bimolecular reaction is to use
binding energy to simply bring the two
reactants in close proximity.
If DG‡ is the change in free energy between
the ground state and the transition state, then
DG‡=DH‡–TDS‡.
In solution, the transition state would be
significantly more ordered than the ground
state, and DS‡ would therefore be negative.
Catalysis by Approximation
The formation of a transition state is
accompanied by losses in translational
entropy as well as rotational entropy.
Enzymatic reactions take place within the
confines of the enzyme active-site wherein
the substrate and catalytic groups on the
enzyme act as one molecule.
Therefore, there is no loss in translational or
rotational energy in going to the transition
state.
This is paid for by binding energy.
Approximation Catalysis
a)
Bimolecular reaction (high
activation energy, low rate).
b)
Unimolecular reaction, rate
enhanced by factor of 105 due to
increased probability of
collision/reaction of the 2 groups
c)
Constraint of structure to orient
groups better (elimination of
freedom of rotation around bonds
between reactive groups), rate
enhanced by another factor of 103,
for 108 total rate enhancement
over bimolecular reaction
Approximation Catalysis
Desolvation:
•
When substrate binds to the enzyme surrounding water in
solution is replaced by the enzyme. This makes the substrate
more reactive by destablizing the charge on the substrate.
•
Expose a water charged group on the substrate for interaction
with the enzyme.
•
Also lowers the entropy of the substrate (more ordered).
Approximation Catalysis
Strain
and Distortion:
When substrate bind to the enzyme, it may induces a
conformational change in the active site to fit to a
transition state.
Frequently, in the transition state, the substrate and the
enzyme have slightly different structure (strain or
distortion) and increase the reactivity of the substrate.
cyclic phosphate ester
Rate:
108
Acylic phospodiester
1
Strain and Distortion
1. Intramolecular Catalysis
Consider the hydrolysis of phenylacetate.
This reaction, a nucleophilic subsitution
reaction, could be catalyzed by the
addition to solution of the general base
acetate, as described above.
Intramolecular Catalysis
Since this reaction would double with the
doubling of the solution acetate, the
reaction is bimolecular (first order in
reactant and catalyst).
Now consider the same reaction only
when the the general base part of the
catalyst, the carboxyl group, is part of the
reactant phenylacetate.
Intramolecular Catalysis
Such a case occurs in the acetylated
form of salicylic acid - i.e. aspirin. When
the carboxy group is ortho compared to
the acetylated phenolic OH, it is in
perfect position to accept a proton from
water, decreasing the charge
development on the O in the transition
state.
Intramolecular Catalysis
The general base does not have to diffuse
to the appropriate site when it is
intramolecular with respect to the carbonyl
C of the ester link.
The rate of this intramolecular base
catalysis is about 100 fold greater than of
an intermolecular base catalyst like
acetate.
Intramolecular Catalysis
It is as if the effective concentration of
the intramolecular carboxyl base catalyst
is much higher due to its proximity to the
reaction site.
Intramolecular Catalysis
Another type of reactions involving a carboxyl
group (in addition to simple proton transfer) is
when the negatively charged carboxyl O acts as
a nucleophile and attacks an electrophilic
carbonyl carbon.
When the carbonyl is part of an ester, the
carboxyl group engages in a nucleophilic
substitution reaction, expelling the alcohol part
of the ester as a leaving group.
Intramolecular Catalysis
The remaining examples below consider
the nucleophilic (carboxyl) substitution on
phenylesters, with phenolate as the
leaving group. The reactions in effect
transfer an acyl group to the carboxyl
group to create an anhydride.
Intramolecular Catalysis
First consider acyl transfer with aspirin
derivatives.
Aspirin, as you know, contains a carboxyl group
ortho to an ester substitutent.
Hence the carboxyl group can act as a
nucleophile and attack the carbonyl carbon of
the ester in a nucleophilic substitution reaction.
Intramolecular Catalysis
The net effect is to transfer the acetyl
group from the phenolic OH to the
carboxyl group converting it to an
anhydride.
This is an intramolecular reaction.
Compare this reaction to a a comparable
bimolecular reaction shown below.
Acyl Transfer Aspirin Derivatives
Intramolecular
Intermolecular
Intramolecular Catalysis
The first order rate constant of the
intramolecular transfer of the acetyl group
to the carboxyl group, k1 = 0.02 s-1.
The analogous bimolecular reaction rate
constant k2~ 10-10 M-1s-1.
Intramolecular Catalysis
Dividing k1/k2 gives the relative rate
enhancement of the intramolecular over the
intermolecular reaction.
With units of molarity, this ratio can be
interpreted as the relative effective
concentration of the intramolecular
nucleophile.
This makes the effective concentration of the
carboxylate in the aspirin derivative 2 x 107 M.
Mechanism of Acetate with
Phenylacetate
2. Intermolecular Catalysis
Now consider the cleavage of phenylacetate
using acetate as the nucleophile.
The products are acetic anhydride and
phenolate.
This is a bimolecular reaction (a slow one at
that), with a bimolecular rate constant, k2 which I
will arbitrarily set to 1 for comparison to some
similar reactions.
Intermolecular Catalysis
Now consider a monoester derivatives of
succinic acid - phenyl succinate - in which
the free carboxyl group of the ester attacks
the carbonyl carbon of the ester derivative.
Intermolecular Catalysis
Intermolecular Catalysis
If you assign a second order rate constant k2
= 1 M-1s-1 to the analogous intermolecular
reaction of acetate with phenylacetate (as
described above), the first order rate
constant for the intramolecular reaction of
phenylsuccinate is 105 s-1.
The ratio of rate constants, k1/k2 = 105 M.
Intermolecular Catalysis
That is it would take 105 M concentration
of acetate reacting with 1 M
phenylacetate in the first bimolecular
reaction to get a reaction as fast as the
intramolecular reaction of
phenylsuccinate.
An even more sterically restricated
bicyclic phenylcarboxylate shows a k1/k2
= 108 M.
Intramolecular Catalysis
Intermolecular Catalysis
Another example is anhydride formation
between two carboxyl groups.
The DGo for such a reaction is positive,
suggesting an unfavorable reaction.
Consider two acetic acid molecules
condensing to form acetic anhydride.
For this intermolecular reaction, Keq =
3x10-12 M-1.
Intermolecular Catalysis
Now consider the analogous intramolecular
reaction of the dicarboxylic acid succinic acid.
It condenses in an intramolecular reaction to
form succinic anhydride with a Keq = 8x10-7 (no
units).
The ratio Keq-intra/Keq inter = 3 x 105 M.
It is as if the effective concentration of the
reacting groups. because they do not have to
diffuse together to react, is 3 x 105 M.
3. Enzyme Catalysis
How does this apply to enzyme catalyzed
reaction?
Enzymes bind substrates in physical steps
which are typically fast.
The slow step is chemical conversion of
the bound substrate, which is effectively
intramolecular.
Enzyme Catalysis
These three kinds of reactions,
intermolecular, intramolecular, and
enzyme-catalysed can be broken down
into two hypothetical steps, a binding
followed by catalysis.
Intramolecular, Intermolecular
and Enzyme-Catalyzed Reaction
Intramolecular, Intermolecular and
Enzyme-Catalyzed Reaction
If the rate constants for the chemical
steps are all identical, the advantage of
the intramolecular and enzymecatalyzed reaction over the
intermolecular reaction is KINTRA/KINTER
and KENZ/KINTER, respectively.
Intramolecular, Intermolecular
and Enzyme-Catalyzed Reaction
The advantage of intramolecular
reactions can be seen by studying the
Ca-EDTA complex.
Calcium in solution exists as a
octahedrally coordinated complex with
water occupying all the coordination
sites.
Intramolecular, Intermolecular
and Enzyme-Catalyzed Reaction
EDTA, a multidentate ligand, first
interacts through one of its potential six
electron donors to Ca in a reaction
which is entropically disfavored from the
the Ca-EDTA perspective, although one
water is released.
Intramolecular, Intermolecular
and Enzyme-Catalyzed Reaction
Once this first intramolecular complex is
formed, the rest of the ligands on the EDTA
rapidly coordinate with the Ca and release
bound water.
The former is no longer entropically
disfavored since it is now an intramolecular
process while the later is favored through the
release of the remaining five water
molecules.
Mechanism of Binding of Ca2+
and EDTA
Intramolecular, Intermolecular
and Enzyme-Catalyzed Reaction
We modeled the catalytic advantage
offered by intramolecular reaction in terms of
a dramatic increase in the effective
concentration of reactants, which sometimes
reached levels of 108 M.
Another way is to look at entropy changes
associated with dimer formation.
Entropy and Catalysis
H 2O
O
C
k
ROH
k
O
OR
C
hydrol
hydrol (glutarate)
OH
1.0
C
O
C
R=C6 H5 –
O
O
O
glutarate
phenylglutarate
O
H 3C
C
C
H 3C
OR
20
O
O
O
C
OR
C
23 0
O
O
O
C
OR
10 ,000
C
O
O
O
C
OR
C
O
O
O
53 ,000
Orientation Effects
In the non-enzymatic lactonization reaction shown below, the relative rate
when R = CH3 is 3.4 x1011 times that when R = H. What is the explanation?
OH
COOH
CH 2
O
O C
CH 2
R R
R
R
R
R
+ H2O
Models of Approximation (1)
H 2O
O
P
C2H 5OH
O
OC 2H 5
OC 2H 5
C
P
t1/2 = 15 min at 36°C & pH 5.0
OH
OC 2H 5
C
O
OH
O
O
O
P
OC 2H 5
OC 2H 5
O
No detectable hydrolysis after 77,960 hours
–9 years!
C
O
O
P
OH
OC 2H 5
C
OCH 3
No reaction
O
What two mechanisms can you write to account for this?
Catalysis by Approximation
1. In order for a reaction to take place between two molecules,
the molecules must first find each other.
2. This is why the rate of a reaction is dependent upon the
concentrations of the reactants, since there is a higher
probability that two molecules will collide at high
concentrations.
3. As an example, look at the hydrolysis of paranitrophenyl ester
again catalyzed by imidazole. This reaction depends on both
the concentration of imidazole and paranitrophenyl ester,
therefore, it proceeds with a Second Order Rate Constant of 35
M-1min-1.
Catalysis by Approximation
4. In the second reaction, the imidazole catalyst is actually part of
the substrate that is being hydrolyzed. Therefore, the rate of
hydrolysis is dependent only on the substrate, and therefore
proceeds with a First Order Rate Constant of 839 min-1.
5. Rate constants of different order cannot be compared.
However, the ratio of the first order rate constant to the second
order rate constant gives an effective Molarity.
6. In order for the second order reaction to be as fast as the first
order reaction, it would be necessary to have imidazole at a
concentration of 24 M!
Catalysis by Approximation
Effective Concentration
O
C
O
C
O
k1 = 0.8 s - 1
N O2
O
O
C
+ HO
O
Effective concentration is
k1/k2 = 2 x 105 M
N O2
C
O
O
+
CH3 CO 2
O
H 3C
N O2
O
O
+
H 3C
K 2 = 4 x 10 - 6 s- 1 M- 1
O
HO
N O2
CH 3
CO 2H
CO 2H
k1 •
0.02 s
CO 2
O
O
-1
O
O
O
O
CH 3
CO 2
k2 = 10- 10 s- 1 M- 1
O
O
Effective concentration
CH 3
O
O
O
= 2 x 107 M
CH 3
O
+
CH 3
2. Preferential Binding of TS
Catalysis
Enzymes typically bind the TS of the reactions
with greater affinity than the substrates or
products
This increases the effective concentration of the
TS with proportionally increases the reaction
rate
TS analogs are extremely potent enzyme
inhibitors
Importance of Binding Energy
On the left are
examples of reaction
coordinates of an
uncatalyzed reaction,
and one that is
enzyme catalyzed.
The active sites of
enzymes tend to be
more complementary
to the transition states
of their respective
reactions than they are
to the actual
substrates.
Importance of Binding Energy
This results in lowering
the energy of the
enzyme–transition
state complex,
meaning, a lowering of
the activation energy.
In order for catalysis to
be effective, the
energy barrier
between ES and EXt
must be less than S
and Xt.
Importance of Binding Energy
Notice that the binding
of substrate to enzyme
lowers the free energy
of the ES complex
relative to substrate.
If the energy is
lowered too much,
without a greater
lowering of EXt, then
catalysis would not
take place.
Transition State Stabilization
Linus Pauling postulated long ago that the only
thing that a catalyst must do is bind the transition
state more tightly than the substrate.
That this must be the case can be seen from the
diagram below, which shows how S and S* (the
transition state) can react with E to form a
complex which then proceeds to product, or can
go to product in the absence of E.
Transition State Stabilization
For an enzyme to be a catalyst the
activation energy for the reaction in the
presence of E, d, must be less than in the
absence of enzyme, c.
Therefore c-d = a-b > 0.
Since DGo = -RTln Keq, Keq for binding of
S* to E is greater than for S binding to E.
Enzyme Bind the TS Tightly
Transition State Stabilization
The stability of the transition state also affects the
reaction kinetics (which makes sense given that the
activation energy clearly affects the speed of a
reaction).
As you probably remember from organic chemistry, SN2
reactions are slow when the central atom where the
substitution will occur is surrounded by bulky
substitutents. (Sterics once again.)
We discussed this in context to nucleophiliic substitution
on a sp2 hybridized carbonyl carbon in carboxylic acid
derivatives versus on a sp3 hybridized phosphorous in
phosphoesters and diesters.
Transition State Stabilization
The explanation for this phenomena has usually been
attributed to hindered access of the central atom caused
by bulky substituents (intrinsic effects). Is this true?
Recent studies on SN2 reactions of
methylchloroacetonitrile and t-butylchloroacetonitrile
(with the reagent labeled with 35Cl) using 37Cl- as the
incoming nucleophile in the gas phase
It shown that the more hindered t-butyl derivative's
activation energy was only 1.6 kcal/mol higher than the
methyl derivative, but in aqueous solution, the difference
is much greater for comparable reactions.
Transition State Stabilization
The explanation for this phenomena has usually
been attributed to They attributed the differences
to solvation effects of the transition state.
The bulkier the substituents on the central atom,
the more difficult it is to solvate the transition
state since water can't reorient around it as
well. In effect there is steric hindrance for both
reactant and solvent.
Transition State Analogs
Stabilizing the Transition State
Rate acceleration by an enzyme means that
the energy barrier between ES and EX‡ must
be smaller than the barrier between S and X‡
This means that the enzyme must stabilize
the EX‡ transition state more than it stabilizes
ES
3. Electrostatic Catalysis
Water is generally excluded from an
enzyme active site
The chemical environment of an enzyme
active site is more like that of found in
organic solvents
The proximity interactions perturbs the
pKs of the amino acid side chains
Interactions generally favor the TS
Electrostatic Catalysis
Electrostatic Catalysis
Electrostatic Catalysis
4. General Acid-base Catalysis
1.
General Acid Catalysis
It donates a proton to substrate
Enzyme active site residue must be protonated
2.
General Base Catalysis
It accepts a proton from the substrate
Enzyme active site residue must be deprotonated
3.
Concerted Acid-base Catalysis
It is acid and base both participate in the
reaction
General Acid-base Catalysis
1. General acid
(a) Partial transfer for a proton from a Bronsted
acid lowers the free energy of TS
(b) Rate of reaction increases with decrease in
pH and increase in [Bronsted acid]
2. Specific acid
Protonation lowers the free energy of the TS
Rate of reaction increases with decrease in pH
General Acid-base Catalysis
3. General base
Partial abstraction of a proton by a Bronsted
base lowers the free energy of TS
Rate of reaction increases with increase in
Bronsted base
4. Specific base
Abstraction of a proton (or nucleophilic attack)
by OH- lowers the free energy of TS
Rate of reaction increases with increase in pH
General Acid-Base Catalysis
General base catalysis
Specific base catalysis
General acid-base catalysis is
involved in a majority of
enzymatic reactions. General
acid–base catalysis needs to be
distinguished from specific
acid–base catalysis.
Specific acid–base catalysis
means specifically, –OH or H+
accelerates the reaction. The
reaction rate is dependent on
pH only, and not on buffer
concentration.
In General acid–base catalysis,
the buffer aids in stabilizing the
transition state via donation or
removal of a proton. Therefore,
the rate of the reaction is
dependent on the buffer
concentration, as well as the
appropriate protonation state.
General Acid-Base Catalysis
Conventions for Describing
General Acid/Base Catalysis
The dehydration reaction below is catalyzed by an enzyme at pH 7 and 25°C. This reaction
does not occur nonenzymatically under these conditions. Sketch a mechanism to show how
an enzyme can easily catalyze this reaction.
O
–H2O
OH
+H2O
O
Models for General Acid-Base Catalysis
O
O
H
O H
O
O
CH 3
OH
O
aspirin
O
+
HO
CH 3
O
Hydrolysis rate 100 fold faster than
O
CH 3
O
If base had pKa of 7 rather than 3.7 (carboxylate), the rate enhancement
would be 5000.
General Acid-base Catalysis
Charge development in the TS can be
decreased by either donation of a proton
from general acids (like acetic acid or a
protonated indole ring) to an atom such as
a carbonyl O which develops a partial
negative charge in the TS when it is
attached by a nucleophile.
General Acid-base Catalysis
Proton donation decreases the developing
negative in the TS.
Alternatively, a nucleophile such as water
which develops a partial positive charge in
the TS as it begins to form a bond to an
electrophilic C in a carbonyl
General Acid-base Catalysis
Can be stabilized by the presence of a
general base (such as acetate or the
deprotonated indole ring).
Proton abstraction decreases the
developing positive charge
Charge Development in TS for
Ester Hydrolysis
Mechanism of General Acid
Catalysis
Mechanism of General Base
Catalysis
Acid-bases Catalysis
The rate of proton transfer
Mechanism:
B + H A
B
(1)
H A
B H
(2)
A
B H + A
(3)
1) The diffusion-controlled formation of a hydrogen bond
between the base B– and the acid HA;
2) The transfer of a proton, leading to the formation of a new
hydrogen bonded complex;
3) The diffusion-controlled dissociation of the product.
- proton transfer to N, O, S is fast;
- proton transfer to carbon (C) is slow
- proton transfer from H3O+ to N or O is diffusion-controlled:
see the Table on p 31, left column, k-1 1011 l.mol-1.s-1
AH + H2O
k1
k -1
A + H3O
Formation of the hydrogen bond between the proton donor
and the proton acceptor is the rate determining step: this step
is slower than the actual proton transfer!
Typical for cases where a proton is both bound to and
transferred to oxygen or nitrogen atoms, where the negative
charge is localised on one atom.
Nevertheless, diffusion of H+ in water is much faster than
diffusion of other ions:
acid:
base:
H
O+ H
H
H
O
H
O
H
A
H
H O
H A
H
O
H
H
O H
H A
H O
H
H
O
H
A
Proton transfer from/to carbon acids/bases
H3C
O
C N
H2 O
H3C
O
C N
H
O
k1
+ H2O
k -1
H3C
O
C N
H
O
+ H3O+
k1 = 4x10-8 l.mol-1.s-1, slow process due to:
- lack of hydrogen bond formation before proton transfer
- low acidity of the hydrogen atoms.
k-1 = 16 l.mol-1.s-1, also much slower than diffusion rate, because:
- upon protonation a considerable redistribution of charge has to take place,
including the concomitant change of solvation.
Another example:
H
O
O
H3C
CH 3
H H
1
1
2
O
4x104
107
O
H3C
CH 3
H
2
O
107
1010
O
H3C
CH 3
H
3
2: carbon acid, k1 and k-1 relatively small
3: oxygen acid, k1 and k-1 relatively large (H-bond formation)
Influence of pH on reaction rate
The hydrolysis of esters is catalysed by both acid and base:
H3C
H2O
O CH 3
O
H3C
O CH 3
H2O
H
OH
H3C
OCH 3
OH2
H3C
OH
H3C
OCH 3
O
H
OH
O
OCH 3
OH H
O
H3C
+ CH3O
OH
log kobs
OH
7.0
pH
acid catalysed
hydrolysis
base catalysed
hydrolysis
Another example: the mutarotation of glucose:
HOH 2C
O H
HO
HO
OH
HO
HOH 2C
O H
HO
OH
HO
HO
acid catalysed
HOH 2C
O
HO
HO
HO
HOH 2C
O
HO
HO
HO
OH
uncatalysed
HOH 2C
O
HO
HO
HO
O
O H
OH
log kobs
HOH 2C
O
HO
HO
HO
OH
7.0
pH
+ H2O
base catalysed
Two mechanisms for acid catalysis
Specific acid catalysis:
- A proton is transferred to the substrate in a rapid preequilibrium;
subsequently, the protonated substrate reacts further to the
product(s) in the rate determining step:
S + HA
SH+
fast
slow
SH+ + A
products
General acid catalysis:
- Proton transfer occurs in a slow, rate determining step;
subsequently, the protonated substrate rapidly reacts to give the
product(s):
slow
SH+ + A
S + HA
SH+
fast
products
Specific acid/base catalysis
Usually found for electronegative elements (O, N), where proton
transfer is fast:
K
E + HA
EH+ + A
fast
The second step is rate determining and can be mono- or
bimolecular:
EH+
k1
H2O
products
or EH+ + H2O
k2
products
Reaction rate:
ν = k1[EH + ] = k1K
[E][HA]
[A - ]
we can now write:
since
slow
(A1 mechanism)
slow
(A2 mechanism)
[H 3O+ ][A - ]
Ka =
[HA]
k 1K
ν=
[E][H 3O + ] = k'[E][H 3O + ]
Ka
So the rate is only dependent on the pH, not on [HA] !!
Example of specific acid catalysis: hydrolysis of acetals (A1 mech.)
OCH 3
OCH 3
H
O
H+
H2O
x
x
+ 2 CH3OH
x
H
kobs
x
x
x
kobs is directly proportional to [H+];
addition of more acid (buffer) at constant
pH has no effect on kobs.
Proton transfer is not rate limiting,
so the mechanism probably reads:
OCH 3
OCH 3 + H3O+
H
OCH 3
H
fast
H2O
H
fast
etcetera
OCH 3
OCH 3
H
[H+]
x x
x
x
x
kobs
slow
- CH3OH
[ClCH2COOH/
ClCH2COO-] (2:1)
x
Example of a reaction that is specific acid catalysed according
to the A2 mechanism:
the hydrolysis of ethyl acetate:
O
H3C
OC2H5
HO OH
H3C
OC2H5
H
+ H3O
+
O
fast
H3C
H
OC2H5
OH
fast
H3C
OH
+ HOC2H5
+ H2O
slow
H2O OH
H3C
OC2H5
fast
Specific base catalysis
Example: the retro-aldol reaction of I:
x
x
O
OH
CH 3
CH 3
H3C
I
base
O
2
x
kobs
H3C
CH 3
x
x
x
[OH-]
kobs is directly proportional to [OH-]. Addition of more base
(in buffer) at constant pH has no effect on kobs;
[OH-] is the only base that occurs in the rate equation.
General acid/base catalysis
Proton transfer is the rate determining step.
Example: the hydrolysis of ortho esters:
H3C
OC2H5
OC2H5 + H2O
OC2H5
H+
O
H3C
+ 2 C2H5OH
OC2H5
III
The reaction is studied in a series of
buffers (m-NO2-Ph-OH/m-NO2-Ph-O–):
reaction rate increases with increasing
buffer concentration, even if the pH
remains constant
k(buffer)
n
{k(H3O+)[H3O+] + k(H2O)[H2O]}[III]
[buffer]
n = {k(H2O)·[H2O] + k(H3O+)·[H3O+] + k(m-NO2-Ph-OH)·[m-NO2-Ph-OH]}·[III]
The relation between general and specific catalysis
Why is there sometimes general and sometimes specific acid/base
catalysis?
• pH: [H+] and [OH-] are very low in neutral solution, whereas
[HA] or [B-] can be high beneficial for general catalysis
• rate of proton transfer: H transfer to and from C atoms is slower
than transfer to N, O, etc. beneficial for general catalysis
• stability of reaction intermediates plays an important role.
Example: compare the hydrolysis of ortho esters and acetals
General acid/base catalysis by enzymes
Enzymes often use general acid or base catalysis:
• They work at neutral pH, so low [H+] and [OH-]
• High effective concentration of general acid/base
• Correct orientation of the acidic/basic group around the
substrate
• Optimum catalysis at pH around pKa
Amino acid residues often have a pKa that is close to neutral
pH and are therefore able to act as a general acid or base
catalyst:
Prototropic groups of enzymes
Amino acid
Acidic group Basic group
N-terminus
a-NH3+
a-NH2
C-terminus
a-COOH
a-COO–
aspartic acid
b-COOH
b-COO–
glutamic acid
g-COOH
g-COO–
histidine
imidazolium ion imidazole
cysteine
–SH
–S–
tyrosine
–C6H4OH
–C6H4O–
lysine
e-NH3+
e-NH2
serine
b-OH
b-O–
threonine
b-OH
b-O–
arginine
–NH–(C=NH2+)NH2 –NH–(C=NH)NH2
peptide bond
R–CO–NH–R’
R–CO–N––R’
pKa
7.8
3.8
4.4
4.6
7.0
8.7
9.6
10.4
13
13
12.5
14.8
The pKa is strongly influenced by its environment: e.g., in
enzymes the pKa of lysine can drop to ~7
5. Nucleophilic-Electrophilic
(Covalent) Catalysis
One way to change the activation energy of the
reaction is to change the reaction mechanism in
ways which introduces new steps with lower
activation energy.
A typical way is to add a nucleophilic catalyst
which forms a covalent intermediate with the
reactant.
The original nucleophile can then interact with
the intermediate in a nucleophilic substitution
reaction.
Nucleophilic power
There is no simple correlation between chemical structure and
nucleophilic power. Nucleophilicity, among others, depends
on:
1. The solvation energy of the nucleophile (which is
influenced by the solvent);
2. The strength of the chemical bond to the electrophile
(the C-Nu bond);
3. The size (steric hindrance);
4. The electronegativity and the polarisability of the
nucleophilic atom in the nucleophile
The effects of the latter factors have been quantified by Edwards:
Edwards equation:
k
= αP + βH
Nucleophilic power = log
k0
- k0 is the rate constant of the reaction with a standard
nucleophile (H2O)
- P = polarisability, related to the refractive index:
R Nu
P = log
R H 2O
(RNu = refractive index of the nucleophile)
- H = basicity, related to the pKa: H = pKa + 1.74
- a and b are dependent on the reaction (usually a >> b)
a and b can be determined by performing a reaction of a
substrate with a set of nucleophiles, like:
Nu + CH3Br
CH 3-Nu + Br
What kind of groups in enzymes
are good nucleophiles:
Aspartate
caboxylates
Glutamates
caboxylates
Cystine
thiol-
Serine
hydroxyl-
Tyrosine
hydroxyl-
Lysine
amino-
Histadine
imidazolyl-
Nucleophile Groups
Electrophilie Groups
Covalent Catalysis
If the nucleophilic catalyst is a better nucleophile
than the original nucleophile (usually water)
then the reaction is catalyzed.
The nucleophilic catalyst and the original
nucleophile usually interact with a carbonyl C in
a substitution reaction, initially forming the
tetrahedral oxyanion intermediate.
Covalent Catalysis
Covalent Catalysis
If an amine is used as the nucleophilic catalyst,
then the initial addition product (a carbinolamine)
can become dehydrated,
Since the free pair of electrons on the N are
more likely to be shared with the carbon to form
a double bond than electrons from the original
carbonyl O, which is more electronegative than
the N).
An imine or Schiff Base forms, with a pKa of
about 7.
Mechanism of Schiff Base Formation
Mechanism of Schiff Base
Formation
This is easily protonated to form a positively
charged N at the former carbonyl O center.
This serves as an excellent electron sink for
decarboxylation reactions of beta-keto acids and
illustrates an important point.
Electrons in chemical reactions can be viewed
as flowing from a source (such as a carboxyl
group) to a sink (such as an nucleophilic
carbonyl O or a positively charged N in a Schiff
base).
Electron Flow (Electron Sink)
In a subsequent section, we will discuss how
protein enzymes use these same catalytic
strategies.
An intriguing question arises: how much of the
structure of a large protein is really needed for
catalysis? Much work has been directed to the
development of small molecule catalysis
mimetics of large protein enzymes.
Just how small can you go in reducing the size
of a protein and still get catalysis.
Electron Flow (Electron Sink)
One important feature of enzyme catalysis is
that they catalyze reactions in which only one
enantiomer is produced. That is, the synthesis
is assymertric.
This is typically a consequence of the
asymmetric enzyme (itself chiral) binding only
one enantiomer as a reactant and/or the
imposition of steric restrictions on the possible
reactions of the bound substrate.
Recently, it has been show that L-Pro alone can
act as such an assymetric catalyst in an aldol
condensation reaction.
Mechanism of Electron Sink
L-Proline Catalysis of Aldol
Condensation Mechanism
How to distinguish between nucleophilic
catalysis and general base catalysis?
1. “Common ion effect”
Add anions that are identical to the leaving group in the reaction
(assuming that the pKb of the leaving group is such that the
group effectively acts as a base) and determine the reaction rate:
- faster: general base catalysis, since addition of the leaving
group increases the concentration of base in solution and
n = S[Bi][S].
- slower: nucleophilic catalysis, addition of extra leaving group
drives the reaction equilibria back from product to the
covalent intermediate.
Example:
O
H3C
F
O
F
H2O
H3C
OH
+ HF
General base catalysis would involve an intermediate like:
OH
H3C
F
O
H F
Mechanism of nucleophilic catalysis:
O
H3C
O
O
F
F
H3C
F
F
H3C
F
+ F
A rate enhancement was found upon addition of F-
general base catalysis
2. Detection of a covalent intermediate is a proof for nucleophilic
catalysis. The existence of the intermediate can be proven by:
- isolation
- spectroscopic detection:
O2N
O
CH 3
+ N
H3C
NH
N
O
NH
+ O2N
O
detectable
by UV
H2O
OH + N
NH + O2N
O
H3C
O
OH
- trapping, i.e. the in situ modification of the intermediate by a
“trapping agent” that is deliberately added to the reaction mixture.
With caution, the failure to detect an intermediate can be used as
a proof for the occurrence of general base catalysis, e.g.:
Cl
H2O
O
Cl
CH 3
O
OH
O
CH 3
O
H
general
base
catalysis
Cl
O
+ CH3-COOH
H3C COO
CH3-COO
nucleophilic catalysis
CH 3
H2O
2 CH3COOH
O
Cl
O
+
O
O
CH 3
O
Ph-NH2
"trapping"
N
H
+ CH3-COOH
CH 3
not detected
3. Nonlinearity of the Brønsted plot:
o
In general base catalysis there is a
good correlation, data points (x) are
on a straight line; in nucleophilic
catalysis there are sometimes strong
deviations (o).
x
o
x
log k
o
x
x
o
pKa (cat.)
Reasons for deviations in the Brønsted plot:
a) A difference in polarisability at the same pKa.
Substrate
catalysis
kim/phosphate
(~same pKa)
ethyl acetate
ethyl dichloroacetate
p-nitrophenyl acetate
acetic anhydride
0.25
1.9
4700
860
kOH-/im
type of
(~same nucleophilicity)
910 000
650 000
16
7.2
general base catalysis
general base catalysis
nucleophilic catalysis
nucleophilic catalysis
b) Steric hindrance
Not important for base catalysis (H-transfer), but very important
in nucleophilic catalysis, e.g.:
CH3
O
O
O
O
CH3
+
+ H3C COO
N
H2O
+ 2 CH3COOH
CH3
N
N
This reaction is not catalysed by sterically hindered bases like:
CH3
N
CH3
2-picoline
c) The a-effect.
H3C
N
CH3
2,6-lutidine
H3C
N
CH3
2,4,6-collidine
4. Determine the solvent isotope effect (H2O vs. D2O):
The rate determining step in general base catalysis = cleavage
of a O-H (O-D) bond, which is not the case in nucleophilic
catalysis. E.g.:
Substrate
ethyl dichloroacetate
p-nitrophenyl acetate
kH/D
3
1
type of catalysis
general base catalysis
nucleophilic catalysis
N.B.: the isotope effect can be obscured by solvation effects!
6. Metal Ion Catalysis
A metal such as Cu2+ or Zn2+ can also
stabilize the TS.
The metal must be able to be bound to the
charged intermediate and hence the TS.
6. Metal Ion Catalysis
The tetrahedral oxyanion intermediate of the
reaction of an electrophilic carbonyl C can
interact with a metal if there is an O on an
adjacent atom which can help coordinate the
metal ion. T
His charge stabilization of the developing
negative in the TS and the full negative in the
intermediate is often called electrostatic
catalysis.
6. Metal Ion Catalysis
This method is likely to be found in many
enzymes since nearly 1/3 of all enzymes
require metal ions.
A classic example of an enzyme using
metal ion catalysis is carboxypeptidase A.
Metal ion catalysis
Roles of metals in catalysis:
1. As “super acid”: comparable to H+ but stronger
2. As template: metal ions are able to coordinate to more than 2
ligands and can thereby bring molecules together
3. As redox catalyst: many metal ions can accept or donate
electrons by changing their redox state
Super acid catalysis
Features:
Introduces positive charge into the substrate, making it more
susceptible toward nucleophilic attack.
Exchange of metal ions is fast (105-109 s-1), but slower than
exchange of H+ (1011 s-1)
Metal ion catalysis in C-C bond cleavage
Decarboxylation of oxalosuccinate by isocitrate dehydrogenase:
O
O
O
O
- CO2
a
O
b
O
g
Mn2+ enzyme
O
H+
a
a
b
O
O
O
O
O
g
O
b
O
Mn2+ enzyme
O
g
O
O
Mn2+ enzyme
Mn2+ is very well able to accept the developing negative charge
(“electron sink”); M3+ like Al3+ are also good, M+ like Na+, K+
(and H+!) are much less effective.
2+
2+ O
O
Other acceptable substrates:
Mn O
Mn
O
- both COO- and C=O are needed for
correct binding of Mn2+
- cleaving COO- group on b-position
O
COOH
oxaloacetic acid
O
COOH
3-ketoglutaric acid
Metal ion catalysis in additions to C=O(N) bonds
Cu2+ ions are very effective catalysts for the hydrolysis of aamino acid esters:
Cu
2+
H2N
R H
Cu
O
OH
OCH 3
H2N
2+
O
OCH 3
R H OH
H+
- CH3OH
hydrolysis
products
They are less effective in the hydrolysis of amides, because of
a tighter bond between the metal and the substrate (= ground
state stabilisation):
H2
N
O
uncatalysed
Mn+
N
H2
ester
E
amide
catalysed
amino acid
amide
reaction co-ordinate
Metal ion catalysis in the hydrolysis of
phosphate esters and anhydrides
Hydrolysis of phosphate esters (e.g. acetyl phosphate) or anhydrides
(e.g. ATP) is always catalysed by metal ions, usually Mg2+:
Mg
O
H3C
2+
O
P O
O
O
Mg
2+
O
H3C
O
O
O
P
H2O
O
fast
H2PO4 + CH3COO
The role of the metal ion is twofold:
- neutralisation of the negative charge in the substrate, to enable the
the approach of the
Ad
Ad
O
O
nucleophile;
O P O
O P O
- stabilisation of the
H2O
O
O
O
O
O
O
H2PO4
P
P
P
P
leaving group
O
O
HO
O
O
O
O
O
2+
(neutralisation of
Mg
Mg 2+
charge)
ATP
ADP
Metal Ion Catalysis
(Stabilization of TS)
Binding Energy of ES
Competing effects determine the position of ES
on the energy scale
Try to mentally decompose the binding effects
at the active site into favorable and
unfavorable
The binding of S to E must be favorable
But not too favorable!
Km cannot be "too tight" - goal is to make the
energy barrier between ES and EX‡ small
Entropy Loss and
Destabilization of ES
Raising the energy of ES raises the rate
For a given energy of EX‡, raising the
energy of ES will increase the catalyzed rate
This is accomplished by
(a) loss of entropy due to formation of ES
(b) destabilization of ES by
strain
distortion
desolvation
Transition State Analogs
Very tight binding to the active site!
The affinity of the enzyme for the
transition state may be 10 -15 M!
Can we see anything like that with stable
molecules?
Transition state analogs (TSAs) do pretty
well!
Proline racemase was the first case
Mechanism of Ribonuclease A
His (119)
BH
N
N
N
H
RNA
N
H
His (12)
Pyr imidine (U or C)
Pyr imidine (U or C)
O
O
H
H
H
O
H
O
P
H
H
B:
N
H
O
O
O
RNA
N
H
H
O
H
N
H
N
O
BH
O
O
Lys (41)
P
NH3
O
O
O
H
H
Pyr or Pur
H
OH
Pyr or Pur
O
O
H
H
O
H
H
H 2O
RNA
H
OH
Free 5'-Hydroxy group
Divalent TS stabilized by Lys-41
O
OH
H
H
O
OH
H
RNA
H
N
B:
Pyr or Pur
H
N
2,3’-cyclic phosphate
Pyr imidine (U or C)
RNA
O
O
H
H
O
H
H
H
O
H
H
N
O
O
N
P
BH
O
2', 3'-cyclic phosphate
His (119)
Pyr imidine (U or C)
RNA
O
O
H
N
N
H
His (12)
OH
H
N
H
H
N
H
O
Pyr imidine (U or C)
O
P
N
O
RNA
O
O
OH
N
H
H
H
H
H
B:
O
N
O
BH
O
Lys (41)
P
NH3
O
Divalent TS stabilized by Lys-41
H
N
OH
Mechanism of Acetoacetate
Decarboxylase
Lys
H2N:
BH+
BH+
O
Enz
H
H3 C C C COO
H
Acetoacetate
HO
Lys
O
H
H3 C C C C O
H
N
H
H2O Imine (Schiff base)
H
H3 C C C COO
H
..
N
H
Carbinolamine
Lys
CO2
.. BH+
NH
Lys
H3 C C
CH2
Enzyme-covalently bound
Lys
NH
H3 C C
O
CH3
Imine (Schiff base)
H 2O
H3 C C CH3
Acetone
Lys
Enz
H2 N
Mechanism of Enolase
Rxn
O
O
C
H
HO
C
CH2
O
O
O
O
C
Enolase
P
O
C
O
O
O
P
O
+ H 2O
O
CH2
Phosphoenolpyruvate (PEP)
2-Phosphoglycerate (2-PG)
Lys
345
NH2
Mg2+
Mg2+
O
O
O
OH
C
H
HO
345
Glu-211
C
O
C
O
P
O
O
CH2
Lys
NH3
Mg2+
Mg2+
Glu-211
O O
O
O P
C
C
O
O
O
O H
C
H2 C
OH
O
O
C
C
O
O
P
O
345
Lys
NH3
O
Glu-211
C
O
O
+ H2 O
CH2
Phosphoenolpyruvate (PEP)
Mechanism of Carboxypeptidase A
His-196 Glu-72
His-69
O
Glu-270
C
Tyr -248
Zn2+
O
O H H
H
H3N
HN
R-C-N-C- COO
H R'
O
Arg- 145
O
C
O
O
H
Zn2+
O
O H H
H
H3N
HN
R-C-N-C- COO
H
O
H R'
H
Terahedral
H
O
H2O
R-C-OH
NH2-R'
H
O
C
O
H
Zn2+
O
O
H
R-C
H3N
HN
O
H
O
H
O
C
O
Zn2+
O
O
H
R-C
O H
O
H
H
H3N
HN
Mechanism of Carboxypeptidase A
Zn2+ is acting as a Lewis acid
It coordinates to the non-bonding
electrons of carbonyl group
Including charge separation and making
the carbon more electrophilic or
More susceptible to nucleophilic attack
Mechanism of Carbonic Anhydrase
His
His
His
His
His
His
Zn2+
Zn2+
CO2
B:
O
B:
O
H
O
H
O
C
His
His
His
Zn2+
B:
O
H
C
O
O
H 2O
His
His
His
Zn2+
B:
O
O
H
H
C
O
H
O
O
His
His
HO C O
Bicarbonate
His
Zn
Zn2+
O
H
His
His
BH
His
2+
BH
O
H
O
Tetraherdal
O
C
H
O
Mechanism of Carbonic Anhydrase
Zn2+ function to make potential
nucleophiles (such as water) more
nucleophilic group.
For example, the pKa of water drop from
15.7 to 6-7 when it is coordinate to Zn2+
OH- is 4 orders of magnitude more
nucleophilic than is water
Mult-Substrate Enzyme
Mechanism
In reality, many enzymes have more than one
substrate (A, B) and more than one product (P,
Q).
For example, the enzyme alcohol
dehydrogenase catalyzes the oxidation of
ethanol with NAD (a biological oxidizing agent)
to form acetaldehyde and NADH.
How do you do enzymes kinetics on these more
complicated systems?
Mult-Substrate Enzyme
Mechanism
The answer is fairly straightforward. You keep
one of the substrates (B) fixed, and vary the
other substrate (A) and obtain a series of
hyperbolic plots of v vs A at different fixed B
concentrations.
This would give a series of linear 1/v vs 1/A
double-reciprocal plots (Lineweaver-Burk plots)
as well. The pattern of Lineweaver-Burk plots
depends on how the reactants and products
interact with the enzyme.
1. Sequential Mechanism:
In this mechanism, both substrates must
bind to the enzyme before any products
are made and released.
The substrates might bind to the enzyme
in a random fashion (A first then B or viceversa) or in an ordered fashion (A first
followed by B).
Sequential Mechanism:
An abbreviated notation scheme is shown
below for the sequential random and
sequential ordered mechanisms.
For both mechanisms, Lineweaver-Burk
plots at varying A and different fixed values
of B give a series of intersecting lines.
Sequential Mechanism:
Bi-substrate Enzyme Kinetics
Sequential
B
A
1. ordered
2. random
E
EA
P
EAB
Ping-pong
P
A
E
EPQ
EA
E*P
Q
EQ
B
E*
E
Q
E*B
EQ
E
Equations for Bi-substrate Kinetics
Ping Pong Mechanism
[B]
1/v
v=
Vmax[A][B]
Ka[B] + Kb[A] + [A][B]
1/[A]
Sequential Mechanism
v=
Vmax[A][B]
[B]
1/v
[A][B] + Ka[B] + Kb[A] + KaKb
1/[A]
Secondary plot (Replot)
Ping Pong Mechanism
Intercept
Ka/V
Slope
Kb/V
1/V
1/[B]
-1/Kb
1/[B]
Sequential Mechanism
Intercept
Slope
Kb/V
KiaKb/V
Ka/V
1/V
1/[B]
-1/Kb
1/[B]
Sequential Kinetics
• Sequential kinetics can be
distinguished from ping-pong
kinetics by initial rate studies.
• In practice, measure initial
rates as a function of the
concentration of one substrate
while holding the concentration
of the second constant. Next,
vary the concentration of the
second substrate and repeat.
Sequential Kinetics
• Lineweaver-Burk (doublereciprocal) analysis should
yield a family of lines that
intersect at the left of the yaxis of the graph.
• Within the realm of sequential
reactions lies ordered
sequential and random
sequential at the extreme
ends.
• The equations for the two are
identical; therefore, simple
initial rate studies cannot
differentiate between the two.
Sequential Kinetics
• In ordered sequential
reactions, one substrate is
obligated to bind to the
enzyme before a second
substrate.
• In random sequential
mechanisms there is no
preference. In practice,
there is usually some
degree of order in binding.
Adenylate Kinase Kinetic Pathway
Adenylate kinase displays a random ordered kinetic mechanism. In this case, the two
substrates are bound randomly, and are in equilibrium with the “ternary complex”
(E•MgATP•AMP). As in our derivation, this necessitates that the off rate for each of the
substrates is less than the forward rate constant for the chemical step. This allows us to
replace Km with Ks. However, it would not be incorrect to use Km values. Below is
typical shorthand notation for kinetic schemes.
MgATP
AM P
KsM gA TP
AD P
Ks'A MP
Ks'ADP
E
E • MgATP • AM P
E
E • MgADP • ADP
E • AMP
AM P
KsM gA DP
E • MgADP
E • MgATP
KsAMP
MgAD P
E • ADP
Ks'M gAT P
MgATP
Ks'M gADP
MgAD P
KsADP
AD P
Random
S1
E
S2
E•S1 S2
E•S1•S2
E + P1 + P2
E•S2 S1
e.g. hexokinase (E) catalyzed phosphorylation of
glucose (S1) by ATP (S2)
Ordered
S2
E
E•S1
E•S1•S2
E + P1 + P2
e.g. oxidation reactions involving nicotinamide
adenine dinucleotide coenzyme
2. Ping Pong Mechanism
In this mechanism, one substrate bind first to the
enzyme followed by product P release. Typically,
product P is a fragment of the original substrate
A. The rest of the substrate is covalently
attached to the enzyme E, which we now
designate as E'.
Now the second reactant, B, binds and reacts
with the enzyme to form a covalent adduct with
the covalent fragment of A still attached to the
enzyme to form product Q.
Ping Pong Mechanism
This is now released and the enzyme is restored
to its initial form, E. This mechanism is term
ping-pong.
An abbreviated notation scheme is shown
below for the ping-pong mechanisms. For this
mechanisms, Lineweaver-Burk plots at varying A
and different fixed values of B give a series of
parallel lines.
Ping Pong Mechanism
Water (B) then comes in and covalently attacks
the enzyme, forming an adduct with the
phosphate which is covalently bound to the
enzyme, releasing it as inorganic phosphate.
In this particular example, however, you can't
vary the water concentration and it would be
impossible to generate the parallel LineweaverBurk plots characteristic of ping-pong kinetics.
Ping Pong Mechanism
Ping Pong Mechanism
P1
E
E•S1
S2
E •S2
E*
P2
e.g. Cleavage of polypeptide chain by serine
protease
H2O
OH
+ H
E
Ping-Pong Reaction
O
H OH
H O
HO
HO
OH
HO
H
H
OH
O
H
H O
HN
O
P
O
O
O
O
P
H
HO
N
H
H
O
H
OH
O
O
O
H
O
P
O
H
H
OH
H
OH
O
galactose 1-phosphate
UDP -Glucose
HO
O
OH
H O
H
HO
H
H
H
OH
O
H OH
HN
O
P
O
O
O
O
P
O
N
O
O
H
UDP -Galactose
H O
HO
HO
H
H
H
H
OH
H
OH
H
OH
O
O
P
O
O
glucose 1-phosphate
Galactose-1-P
Uridylytransferase
O
H OH
H O
HO
HO
H
H
OH
O
H
O
P
O
O
O
O
P
O
H O
N
HO
HO
Ping
O
O
H
E
H OH
HN
His
H
H
OH
O
H
H
H
OH
H
OH
O
P
O
O
glucose 1-phosphate
O
UDP-Glucose
HN
+
HO
H O
H
HO
H
H
E
O
OH
HN
H
OH
O
O
P
O
O
O
O
P
O
HO
N
O
O
H
H
UDP-Galactose
OH
H
H
OH
O
O
Pong
His P
O
H
H O
H
HO
H
H
O
O
OH
H
OH
O
O
P
O
O
galactose 1-phosphate
N
H
H
OH
H
OH
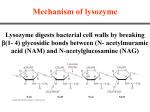
Lysozyme
Lysozyme hydrolyzes polysaccharide
chains and ruptures certain bacterial cells
by breaking down the cell wall
Hen egg white enzyme has 129 residues
with four disulfide bonds
The first enzyme whose structure was
solved by X-ray crystallography (by David
Phillips in 1965)
Substrate Analog Studies
Natural substrates are not stable in the
active site for structural studies
But analogs can be used - like (NAG)3
Fitting a NAG into the D site requires a
distortion of the sugar
This argues for stabilization of a transition
state via destabilization (distortion and
strain) of the substrate
The Lysozyme Mechanism
Studies with 18O-enriched water show that
the C1-O bond is cleaved on the substrate
between the D and E sites
This incorporates 18O into C1
Glu35 acts as a general acid
Asp52 stabilizes a carbonium ion
intermediate
Mechanism of Lysozyme
O
(52) Asp
O
CH2OH
O
O
O
H
OR
H
OH
H
H
H
O
CH2OH
O
H
HO
NHCOCH3
O
Site E
H
H
H
H
NHCOCH3
General Acid
18
CH3
R = H (NAG) or
Glu (35)
O
Site D
H2 O
CH2OH
CH (NAM)
H
O
H
OH
COO
O
H
HO
H
H
NHCOCH3
O
(52) Asp
Asp-52 acting to
stabilize positively
charged intermediate
at TS
Glu (35)
O
O
O
CH2OH
O
H
H
OR
O
H
H
O
H
General Base
H 18 H
NHCOCH3
18
CH2OH
O
H
O
H
OR
H
(52) Asp
H
NHCOCH3
O
Glu (35)
O
O
HO
OH
H