* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Functional Groups - Effingham County Schools
Survey
Document related concepts
Transcript
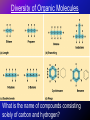
Carbon and the Molecular Diversity of Life • Biological macromolecules – carbohydrates, protein, lipids, and nucleic acids (DNA & RNA) - are all composed of carbon atoms • Other important elements include: – Hydrogen (H) – Oxygen (O) – Nitrogen (N) – Sulfur (S) – Phosphorus (P) Organic Chemistry • Organic Chemistry is the study of carbon compounds – compounds of living things • Carbon has 4 valence electrons –usually forms 4 covalent bonds with other atoms –allows molecules to branch off in up to 4 directions –makes large, complex molecules possible Diversity of Organic Molecules • Carbon atoms readily bond with each other, producing chains or rings of carbon atoms • Carbon chains form the backbones of most organic molecules • These carbon backbones can vary in length, branching, placement of double bonds, and location of atoms of other elements • The variations in these skeletons contributes to the complexity and diversity of living matter. Diversity of Organic Molecules What is the name of compounds consisting solely of carbon and hydrogen? Hydrocarbons • Organic molecules consisting of only carbon and hydrogen • Major component of petroleum (fossil fuels) – partially decomposed remains of organisms that lived millions of years ago • Are hydrophobic because the bonds between C and H are nonpolar • Store a relatively large amount of energy (fat is stored energy for the body) Isomers • Compounds that have the same molecular formula, but different structural formulas; therefore, different properties • Three types: structural isomers, geometric isomers, and enantiomers • Structural isomers of butane, C4H10 Structural Isomers Butane Isobutane Both have the same molecular formula, but different structural formulas • Differ in the covalent arrangement of their atoms • C20H42 has 366,319 possible structural isomers • May differ in the placement of the double bond Geometric Isomers • Same covalent partnership, differ in spatial arrangement • Due to inflexibility of double bonds – do not allow atoms to rotate about the axis • The subtle difference in shape can dramatically affect the biological activities of the molecule • Example – rhodopsin in the eye Enantiomers • Mirror images of each other • Differ in spatial arrangement around an asymmetric carbon (a carbon atom bonded to 4 different kinds of atoms or groups of atoms) • Each enantiomer may behave differently in the body Functional Groups • Components of organic molecules most commonly involved in chemical reactions • Attachments that replace one or more of the hydrogens bonded to the carbon skeleton of the hydrocarbon • Behaves consistently from one organic molecule to another • Number and arrangement of groups give each molecule its unique properties • Example: estradiol and testosterone Functional Groups Hydroxyl Group • Organic compounds containing hydroxyl are alcohols • Name usually ends in –ol (i.e. ethanol) • Hydrophilic – soluble in water Functional Group Formula Name of Compound Example Functional Groups Carbonyl Group • A carbon atom joined to an oxygen atom by a double bond • If the carbonyl group is on the end, the compound is an aldehyde • Anywhere else, the compound is a ketone (i.e. acetone) • Hydrophilic – soluble in water Functional Group Formula Name of Compound Example Functional Groups Carboxyl Group • An oxygen atom double-bonded to a carbon atom that is also bonded to a hydroxyl group • Compounds containing a carboxyl group are called carboxylic acids or organic acids • Hydrophilic – soluble in water • Have acidic properties Functional Group Formula Name of Compound Example Functional Groups Amino Group • A nitrogen atom bonded to two hydrogen atoms and to the carbon skeleton • Compounds containing an amino group are called amines (i.e. glycine – an amino acid) • Hydrophilic – soluble in water • Have basic properties Functional Group Formula Name of Compound Example Functional Groups Sulfhydryl Group • A sulfur atom bonded to an atom of hydrogen • Compounds containing a sulfhydryl group are called thiols • Hydrophilic – soluble in water • Interact to stabilize the structure of a protein Functional Group Formula Name of Compound Example Functional Groups Phosphate Group • A phosphate ion covalently attached by one of its oxygen atoms to the carbon skeleton • Transfers energy between organic molecules (a process exhibited in cellular respiration) • Hydrophilic – soluble in water Functional Group Formula Name of Compound Example