* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Solvent Properties of Ground Substance Studied by
Survey
Document related concepts
Transcript
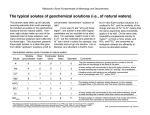
Solvent Properties of Ground Substance Studied by Cryomicrodissection and Intracellular Reference-Phase Techniques SAMUEL B. HOROWITZ and DAVID S. MILLER Laboratories of Cellular Physiology and Metabolic Control, Department of Physiology and Biophysics, Michigan Cancer Foundation, Detroit Michigan 48201 Water, sodium, potassium, ATP, amino acids, and sugars are not uniformly distributed in Rana pipiens oocytes . Concentration differences exist between nucleus (germinal vesicle) and ooplasm and between animal and vegetal ooplasmic regions . The mechanisms responsible for these differences were investigated using intracellular reference-phase (iRP) analysis . The iRP is an artificial "organelle" that has the solvent properties of a dilute salt solution and is in diffusional equilibrium with water and solutes present in other cellular compartments . Ooplasm/iRP solute distributions show that ooplasm differs from ordinary aqueous solutions-exhibiting both solute exclusion and solute binding . Yolk platelets are an important cause of this behavior, largely because their proteins are present as hydrate crystals, which are rich in anionic sites and which interact intensely with associated water. Because of yolk's abundance, it obscures the solvent and binding properties of ooplasmic ground substance . The oocyte nucleus is yolk and organelle free and the nuclear envelope is readily permeable . Consequently, nucleus/iRP solute concentration differences reflect the binding and solvent properties of nuclear ground substance . Nucleoplasm binds -19 meq of potassium. Furthermore, the monosaccharides, 3-O-methylglucose, L-glucose, and o-xylose, are selectively excluded, their nucleus/iRP concentration ratios averaging about 0.7; ratios for other solutes studied are unity . We interpret monosaccharide exclusion to mean that nuclear ground substance water is different in its "instantaneous" structure from ordinary saline water. Because of this difference, hydrogen bond interaction between nuclear water and certain sterically restricted solutes, of which ringed monosaccharides are examples, is reduced . Some implications of modified ground substance water and selective solute exclusion are discussed . ABSTRACT We will need to possess a detailed picture of the ground substance's interactions with water and solutes ifwe are ever to understand its role in metabolism and physiology. This picture will be difficult to obtain, because many important molecules are small (amino acids, nucleotides, sugars, inorganic salts) and diffuse rapidly from normal in vivo locations when the cell's integrity is disturbed . Thus, experimental tools are required that prevent artifactual redistributions and permit water and microsolutes to be measured locally within the cell . An essential first step is to fix the substance in place. Chemical fixation, suitable for macromolecules, does not work because fixative, water, and metabolites interdiffuse at 172s comparable rates. The only reliable approach is cryofixation; specifically, the rapid removal of heat so that diffusion is effectively stopped . A second essential step is the measurement of water and microsolute concentrations without undue loss of spatial resolution . Available techniques include drymount and low-temperature autoradiography (1-4), microprobe analysis (5, 6), nonaqueous fractionation (7), and cryomicrodissection (8-10) . Cryomicrodissection is potentially the most universally applicable of these approaches. In principle, one can freeze any cell and isolate samples for analysis ofwater and any solute. In the future, it may be possible to excavate or mine a frozen cell, removing and cleaning individual organelles and isolating THE JOURNAL OF CELL BIOLOGY " VOLUME 99 NO . 1 PT . 2 JULY 1984 172s-179s ® The Rockefeller University Press - 0021-9525/84/07/0172x/08 $1 .00 FIGURE 1 organelles . An advanced cryomicrodissection project with the excavation and mining of cytoplasmic ground substance and chunks of frozen pure ground substance (Fig. 1). Scaled down methods for measuring the water and solute content of minute bits of material will also be needed. At present, neither the tools nor the personnel are available to fulfill this vision of the future. But a start has been made. Oocyte Cryomicrodissection Fig. 2 diagrams the beginning made on the cryomicrodissection of a giant cell, the amphibian oocyte (10, 11) . The first panel (A) schematizes the full-grown ovarian oocyte as it appears just before the initiation of meiotic maturation . In Rana and Xenopus, the cell is 1,500 um in diameter and has an eccentrically located, -400-um nucleus (germinal vesicle). The extranuclear ooplasm contains the ground cytoplasm and organelles, which exhibit animal-vegetal gradients in distribution . The most prominent organelle is the yolk platelet. These are more concentrated in the vegetal than animal hemisphere, and, with fatty yolk globules, occupy overall about 70% of the ooplasmic volume. Other organelles (e.g., mitochondria, endoplasmic reticulum, ribosomes, and pigment granules) occupy 5-10% of the ooplasm, and the ground substance occupies the remaining 20-25% . The nucleus contains numerous nucleoli and a minute chromosome frame, but the volume fraction these occupy is negligibly small (<I%). For present purposes, the nucleus is considered to consist entirely of ground substance, the most abundant cytoskeletal protein of which is actin (12). For cryomicrodissection, the oocyte is placed on a metal mounting and frozen rapidly to the temperature of liquid nitrogen (Fig. 2 B). Cell and mounting are placed on the lowtemperature (-45°C) stage of a microdissection apparatus (Fig. 2 C). Nucleus and regional samples of ooplasm are isolated by free-hand dissection (Fig. 2 D). After their separation, the frozen samples are placed in tared foil packets. Wet and dry weights of the samples are determined, the contents are extracted, and the solutes are measured by microanalytic techniques . Ovarian oocytes at stages earlier than full-grown can also be cryomicrodissected, though this is not yet routine. The smallest oocytes from which nuclei and cytoplasm have been isolated and analyzed are yolk free and measure -425 um in diameter (13). HOROWITZ AND MILLER Solvent Properties of Ground Substance 173s C Current cryomicrodissection technology . (A) Schematic of a full-grown frog oocyte, which is (B) mounted and frozen on a brass block and (D) partially dissected at cryogenic temperatures using microtools . The microdissection apparatus (C) is an insulated box with glove ports (c), to allow free-hand microdissection of oocytes on a low-temperature stage (a), which is mounted in a Dewar flask (b). Temperature is maintained by a thermostatically regulated flow of nitrogen gas into the Dewar. The work is observed through a dissecting microscope (d). Isolated nuclei and ooplasmic samples are weighed at -20' to -10°C in a compartment (e) mounted below an Electrobalance weighing chamber (f). FIGURE 2 Table I A gives nuclear, animal, and vegetal ooplasmic water concentrations in full-grown oocytes . Note that nuclei contain only - I I % dry material, consistent with the absence of organelles. Ooplasm contains >50% dry material, reflecting the abundance of organelles, chiefly yolk. Because of ooplasm's composite nature, measured concentrations are the weighted average of the solute and solvent content of the many different components. Table I A gives regional cellular contents for six metabolites or model compounds, expressed as concentrations on a water basis . Note first, that no simple rule determines solute distributions between ooplasm and nucleus . For example, al- though sodium is more concentrated in the ooplasm than in the nucleus, the reverse is true for potassium, ATP, sucrose, a-aminoisobutyric acid (AIB)' and 3-O-methylglucose (30MG). Even among these solutes, nucleus/ooplasm concentration ratios vary over a greater than twofold range. No obvious relationship between molecular weight and distribution is seen. Furthermore, because Na and K are cations, AIB is a zwitterion, sucrose and 30MG are nonelectrolytes, and ATP is an anion, nuclear/ooplasmic distributions are not simply a matter of electrical charge, which would be the case if Donnan considerations dominated . The diversity ofsolute distributions seen in Table I A should ' Nucleus/ooplasmic concentration differences do not imply active transport by the nuclear envelope. The effective radius of oocyte nuclear pores is -4.5 nm, so that, although the nuclear envelope can segregate large macromolecules and formed elements (which themselves interact with microsolutes and create asymmetries), it is too porous to play a direct role in establishing asymmetric solute distributions (14, 15). 'Abbreviations used in this paper: AIB, a-aminoisobutyric acid ; HCS, hydrate crystal solute; iRP, intracellular reference phase; 30MG, 3O-methylglucose . I 74s THE JOURNAL OF CELL BIOLOGY " VOLUME 99, 1984 TABLE I Typical Water and Solute Concentrations in Rana pipiens Oocytes Water A B Nucleus (N) Animal ooplasm (AD) Vegetal ooplasm (Vo) Medium 89 ± 1 46 ± 1 41 ± 1 ~100 Na mm 19 ± 4 63 ± 3 98 ± 3 115 .5 K mm 144 ± 3 134 ± 4 105+4 2 .5 ATP Sucrose AIB 30MG 5 .6 ± 0 .9 2 .8 ± 0 .3 1 .4 ± 0 .1 0 0 1 .55 ± 0.16 0 .82 ± 0.04 0 .50 ± 0 .04 1 .0 0 .21 ± 0 .0S 0 .15 ± 0 .03 0.12 ± 0.02 1 .0 mm mm mm 16±2 125±5 6 .3±1 .0 1 .54±0 .08 0.31±0.07 4 .3 ± 0 .3 1 .1 ± 0 .03 0 .S0 ± 0 .0S 0 .44 ± 0 .03 0.S0 ± 0.01 0.S3 ± 0.03 6 .8±0 .6 0 .8±0 .03 0 .33±0 .04 0 .30±0 .02 0.33±0.01 0 .41 ±0 .02 1 .3±0 .3 1 .1 ±0 .03 1 .00±0 .03 1 .00±0 .01 1 .01 ±0 .01 0.80±0.04 <0 .01 NS NS NS NS <0.01 Na and K were measured by atomic-absorption spectroscopy, ATP by luciferin-luciferase and radiolabeled sucrose, and AIB and 30MG by liquid scintillation counting. Sucrose was introduced into oocytes by microinjection and AIB and 30MG by preincubation in Ringer's solution. The 4-h preincubation used here was insufficient for AIB or 30MG transport to reach steady state . At steady state, cell/medium ratios for both solutes are considerably higher . However, intracellular ratios (Ao/iRP, Vo/iRP, and N/iRP) were unchanged, showing they are concentration-independent, equilibrium values (11) . NS, not significant . iRP Ao/iRP Vo/iRP N/iRP P (N vs . iRP) 89±1 - not be surprising. First, there is rich variety of organelles in ooplasm. Some are membrane-bounded, suggesting that they are semipermeable and may engage in active transport; some contain protein crystals, whose water is known to be modified ; and some contain lipid, with hydrophobic solubility properties. Even if organelles were absent, one might expect some of the diversity seen in Table I A because of specific interactions between water, solutes, and the ground substance . As the papers in this supplement attest, the type, concentration, and organization (e.g., polymerization state) of cytomatrix macromolecules vary with cellular region, and each potentially has a different spectrum of interactions with water and solutes. Intracellular Reference-Phase (iRP) Analysis If we lived in the future depicted in Fig. 1 and could mine pure ground substance from a frozen cell, and if we could also thaw the frozen samples without changing their in vivo properties, we would be able to answer directly the questions that interest us here: How do water's interactions with the cytomatrix affect the solvent properties of ground substance water? What solutes bind to the cytomatrix? How do water and solute interactions with the cytomatrix vary with cellular region? Using a dialysis membrane, we would separate the ground substance from an appropriate intracellular "Ringer's" solution, and determine how the distribution ofsolutes in the separate phases changed as the solutes' concentrations were varied. These isotherms would disclose how cytomatrix proteins influence water, what solutes adsorb to the cytomatrix, andwith what specificities and affinities. Present experimental capabilities fall considerably short of this ideal. Two aspects of this shortfall seem especially critical and yet inadequately appreciated : (a) we cannot isolate ground substance and be confident that it has not been modified and (b) we do not know how to formulate an appropriate intracellular saline solution . The iRP providesa partial solutionto these problems (16, 17) . It can be thought of as a technique by which the cell is made to form a localized internal salt solution against which its solutes are dialyzed. In the iRP technique, summarized in Fig. 3, a 13% gelatin solution is injected into ooplasm and then cooled (A). This creates a region ofgel (B; about 2-5 % of cell volume), which, A MICROPIPETTE B GELATIN (gelled) GELATIN (liquid) FIGURE 3 Internal reference phase methodology showing (A) gelatin injection, (B) gelation and diffusion of endogenous solutes into iRP, (C) solute equilibration and freezing, and (D) iRP isolation by cryomicrodissection . due to its fibrous protein network, excludes organelles and the cytoplasmic matrix (not shown) but still allows free entry of salts and metabolites (stippling) . With time (usually 1-4 h), the iRP and cell interior reach diffusional equilibrium (Q. The cell is frozen and the iRP, nucleus, and ooplasmic samples are isolated by cryomicrodissection (D) and analyzed for water and solutes . Fig. 4 shows the typical internal arrangement of iRP, nucleus, and animal and vegetal ooplasm . Comparison of iRP concentrations with those of the "unknown" intracellular phases provides information on how cellular material differs from a gelatin gel . This is important, not only because (for most solutes) the solvent properties of dilute gelatin gels resemble those of ordinary aqueous solutions but also because gelatin gels can be used in conventional dialysis experiments to "refer" intracellular concentrations to those in standard saline solutions. Table I B gives iRP concentrations for water and those solutes for which nuclear and ooplasmic data are given in Table I A. Sodium can serve as a useful starting point for discussing reference-phase analysis. Table I shows that while the Ringer's solution contains 115 .5 mM sodium, the iRP concentration is 16 mM, only 12% as great. Since the solvent properties of HOROWITZ AND MILLER Solvent Properties of Ground Substance 175s Partially cryomicrodissected Rana pipiens oocyte showing a nucleus in situ in the pigmented (darker) animal ooplasm and an iRP embedded in ooplasm near the center of the cell, x 40 . FIGURE 4 Na cm U c 0 U loo U E0 a 0 O 0 I 0 I 100 I 200 300 M IM Reference Phase Concentration FIGURE 5 Animal ooplasm vs . iRP isotherm for sodium . The range of concentrations normally found in healthy oocytes was extended by sodium chloride microinjection . iRP water are indistinguishable from Ringer's solution, the steady-state iRP/Ringer's concentration ratio less than unity demonstrates the role of active, plasma membrane transport in the maintenance of intracellular Na (15-18) . However, the iRP Na concentration (16 mM) is also considerably lower than that of the animal ooplasm (63 mM). Yet, the two lack a separating membrane and are in diffusional equilibrium . The explanation for this difference is complex and is best discussed by referring to the animal ooplasmic isotherm for Na (Fig. 5). If ooplasm and iRP exhibited identical solvent properties, the isotherm would be linear with a slope of unity and an intercept of zero (isomolar line). This is not the case. 176s THE JOURNAL OF CELL BIOLOGY " VOLUME 99, 1984 The isotherm is biphasic, exhibiting curvature (saturation) when iRP Na <40 mM and linearity when iRP Na >40 mM . Thus, Rana ooplasm (like that of Desmognathus [ 17]) differs in two respects from a simple aqueous solution . First, the saturable segment of the isotherm demonstrates binding to nondiffusive sites . From the intercept of the linear portion of the isotherm, the binding capacity of animal ooplasm is -75 mM. Nondiffusive or bound Na is associated closely with yolk, probably as counterion to abundant phosphate and carboxylic residues ; it can be viewed as one of the metabolic stores provided by this organelle (13, 17, 19, 20). Isotopic exchange with yolk sodium is very slow . The Na concentration in yolk water is about 100 mM, which makes it 6-10 times more concentrated than in cytoplasm or nucleoplasm . This large difference between yolk and ground substance sodium concentrations is chiefly responsible for the inhomogeneous distribution seen for this cation .' Second, the linear portion of the sodium isotherm has a slope of -0.35 (Fig. 5). This slope is substantially below that of an isomolar line, indicating that only -35% of ooplasmic water is available as solvent . That is, because of reduced solvent capacity, diffusive sodium is excluded from -65% of the ooplasmic water. Ooplasmic exclusion is largely explained by another property of yolk . Yolk contains two dominant proteins (phosvitin and lipovitellin), present as a hydrate crystal (22-24) . Water in protein crystals is compartmentalized by the protein backbone and modified by numerous coulombic, hydrogen bond, and "hydrophobic" interactions with functional groups (25, 26) . These interactions reduce water's capacity to dissolve most polar solutes, giving rise to a property which can be called hydrate crystal solute (HCS)-exclusion . Because a large portion of ooplasm is yolk and the water in this portion exhibits HCS-exclusion, the fraction of ooplasm's water available as a solvent is much less than that in the reference phase . With the exception of urea, every polar solute studied is excluded significantly by ooplasmic water. Because exclusion is a solvent property it is, to a first approximation, independent of solute concentration and (as in the case of sodium), described by the linear (nonsaturable) portion of an isotherm . For solutes that do not bind, for example, sucrose and 30MG, the ooplasm/iRP isotherm is linear throughout, with a slope less than unity and an intercept at the origin (Fig. 6) . HCSexclusion is seen in the ooplasm/iRP equilibrium distribution ratios for a nucleotide (ATP), a disaccharide (sucrose), an amino acid (AIB), and a monosaccharide (30MG; Table 1), and exclusion is also found for inorganic ions, polyols, polysaccharides, and polypeptides. Furthermore, because of the greater regional density of yolk platelets, HCS-exclusion is more intensive in vegetal than in animal ooplasm . Solute exclusion and binding by yolk are interesting because they are examples of the behavior of one of the "states" accessible to cellular proteins-the hydrate crystal . On the other hand, because of their abundance, yolk platelets create a problem ; they mask the properties of ooplasmic ground substance. However, the oocyte nucleus lacks yolk and a significant volume of other organelles. Thus, reference phase ' Inhomogeneity in potassium's distribution isalso largely determined by the distribution of yolk (Table I). Like sodium, yolk potassium is mostly "bound ." However, potassium's concentration in yolk platelets (-69 mM) is lower than in cytoplasm and nucleoplasm (13, 15, 17,21). c U c 0 U v acts O O RP Sucrose Concn Animal (Ao) and vegetal ooplasm (Vo) vs . iRP isotherms for sucrose (filled circles) and 30MG (open circles). Slopes of regression lines for sucrose are Ao 0.45 ± 0.06, Vo 0.30 ± 0.05; for 30MG, they are Ao 0.55 ± 0.01, Vo 0.41 ± 0 .07. All slopes are significantly lower than unity (P < 0 .001). Sucrose slopes in both AD and Vo are significantly lower than respective 30MG slopes (P FIGURE 6 < 0.01) . analysis of nucleoplasm is the best hope for defining the solvent properties of ground substance in full-grown oocytes . Nuclear Solute Binding and Exclusion The oocyte nucleus, with a water content of 89%, is a relatively dilute protein water phase, and its envelope is freely permeable to small solutes (14, 27). Hence, we might expect solute concentration ratios between nucleus and iRP water to be unity. This is the case for sucrose, AIB and ATP, and is approximately true for sodium, but not for potassium and 30MG (Table I B). Nuclear potassium significantly exceeds the iRP concentration, whereas nuclear 30MG is lower than in the iRP. The Rana pipiens oocyte nucleus has a potassium concentration 19 mM greater than the iRP (Table I). This confirms an earlier finding in the salamander, Desmognathus ochrophaeus, and demonstrates nuclear potassium binding (15). Analysis of the Rana pipiens oocyte's potassium economy is still incomplete; however, several points are clear . First, although ooplasm and nucleus share common membrane-controlled diffusive sodium and potassium pools, the proportion of each cation that is bound and free is different. The bound potassium/sodium ratio is greater than unity in the nucleus but less than unity in ooplasm . It follows that nuclear and ooplasmic (chiefly yolk) binding sites differ, the former having a greater potassium affinity than the latter . Second, we estimated the nucleotide-associated phosphate content of the oocyte nucleus using the RNA and DNA data of Finamore et al. (28) and Dawid (29) and found that these anions could neutralize less than 1 meq ofthe observed nuclear potassium excess (Lau, Y.-T., and S. B. Horowitz, unpublished observation). On the other hand, nucleoplasmin, a recently discov- ered phosphorylated peptide that comprises about 10% of total nuclear protein, could supply the missing fixed anions (30, 31). Nuclear exclusion of 30MG is most clearly seen when 30MG and sucrose isotherms are compared . Fig. 7 shows the results ofan experiment in which [ t'C]sucrose and [3H]30MG were present simultaneously in oocytes . These and other experiments show that, irrespective oflabeling or introduction protocol, the sucrose nucleus/iRP concentration ratio is unity, whereas the 30MG ratio is significantly less than unity. This contrasts with ooplasm, where ooplasm/iRP ratios for both solutes are much less than unity and sucrose is excluded more than 30MG (Fig. 6). Indeed, the finding that a monosaccharide, 30MG, is excluded from nuclear water, whereas a larger disaccharide is not, seems counterintuitive . An explanation begins to emerge when the nucleus/iRP distribution of other solutes is considered . Table II lists equilibrium nuclear/iRP distributions for 13 organic solutes. Of these, three exhibit nuclear exclusion : 30MG, t.-glucose, and D-xylose. The excluded solutes are all hexoses or pentoses and differ from the other solutes (except myo-inositol) in their closed-ring structure, and from myoinositol in the presence of a ring oxygen. The explanation we propose for these data invokes current models of the steric properties of both water and monosaccharides.' Aqueous systems are hydrogen-bonded networks of water and other molecules . Each system contains a different array of microstructures and microstructure lifetimes that depend on temperature, pressure, and the nature of the nonwater components. Hence, every aqueous system has an "instantaneous" structure (32, 33). Because of this, solute-water bond strength (which determines the free energy required to move a solute from one aqueous environment to another, and consequently interphase solute concentration ratios) depends on the solute's ability to fit into the preexisting microstructure and form low-strain hydrogen bonds with water. The less compliant a solute is in its ability to orient to form hydrogen bonds, the greater will be its capacity to recognize structural differences between aqueous phases . Monosaccharides are especially useful because their several hydrogen bonding sites are arrayed in a closed ring configuration that reduces orientational freedom (34). For this reason, monosaccharides are sensitive probes of water structure and of microstructural differences between aqueous systems. The data ofTable II can be explained within this conceptual framework . We have hypothesized, first, that nuclear water is, on average, differently structured than saline or iRP water and, second, that, compared with ordinary saline water, hydrogen-bond opportunities available to ringed monosaccharides are reduced in nuclear water. In other words, the precisely cut monosaccharide key is less able to fit into the modified lock structure of nuclear water than the "native" lock structure of ordinary saline water. Comparison of myoinositol and monosaccharide data (Table II) point to the ring oxygen of the latter as a critical determinant. Afterthoughts What is the significance to the cell of the nuclear water properties disclosed by monosaccharide exclusion? The answer depends largely on discoveries still to be made. If, for example, the water of nucleoplasm and cytoplasm differ, the ' Horowitz, S. B. Manuscript in preparation. HOROWITZ AND MILLER Solvent Properties of Ground Substance 177s FIGURE 7 Nuclear vs . iRP isotherms for sucrose and 30MG . The dashed line is isomolar . The slope of the regression line for sucrose (not shown) is 1 .01 ± 0 .01 ; that for 30MG (solid line) is 0 .74 ± 0 .02, which differs significantly from unity (P < 0 .001). Reference Phase Concentration TABLE II Nucleus (N) to iRP Equilibrium Concentration Ratios in Rana pipiens Oocytes Solute Sucrose 3-O-Methylglucose L-Glucose D-Xylose Mannitol Myo-inositol a-Aminoisobutyric acid Alanine Valine Leucine Isoleucine Glutamine Glutamic Acid ATP NJiRP 1 .00 ± 0 .01 0 .74 ± 0 .03 0 .70 ± 0 .02 0 .70 ± 0 .03 0.93 ± 0 .05 1 .01 ± 0 .02 1 .04 ± 0 .02 1 .05 ± 0 .23 0.95 ± 0 .32 0 .91 ± 0 .41 1 .14 ± 0 .22 0.94 ± 0 .15 1 .05 ± 0 .10 1 .00±0 .03 (N vs . iRP) (77) (46) (9) (9) (10) (13) (25) (16) (12) (11) (12) (15) (17) (11) NS <0.001 <0.001 <0.001 NS NS NS NS NS NS NS NS NS NS Data are given as mean ± SE with the number of oocytes in parentheses . Radiolabeled sucrose, AIB, 30MG, D-xylose, L-glucose, mannitol, and myoinositol were analyzed by liquid scintillation counting, endogenous amino acids by high-performance liquid chromatography, and ATP by a luciferinluciferase procedure. Thin-layer chromatographic analysis of oocyte extracts showed negligible metabolism of labeled solutes. iRP-gelatin/intracellular "Ringer's" concentration ratios for all solutes, determined by equilibrium dialysis, were unity. NS, not significant . kinetics and thermodynamics of every enzymatic reaction that involves water (as solvent, substrate, or product) will also differ in the two regions. Because water participates in almost every reaction, water structure differences could underlie the regional (nucleocytoplasmic) specialization characteristic of eukaryotic cells. It is also possible that nuclear water in other cells exhibits more or less exclusion that that of the oocyte and that the magnitude of exclusion is a function of physiological state. If so, we may expect water-ground substance interactions to have roles in differentiative, metabolic, and physiologic control. Despite monosaccharide exclusion, nuclear water is experienced by most solutes as similar to saline (iRP) water . This has led us to reject discrete state models in which ground substance water is either "bound" or "bulk ." Instead, we view ground substance water in the same way as modern physical chemistry views other aqueous solutions: as containing an instantaneous microstructure that requires a statistical description (32-35) . Monosaccharide exclusion discloses only that nuclear water's instantaneous microstructure is different, presumably because of interaction with specialized nuclear cytomatrix proteins. As we have been reminded (36), this 78S THE JOURNAL OF CELL BIOLOGY " VOLUME 99, 1984 should not be surprising, inasmuch as the cell and its parts are the products of several billion years of intimate watermacromolecule evolution . Perhaps future experiments (Fig. 1) will disclose other examples of cell water's microstructural adaptation. We thank Y.-T . Lau and P. L. Paine for their valuable suggestions and advice, C. Johnson for her secretarial assistance, and S. Harmon, S. Madry, B. Mahalek, L. J. M. Tluczek, and J. White for their skilled technical assistance. This work was supported by National Institutes of Health grants GM 19548 and HD12512 and an institutional grant from the United Foundation of Greater Detroit . REFERENCES 1 . Roth, L. J., and W. E. Stumpf, editors. 1969. Autoradiography of Diffusible Substances. Academic Press. Inc., New York. 2. Kinter, W. B., and T. H. Wilson. 1965 . Autoradiographic study of sugar and amino acid absorption by everted sacs of hamsterintestine. J Cell Biol. 25:19-40. 3. Horowitz, S. B. 1974. The ultra-low temperature autoradiography of water and its solutes. Methods Cell Biol. 8:249-275 . 4. Stirling, C. E., and W. B. Kinter . 1967. High-resolutio n radioautography of galactose'H accumulation in ringsof hamster intestine . J Cell Biol. 35 :585-604. 5. Gupta, B. L., T. A. Hall, and R. B. Moreton . 1977. Electron probe X-raymicroanalysis. In Transport of Ions and Waterin Animals. B. L. Gupta, R. B. Moreton, J. L. Oschman, and B. J. Wall, editors . Academic Press, London, 83-143 . 6. Somlyo, A. V., H. Schuman, and A. P. Somlyo. 1977 . Elemental distributions in striated muscle and the effects of hypertonicity . Electron probe analysis of cryo sections . J Cell Biol. 74 :828-857 . 7. Siebert, G., and G. B. Humphrey. 1965. Enzymology of the nucleus. Adv. Enzymol. Relay. Subj. Biochem . 27:239-288 . 8. Katzman, R., G. M. Lehrer, andC. E. Wilson. 1969. Sodium and potassium distribution in puffer fish supramedullary nerve cell bodies. J Gen. Physiol 54 :232-249 . 9. Century, T. J., I. R. Fenichel, and S. B. Horowitz. 1970 . The concentrations of water, sodium and potassium in the nucleus and cytoplasm of amphibian oocytes. J. Cell Sci. 7:5-13. 10 . Frank, M., and S. B. Horowitz . 1975. Nucleocytoplasmi c transport and distribution of an amino acid, in situ. J. Cell Sci. 19 :127-139. 11 . Horowitz, S. B., and T. W. Pearson. 1981 . Intracellular monosaccharide and amino acid concentrations and activities and the mechanisms of insulin action . Mot Cell. Biol. 1 :769-784. 12 . Clark, T. G., and J. L. Rosenbaum. 1979 . An actin filament matrix in hand-isolated nuclei of X. laevis oocytes. Cell. 19:1 lot-1108 . 13 . Tluczek, L. J. M., Y-T. Lau, and S. B. Horowitz. Water, potassium and sodium during amphibian oocyte development. Dev. Biol. In press. 14 . Paine, P. L., and S. B. Horowitz. 1980. The movement of material between nucleusand cytoplasm. In Cell Biology: A ComprehensiveTreatise . L. Goldstein and D. M. Prescott, editors. Academic Press, Inc., New York . Vol. 4, 299-338 . 15 . Paine, P. L., T. W. Pearson, L. J. M. Tluczek, andS. B. Horowitz . 1981 . Nuclearsodium and potassium . Nature (Land.). 291:259-261 . e phase 16. Horowitz, S. B., P. L. Paine, L. Tluczek, and J. K. Reynhout. 1979 . Referenc analysis of free andbound intracellular solutes . 1. Sodium and potassium in amphibian oocytes. Biophys. J. 25 :33-44. e phase analysis of free and bound 17. Horowitz, S. B., and P. L. Paine. 1979. Referenc intracellular solutes. 11. Isothermal and isotopic studies of cytoplasmic sodium, potassium, and water. Biophys. J 25:45-62 . 18 . Horowitz, S. B. 1984. Testingthe association-induction (All hypothesis. In Biophysical Correlates of Cellular Function . Proceedings of UNESCO Inter Workshop . C. F. Hazelwood, editor. DiPuma Printing, Houston . In press. 19. Century,T. J., and S. B. Horowitz. 1974 . Sodium exchange in the cytoplasm and nucleus of amphibian oocytes. J. Cell Sci. 16:465-471 . 20. Horowitz, S. B., and 1. R. Fenichel. 1970. Analysisof sodium transport in theamphibian oocyte by extractive and radioautographic techniques. J Cell Biol. 47 :120-131 . 21 . Frank, M., andS. B. Horowitz. 1980. Potassium exchange in the whole cell, cytoplasm and nucleus of amphibian oocytes. Am. J. Physiol 238:Ct33-C138 . 22 . Wallace, R. A. 1963 . Studies on amphibian yolk. IV. Analysis of the main body components of yolk platelets. Biochim . Biophys. Acta. 74:505-517. 23 . Wischnitzer, S. 1966. The ultrestructure of the cytoplasm of the developing amphibian egg. Adv. Morphog. 5: 131-179. 24 . Redshaw, M. R., and B. K. Follett. 1971 . Thecrystalline yolk-platelet proteins and their soluble plasma precursor in an amphibian Xenopus laevis. Biochem . J 124:759-766. 25 . Eagland, D. 1975. Nucleic acids, peptides and proteins. In Water, A Comprehensive Treatise. F. Franks, editor. Plenum Press, New York. Vol. 4, 305-518 . 26 . Edsall, J. T., and H. A. McKenzie. 1983 . Water and proteins. II. The location and dynamics of water in protein systems and its relation to their stability and properties . Adv. Biophys. 16 :53-183. 27 . Paine, P. L., L. C. Moore, and S. B. Horowitz. 1975 . Nucleu s envelope permeability . Nature (Land.). 254:109-114 . 28 . Finamore, F. J., D. J. Thomas, G. T. Crouse, and B. Lloyd. 1960. Biochemistry of amphibian oocytes. I. Method of isolation and nucleic acid content of nuclei. Arch . Biochem . Biophys. 88 :10-16 . 29. Dawid, I . B. 1965 . Deoxyribonucleic acid in amphibian eggs. J Mol. Biol. 12:581-599 . 30. Krohne, G., and W. W. Franke . 1980 . Immunological identification and localization of 31 . 32 . 33 . 34 . 35 . 36 . the predominant nuclear protein of the amphibian oocyte nucleus. Proc . Nad. Acad. Sci. USA. 77:1034-1038 . Dingwall, C., S. V. Shamick, and R. A. Laskey. 1982 . A polypeptide domain that specifies migration of nucleoplasmin into the nucleus. Cell. 30 :449-458 . Finney, J. L. 1982. Towards a molecular picture of liquid water. In Biophysics of Water. F. Franks and S. Mathias, editors. John Wiley & Sons, Chichester . 73-95. Beveridge, D. L ., M. Mezei, P. K. Mehrotra, F. T. Marchese, G. Ravi-Shanker, T. Vasu, and S. Swaminathan. 1983 . MonteCarlocomputer simulation studies ofthe equilibrium properties and structure of liquid water. ACS (Am. Chem . Soc.) Adv. Chem . 204:297351. Suggett, A. 1976. Molecular motion and interactions in aqueous carbohydrate solutions. III. A combined nuclear magnetic and dielectric-relaxation strategy . J. Sol. Chem. 5:3346 . Beveridge, D. L., M. Mezei, P. K. Mehrotra, F. T. Marchese, T. Vasu, and G. RaviShanker. 1981 . Liquid state computer simulations of biomolecular solvation problems. Ann. NYAcad. Sci. 376:108-131 . Clegg, J. S. 1982 . Alternative views on the role of water in cell function . In Biophysics of Water. F. Franks and S. Mathias, editors. John Wiley & Sons, Chichester . 365-383 . HOROWITZ AND MILLER Solvent Properties of Ground Substance 179s