* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download A Road Map for HCV Genotyping
Drug design wikipedia , lookup
Exome sequencing wikipedia , lookup
Pharmacometabolomics wikipedia , lookup
Plant virus wikipedia , lookup
Bisulfite sequencing wikipedia , lookup
Molecular Inversion Probe wikipedia , lookup
Pharmacogenomics wikipedia , lookup
SNP genotyping wikipedia , lookup
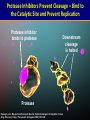
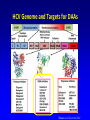
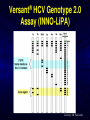
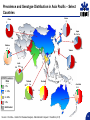
A ROAD MAP FOR HCV GENOTYPING: HOW DO WE GET THERE – AND WHAT WILL WE FIND? Scott Bowden Molecular Microbiology VIDRL Adjunct Professor Dept of Microbiology Monash University HCV Genotyping 1. How we get there – evolution of genotypes 2. What do we find – genotyping technology First Methods of Viral Characterization were Based on Serotyping & Phenotyping • Virus characterization has undergone continual refinement as a consequence of the development of new technologies • Initial methods based on phenotyping growth in eggs, cell culture, CPE etc. • Polyclonal and monoclonal antibodies could serotype isolates which grew in culture Serotyping • 1970s - some classes of viruses could be identified using specific antibodies capable of neutralizing their infectivity. • Serotyping, raised to type-specific antigens, has been an important tool for classifying viruses – poliovirus (types 1, 2 and 3) – hepatitis B virus (adw, adr, ayw and ayr) – dengue virus (types 1, 2, 3 and 4) These methods not suitable for all viruses • The dawn of the molecular biology era allowed viruses to be classified genotypically • Initially using cloning & nucleic acid hybridization technology and more recently with the aid of amplification technology, such as PCR followed by DNA sequence analysis • Next Gen Sequencing not yet widely used • Requires some form of sequence interrogation Definition of Viral Genotype • Genetic sequences within species are subdivided into one or more groups. These classifications are commonly termed genotypes or clades • There can be further subdivisions - subgenotypes • The degree of variation seen between these classifications is species specific and no standardisation in nomenclature exists between viral species • An important job for the ICTV!! Viral Genotypes • HIV groups subdivided into clades which share around 30% nucleotide identity within the envelope region and viruses within a clade vary by around 15%. • HCV genetic diversity is classified on three levels. – level 1 defines genotypes which differ by >35 % in nucleotide sequence over the whole 9.5kb genome (Genotype 1-7) – level 2 divides genotypes into subtypes whose nucleotide sequence differs by >15% (subtypes 1a, b, c, etc) – level 3 defines isolates, within a subtype, which differ by <13%. Hepatitis C Virus Genotypes: World View 1a,1b,2b,3a 1a,3a 1b,1a,2b,3a 4,1a,1b 4 4 4 1,2 4 1b,1a,3a 5a,1b,1a,2 3a 2a 1b 1b,2a,1a 1b,6a 6,6c-6l,1b,2 3a,6a-6b 3k,6g 3a,1a,1b,2 1a,1b,3a HCV Divergence • Adaptive change – selection pressure – Immunoselection – Antiviral therapy > Change of phenotype • Neutral sequence drift – Changes which have little or no effect on fitness – Accumulate and become fixed in the viral population – In geographically distinct regions, become genetically diverse – > GENOTYPES > Little or no change of phenotype Simmonds (2004) J Gen Virol 85:3173 Interferon Therapy for HCV HCV Can Modify the Innate Immune System Evolution of HCV Genotypes • Hypothesis: Immune selection has been a driving force in divergence of HCV genotypes Viral factors inhibiting the immune response have played a role in the genotype-specific response to interferon Genotype 2 older than genotype 3 > 4 > 1 Pang et al 2009 PLoS ONE 4: e6579 Will Genotype be Important in the Era of Direct-Acting Antivirals? Shinazi et al Liver Int 2014 Positive recommendation - ledipasvir/sofosbuvir - sofosbuvir - daclatasvir - genotype 1-6 - all patients (no comment about F stage) - general section rather than S100 - peginterferon containing treatment is not cost-effective Resistance Associated Variants to DAAs 1. HCV exists as a swarm of related variants 2. HCV with amino acid substitutions known to confer resistance pre-exist even in the absence of drug exposure 3. Naturally occurring polymorphisms may encode resistance 4. Minor variants can also be selected and amplified by antiviral therapy Selection of a minor quasispecies Amplification by selection pressure Mode of Action of New HCV DAAs 1. NS3/4 Protease inhibitors 2. NS5A inhibitors 3. NS5B non-nucleoside inhibitors 4. NS5B nucleoside inhibitors 1. NS3/4A Protease – Essential for Initiating Replication Cleavage P7 NS2 NS3/4A Protease NS4B E2 NS5A E1 C Kwong A, et al. Beyond interferon and ribavirin: Antiviral therapies for hepatitis C virus. Drug Discovery Today: Therapeutic Strategies. 2006;3:211-220. NS5B Protease Inhibitors Prevent Cleavage – Bind to the Catalytic Site and Prevent Replication Protease inhibitor binds to protease Downstream cleavage is halted Protease Kwong A, et al. Beyond interferon and ribavirin: Antiviral therapies for hepatitis C virus. Drug Discovery Today: Therapeutic Strategies. 2006;3:211-220. Protease Inhibitor Resistance • Binding site includes R155 (arginine at amino acid position 155 of the protease), A156 & D168 • Thus changes at these positions influence efficacy • Some genotype differences – R155 with HCV 1a (R155K = nt AGG to AAG - for 1b = nt CGG to AAG); A156 with HCV 1b • Simeprevir efficacy reduced by Q80K, a naturally occurring polymorphism (HCV genotype 1a) – In Australia, HCV 1a makes up approx 35% – Q80K frequency is 5-10% HCV Genome and Targets for DAAs Shinazi et al Liver Int 2014 2. HCV NS5A Inhibitors Daclatisvir (BMS); Ledipasvir (Gilead); Ombitasvir (AbbVie) • NS5A is a phosphoprotein involved in replication and host-cell interactions • Mechanism of inhibition unclear (SVR 1b> 1a) • Resistance associated variants map to domain I (of three domains) of the protein – L31 and Y93 • These changes cause cross-resistance among other NS5A inhibitors • Naturally occurring polymorphisms associated with resistance occur in HCV genotype 1b Genotype/Subtype will Remain Important in the Era of Current DAAs Progress of an Online Genotypic Drug Resistance Testing and Monitoring Program for Chronic Hepatitis C Lilly Yuen 1, Francesco Moriconi 2, Michael Beard 3, Scott Bowden 1 and Stephen Locarnini 1 1 2 3 VIDRL, North Melbourne, VIC 3051; Hepatology Unit, University Hospital of Pisa, Italy; IMVS & The University of Adelaide, Adelaide, SA 5000. Aim To develop a platform (SeqHepC) for detecting and measuring antiviral drug resistance to DAA compounds in patients Main Goals • Aid in individualized patient management • Identification of risk factors associated with drug resistance • Identification of novel clinically significant drug associated resistance amino acid substitutions that may emerge in chronic patients on DAA therapies Sequence Analysis Unrooted tree of 228 Full length, Unique, Confirmed and Naïve HCV genomes 4a 5a 1b 6 1c 100 100 100 100 Similar results were obtained from unrooted trees of the HCV regions coding for the DAAs targets: 100 100 1a 100 6a 100 100 100 •NS3 1866nt •NS5A 1530nt •NS5B 1782nt 100 3 100 100 3a 100 100 7a 2a Final Reference Set (n = 32) Input Sequence Name NC004102_H77 (1a) EF407429_7043 (1a) EF407417_4040 (1a) AJ238799_Con1 (1b) D50483_HCVK1S (1b) EU781828_TN28 (1b) AY051292 (1c) AF177036_pJ6CF (2a) AB047639_JFH1 (2a) AY232742_MD2b71 (2b) AB559564 (2b) D50409_BEBE1 (2c) DQ155561_D54 (2i) AB031663_VAT96 (2k) D28917_K3A (3a) GQ275355 (3a) D49374_HCVTr (3b) D63821_JK049 (3k) DQ516084_25 (4a) DQ988075_Eg4 (4a) AF064490_SA1 (5a) DQ480522_6a72 (6a) EU246930_D9 (6a) D84262 (6b) D84263 (6d) c0046_HCV (6f) HK6554_HCV (6g) D84265 (6h) KM41_HCV (6k) EU408330_DH012 (6u) EU798761_KM046 (6v) EF108306_QC69 (7a) Length 9646 9086 9383 9605 9410 9359 9441 9711 9678 9406 9511 9513 9370 9488 9454 9442 9444 9450 9309 9057 9386 9358 9376 9628 9615 9454 9462 9621 9430 9451 9429 9357 HCV full length sequences Report Screen HCV Genotyping 1. How we get there – evolution of genotypes 2. What do we find – genotyping technology Methods to Determine Genotype • • • • • Direct sequencing post PCR - 5’ UTR, core, NS5 RFLP post-PCR Type-specific PCR primers Genotype-specific antibodies (NS4) Real-time PCR and melting curve analysis • AUSTRALIA (TGA) • Hybridization assays (LiPA – 5’ UTR, Core) • Abbott RealTime HCV Genotype II Line Probe Assay (LiPA) – Innogenetics > Bayer > Siemens Versant LiPA 2.0 Requires initial amplification by PCR amplicon tested by LiPA Drawbacks - LiPA 1.0 • Relied on 5’ UTR for determination of genotype • Some banding patterns not compatible with the Interpretation Chart • Could not discriminate many HCV genotype 6 from 1 • Inability to clearly subtype some HCV genotype 1 Unusual Band Interpretation Chart www.VIDRL.org.au > Laboratories > Hepatitis Reference Courtesy JM Pawlotsky Advantages - LiPA 1 versus LiPA 2 • LiPA 2 distinguishes HCV genotype 6 c-l from genotype 1b • Also discriminates HCV 1a from HCV 1a/1b (and HCV 1) Abbott RealTime HCV Genotype II • 5’ UTR for Genotyping – NS5B for HCV 1 Subtyping Drawbacks • Some reports of cross-reactivity. True? • Despite NS5B primers, some subtyping of HCV genotype 1is unsuccessful • Difficulties distinguishing HCV genotype 6 Prevalence and Genotype Distribution in Asia Pacific – Select Countries Korea 3 China 2 1b 2a 1b Japan 2b 3a 1a 2a Pakistan 4 1 1b Taiwan 2 2 3 2010 Prevalence Rate < 1% 1b India 3b 4 1a 1b 2 3a 2b Thailand 1a 3b 6 1b 1 - 1.9% 2 - 2.9% > 3% Vietnam Australia 4 6 6 1 3a 2a 2c 3 1 2a 2 Not Studied Source: Kromite – Center for Disease Analysis, International Conquer C Coalition (I-C3) 39 Abbott RealTime HCV Genotype II versus VERSANT LiPA 2.0 Method Abbott RealTime HVC GT II Versant HCV Genotyping 2.0 assay (LiPA) Genotyping technology Real-time PCR Line Probe Assay: End Point PCR followed by reverse hybridization Total turnaround time - 5 hours for 24 samples - 10 hours for 20 samples Result interpretation Genotyping result generated Subjective interpretation of automatically by the software results Sample identification Samples barcoded for traceability Manual labelling of reactions and strips Internal control in sample to Internal controls of samples Internal controls only for PCR monitor extraction efficiency from extraction to PCR, and steps; Control bands on all and possible inhibition external controls to monitor strips, on external controls. the run. HCV subtypes 1a, 1b Most subtypes LL of D 500 IU/ml ∼1,000 IU/ml Liu et al JCM 2015 53: 1754 Abbott HCV Genotype Plus (RUO) • Designed for detection of HCV 1a,1b & 6 only • Can be used as a reflex assay after obtaining results from HCV Genotype II assay (same RNA) • Or used directly for detection of HCV 1a,1b & 6 HCV Genotype Assay Comparison – 1 Routine Genotype Samples SAMPLE ID VERSANT-LiPA 2.0 Abbott RealTime II 15549869 1a 1a 15513934 1a 1a 15549832 1a 1a 15545272 1a 1a 15548755 1a & 3a 1a Reactivity with 3 15554931 1b 1b 15549558 2b 2 15548113 3a 3 15524732 3b 3 15544270 3a 3 15546297 3a 3 15552064 4 4 High concordance between assays HCV GT Plus RUO 1a; 1b HCV Genotype Assay Comparison – 2 LiPA Problematic Samples SAMPLE ID VERSANT-LiPA 2.0 CORE SEQUENCING Abbott RealTime II HCV GT Plus RUO 15513206 346N 6f 1 6 15546665 34N no product 1a 15546667 band 24 no product 1a 15547928 345A 1a 1a 15548329 band 25 1a 1a 15548332 345N 1a 1a 15508997 346A 1a 1a 15517954 345A 1a 1a 15550297 345A 1a 1a 15550456 band 24 unsuccessful 1a 15553443 3 4 (5) 6 N to be done 1a 15501602 no band 1a 1b 15555305 no band to be done 1b 15531224 band 9 2a 2 15544256 no band no product 3 15545261 band 24 3a 3 The Abbott HCV Genotype II can resolve LiPA problematic genotyping HCV Genotype Assay Comparison – 3 HCV Genotype 6 c-l Samples LAB NO 14541062 14547094 14547624 14548133 14551922 14552770 14553472 14556716 14561666 14571514 14580039 14579962 14582079 14586009 14588162 14588373 14596632 14605594 15502180 15524079 15524727 LIPA 6C-L 6C-L 6C-L 6C-L 6C-L 6C-L 6C-L 6C-L 6C-L 6C-L 6C-L 6C-L 6C-L 6C-L 6C-L 6C-L 6C-L 6C-L 6C-L 6C-L 6C-L HCV GT Plus RUO 6 6 6 1a; 6 6 ND 6 6 6 1a;6 6 6 6 6 6 6 6 6 6 6 6 HCV GT Plus can successfully distinguish HCV genotypes 6c-l Conclusions • Our data and previous published comparisons show both LiPA 2.0 and Abbott Gen II have a high concordance rate • Assays complement each other for problematic samples • Shortcomings of the Abbott Gen II assay are resolved with the HCV GT Plus (RUO) • Both LiPA and the Abbott assays offer accurate genotyping – choice will be based on whichever method suits workflow of individual laboratories Acknowledgements • Lilly Tracy • Prof Alexander Thompson (pinched a few slides) • Staff of Molecular Microbiology • Adam & Jennifer - Abbott