* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Primary Structure Determination (Sanger)
Survey
Document related concepts
Protein folding wikipedia , lookup
Homology modeling wikipedia , lookup
List of types of proteins wikipedia , lookup
Circular dichroism wikipedia , lookup
Intrinsically disordered proteins wikipedia , lookup
Protein mass spectrometry wikipedia , lookup
Transcript
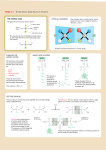
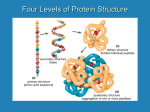
Primary Structure Determination (Sanger) The primary structure is the amino acid sequence plus any disulfide links. • 1. Determine what amino acids are present and their molar ratios. • 2. Cleave the peptide into smaller fragments, and determine the amino acid composition of these smaller fragments. • 3. Identify the N-terminus and C-terminus in the parent peptide and in each fragment. • 4. Organize the information so that the sequences of small fragments can be overlapped to reveal the full sequence. Amino Acid Analysis • Acid-hydrolysis of the peptide (6 M HCl, 24 hr) gives a mixture of amino acids. • The mixture is separated by ion-exchange chromatography, which depends on the differences in pI among the various amino acids. • Amino acids are detected using ninhydrin. • Automated method; requires only 10-5 to 10-7 g of peptide. Partial Hydrolysis of Peptides and Proteins • Cleaving some, but not all, of the peptide bonds gives smaller fragments. These smaller fragments are then separated and the amino acids present in each fragment determined. • Enzyme-catalyzed cleavage is the preferred method for partial hydrolysis. • The enzymes that catalyze the hydrolysis of peptide bonds are called peptidases, proteases, or proteolytic enzymes. Trypsin Trypsin is selective for cleaving the peptide bond to the carboxyl group of lysine or arginine. O O O NHCHC NHCHC R R' lysine or arginine NHCHC R" Chymotrypsin Chymotrypsin is selective for cleaving the peptide bond to the carboxyl group of amino acids with an aromatic side chain. O O O NHCHC NHCHC R R' phenylalanine, tyrosine, tryptophan NHCHC R" Carboxypeptidase Carboxypeptidase is selective for cleaving the peptide bond to the C-terminal amino acid. O O + H3NCHC R O – protein C NHCHCO R End Group Analysis • Amino sequence is ambiguous unless we know whether to read it left-to-right or right-to-left. • We need to know what the N-terminal and Cterminal amino acids are. • The C-terminal amino acid can be determined by carboxypeptidase-catalyzed hydrolysis. • Several chemical methods have been developed for identifying the N-terminus. They depend on the fact that the amino N at the terminus is more nucleophilic than any of the amide nitrogens. Sanger's Method • The key reagent in Sanger's method for identifying the N-terminus is 1-fluoro-2,4dinitrobenzene. • 1-Fluoro-2,4-dinitrobenzene is very reactive toward nucleophilic aromatic substitution NO2 O2N F Sanger's Method • 1-Fluoro-2,4-dinitrobenzene reacts with the amino nitrogen of the N-terminal amino acid. NO2 O2N O O F + H2NCHC NHCHC NHCH2C O2N O NHCHC O NHCHC O NHCH2C CH(CH3)2 CH2C6H5 – NHCHCO CH3 CH(CH3)2 CH2C6H5 NO2 O O O – NHCHCO CH3 Sanger's Method • Acid hydrolysis cleaves all of the peptide bonds leaving a mixture of amino acids, only one of which (the N-terminus) bears a 2,4-DNP group. NO2 O O O O + + + NHCHCOH + H3NCHCO– + H3NCH2CO– + H3NCHCO– O2N CH(CH3)2 CH3 CH2C6H5 H3O+ NO2 O2N O NHCHC O NHCHC O NHCH2C CH(CH3)2 CH2C6H5 O – NHCHCO CH3 B Chain of Bovine Insulin FVNQHLCGSHL SHLV LVGA VGAL ALY YLVC VCGERGF GFFYTPK YTPKA FVNQHLCGSHLVGALYLVCGERGFFYTPKA Edman Degradation • 1. Method for determining Nterminal amino acid. • 2. Can be done sequentially one residue at a time on the same sample. Usually one can determine the first 20 or so amino acids from the N-terminus by this method. • 3. 10-10 g of sample is sufficient. 4. Has been automated. N C 5. Uses phenyl isothiocyanate. S Edman Degradation + O C6H5N C + H3NCHC S peptide NH R • Phenyl isothiocyanate reacts with the amino nitrogen of the N-terminal amino acid. S O C6H5NHCNHCHC NH R peptide Edman Degradation S O C6H5NHCNHCHC peptide NH R HCl S C6H5NH C O C + + N CH R H3N peptide Peptide Bond Formation • Random peptide bond formation in a mixture of phenylalanine and glycine, for example, will give: Phe—Phe Gly—Gly Phe—Gly Gly—Phe Limit the number of possibilities by "protecting" the nitrogen of one amino acid and the carboxyl group of the other. N-Protected phenylalanine C-Protected glycine O X NHCHCOH O H2NCH2C CH2C6H5 Only Phe- Gly is formed Y Protect Amino Groups as Amides • Amino groups are normally protected by converting them to amides. • Benzyloxycarbonyl (C6H5CH2O—) is a common protecting group. It is abbreviated as Z. • Z-protection is carried out by treating an amino acid with benzyloxycarbonyl chloride. Protect Amino Groups as Amides O O + CH2OCCl + H3NCHCO – CH2C6H5 1. NaOH, H2O 2. H+ O CH2OC O NHCHCOH (82-87%) Z-Phe CH2C6H5 Removing Z-Protection • An advantage of the benzyloxycarbonyl protecting group is that it is easily removed by: • a) hydrogenation (H2/Pd) • b) cleavage with HBr in acetic acid The tert-Butoxycarbonyl Protecting Group O (CH3)3COC O NHCHCOH CH2C6H5 is abbreviated as: O BocNHCHCOH CH2C6H5 or Boc-Phe HBr Cleavage of Boc-Protecting Group O (CH3)3COC O NHCHCNHCH2CO2CH2CH3 CH2C6H5 HBr O H3C C H3C CH2 CO2 + H3NCHCNHCH2CO2CH2CH3 CH2C6H5 Br – (86%) Protect Carboxyl Groups as Esters • Carboxyl groups are normally protected as esters. • Deprotection of methyl and ethyl esters is by hydrolysis in base. • Benzyl esters can be cleaved by hydrogenation. (H2/Pd) Forming Peptide Bonds • The two major methods are: • 1. coupling of suitably protected amino acids using N,N'-dicyclohexylcarbodiimide (DCCI) • 2. via an active ester of the N-terminal amino acid. DCCI-Promoted Coupling O O ZNHCHCOH + H2NCH2COCH2CH3 CH2C6H5 DCCI, chloroform O ZNHCHC CH2C6H5 O NHCH2COCH2CH3 (83%) DCCI-Promoted Coupling • The species formed by addition of the Zprotected amino acid to DCCI is similar in structure to an acid anhydride and acts as an acylating agent. • Attack by the amine function of the carboxylprotected amino acid on the carbonyl group leads to nucleophilic acyl substitution. C6H11N NC6H11 C H + O O C6H11N C OCCHNHZ ZNHCHCOH C6H11N CH2C6H5 CH2C6H5 Mechanism of DCCI-Promoted Coupling O O H C6H11N C O + ZNHCHC NHCH2COCH2CH3 CH2C6H5 C6H11NH O H2NCH2COCH2CH3 H C6H11N C6H11N O C OCCHNHZ CH2C6H5 The Active Ester Method A p-nitrophenyl ester is an example of an "active ester." O O NO2 ZNHCHCO + H2NCH2COCH2CH3 CH2C6H5 chloroform O ZNHCHC CH2C6H5 O NHCH2COCH2CH3 (78%) + HO NO2 Solid-Phase (Merrifield) Synthesis • In solid-phase synthesis, the starting material is bonded to an inert solid support. • Reactants are added in solution. • Reaction occurs at the interface between the solid and the solution. Because the starting material is bonded to the solid, any product from the starting material remains bonded as well. • Purification involves simply washing the byproducts from the solid support. The Solid Support CH2 CH CH2 CH CH2 CH CH2 CH2Cl • The side chain chloromethyl group is a benzylic halide, reactive toward nucleophilic substitution (SN2). CH The Merrifield Procedure CH2 CH CH2 CH CH2 O – BocNHCHCO R CH CH2Cl CH2 CH The Merrifield Procedure CH2 CH CH2 CH CH2 O BocNHCHCO • Next, the Boc protecting group is removed with HCl. R CH CH2 CH2 CH The Merrifield Procedure CH2 CH CH2 CH CH2 O H2NCHCO • DCCI-promoted coupling adds the second amino acid R CH CH2 CH2 CH The Merrifield Procedure CH2 CH CH2 CH CH2 O BocNHCHC R' CH O CH2 CH CH2 NHCHCO R • Remove the Boc protecting group. The Merrifield Procedure CH2 CH CH2 CH CH2 O H2NCHC R' CH O CH2 CH CH2 NHCHCO R • Add the next amino acid and repeat. The Merrifield Procedure CH2 CH CH2 O CH CH2 O + H3N peptide C NHCHC R' CH O CH2 CH CH2 NHCHCO R • Remove the peptide from the resin with HBr in CF3CO2H The Merrifield Procedure CH2 CH CH2 CH CH2 CH CH2Br O O + H3N peptide C NHCHC R' O – NHCHCO R CH2 CH The Merrifield Method • Merrifield automated his solid-phase method. • Synthesized a nonapeptide (bradykinin) in 1962 in 8 days in 68% yield. • Synthesized ribonuclease (124 amino acids) in 1969. 369 reactions; 11,391 steps • Nobel Prize in chemistry: 1984 Levels of Protein Structure • Primary structure = the amino acid sequence plus disulfide links • Secondary structure = conformational relationship between nearest neighbor amino acids • • • – pleated sheet – helix planar geometry of peptide bond anti conformation of main chain hydrogen bonds between N—H and O=C Pleated Sheet • • • • Adjacent chains are antiparallel. Hydrogen bonds between chains. small side chains Sheet is flexible, but resists stretching. Helix • Shown is an helix of a protein in which all of the amino acids are L-alanine. • Helix is right-handed with 3.6 amino acids per turn. • Hydrogen bonds are within a single chain. Tertiary Structure • Refers to overall shape (how the chain is folded) • Fibrous proteins (hair, tendons, wool) have elongated shapes • Globular proteins are approximately spherical • most enzymes are globular proteins • an example is carboxypeptidase Protein Quaternary Structure • Some proteins are assemblies of two or more chains. The way in which these chains are organized is called the quaternary structure. • Hemoglobin, for example, consists of 4 subunits. • There are 2 chains (identical) and 2 chains (also identical). • Each subunit contains one heme and each protein is about the size of myoglobin.