* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Amiodarone - pharminfotech
Discovery and development of direct thrombin inhibitors wikipedia , lookup
Drug discovery wikipedia , lookup
Adherence (medicine) wikipedia , lookup
Psychedelic therapy wikipedia , lookup
Electronic prescribing wikipedia , lookup
Neuropsychopharmacology wikipedia , lookup
Pharmaceutical industry wikipedia , lookup
Neuropharmacology wikipedia , lookup
Prescription drug prices in the United States wikipedia , lookup
Pharmacognosy wikipedia , lookup
Pharmacokinetics wikipedia , lookup
Prescription costs wikipedia , lookup
Pharmacogenomics wikipedia , lookup
Psychopharmacology wikipedia , lookup
Drug interaction wikipedia , lookup
Theralizumab wikipedia , lookup
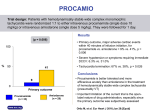
“Q.I.D.”… QUALITY INFORMATION on DRUGS September 2004 Dunedin Medicines Information Service, Pharmacy Department, Dunedin Hospital Telephone: 8876 (Internal), (03) 474 7658 (Direct), Facsimile: (03) 474 7638 Amiodarone Amiodarone is a difficult drug to use due to its long half-life, interactions, adverse effects and incompatibilities with solutions and containers. This bulletin aims to answer some of the frequent questions that arise regarding its use. Amiodarone is used to treat tachyarrhythmias including Wolff-Parkinson-White syndrome, supraventricular, nodal and ventricular tachycardias as well as atrial fibrillation and atrial flutter. Dosing Cardiologists recommend various dosing regimens but the following is one that is commonly used in Dunedin Hospital. Route IV Use Management of acute arrhythmias (Short term only) Loading dose Oral Maintenance dose Dose Starting dose 5mg/kg in 5% Dextrose 250ml over 60 minutes. Repeat infusions up to 1200mg in 24 hours (administered via a pump with cardiac monitoring). 200mg three times daily for one week then reduce to 200mg twice daily for one week then reduce to 100 - 200mg daily Notes on loading amiodarone: IV amiodarone has a limited role in loading, it is used in clinical situations where a rapid response is required. Oral loading is still required. A shortened oral loading regimen can be given after IV dosing at the clinicians discretion for less serious arrhythmia such as AF. The full oral loading regimen should be used in life-threatening arrhythmias such as VF or VT. Kinetics Amiodarone has a very long half-life, reported to be between 14 and 110 days with chronic dosing. Maximal antiarrhythmic effects of oral amiodarone may take up to 10 weeks, even with loading. Amiodarone accumulates in many different tissues with a range of serious side effects e.g. hypo/hyperthyroidism, corneal microdeposits, pulmonary toxicity, hepatotoxicity, peripheral neuropathy, skin discolouration. These effects should be monitored during long-term therapy. Amiodarone may cause photosensitivity so patients should protect themselves from the sun. The effects of the drug can remain for months after it has been discontinued. The effect of a missed dose or change in brand will be minor, and may not be measurable until weeks afterwards. Infusions Amiodarone infusions must be administered carefully for two reasons. The drug itself can be absorbed into PVC infusion bags and administration sets, and plasticisers from some administration sets can leach out into the amiodarone solution. For initial, rapid infusions (60mins), any type of bag can be used as absorption is minimal. For slow infusions use glass or rigid PVC bottles and ‘LoSorb IV’ or ‘vented nitro-glycerine’ giving sets. Hang infusion solution immediately after preparation and discard if not used within 12 hours. Frequently Asked Questions Do you increase the maintenance dose if there is poor control? No, increasing the oral maintenance dose above 200mg is not recommended. Another antiarrhythmic medication can be added as the next step to achieve sinus rhythm. Occasionally, after cardiologist consultation, amiodarone levels can be measured and the dose increased if necessary. What if the patient is nil by mouth? The bioavailability of amiodarone is approximately 50% so if a patient is switched from oral to iv, give approximately half the oral dose. e.g. instead of 200mg tds po give 150mg bd iv. Do you need to reduce the dose in renal impairment? No, very small amounts are excreted in the urine. Do the oral and IV formulations exert different effects? There is some evidence that chronic oral dosing has different effects on conduction pathways than IV dosing. IV amiodarone exerted additional benefits to long-term oral amiodarone therapy in one trial. It is unclear whether cumulative effects, active metabolites or both are responsible for the differences. Can the brands of oral amiodarone be switched safely? Amiodarone is not on the interchangeable medicine list, so it is preferable that a patient remains on the same brand if possible. There have been reports of increases and decreases in amiodarone levels following a change in formulation. If the brand was switched temporarily during a hospital stay, the effect is likely to be minor due to the long half-life. If the brand is switched permanently, patient monitoring is recommended followed by a drug level after a few weeks if the clinician is particularly concerned. Drug Interactions Due to amiodarone’s long half-life, interactions may take several weeks to fully develop and may persist for months after the drug is discontinued. The following drugs should be monitored when amiodarone is added to a stabilised regimen. Interacting Drugs Warfarin Digoxin CYP3A4 substrates eg. diltiazem, cyclosporin simvastatin Drugs that slow AV conduction e.g. beta-blockers, calcium channel blockers Drugs that prolong the QT interval e.g. erythromycin, cisapride antidepressants, antipsychotics, Management Monitor INR closely as the effect of warfarin is likely to be increased. Expect to reduce the warfarin dose by approximately 50% (variable). The interaction can take at least two weeks to fully develop. Amiodarone reduces the excretion of digoxin. The dose of digoxin should be reduced by 1/3 to 1/2 when amiodarone is added to the regimen. Monitor patient for signs of toxicity, digoxin levels can be taken after a few days if toxicity is suspected, otherwise after a week or two to check if the steady state level is not above the therapeutic range. Amiodarone is a CYP3A4 substrate and inhibitor. The levels of drugs metabolised by this enzyme are likely to be increased by the addition of amiodarone. Monitor patient for side effects and toxicity, dose reduction of these drugs should be considered. The pharmacodynamic effects of amiodarone may be additive. Monitor patient for hypotension, bradycardia and ECG changes. Avoid if possible due to the potential to cause ‘torsades de pointes’. References available on request from Medicines Information Service (Ext 8876).