* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Lecture 3. Fluorescence microscopy I
Wave–particle duality wikipedia , lookup
Franck–Condon principle wikipedia , lookup
X-ray fluorescence wikipedia , lookup
Protein–protein interaction wikipedia , lookup
Chemical imaging wikipedia , lookup
Ultrafast laser spectroscopy wikipedia , lookup
Fluorescence correlation spectroscopy wikipedia , lookup
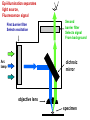
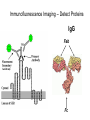
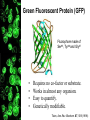
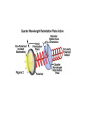
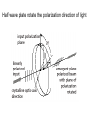
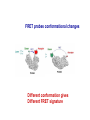
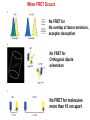
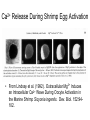
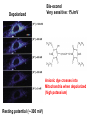
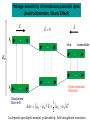
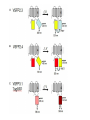
Letting someone else doing my job http://probes.invitrogen.com/resources/education/tutorials/1Intro/player.html Epi-illumination is form of Kohler Illumination: Objective is also condenser White light (regular Kohler) Brightfield, phase, etc Light is focused At back aperture Of the objective, Conjugate to condenser aperture Different illumination And image paths Lamp or laser lens detector Detect at 90 degrees Split with dichroic mirror Greatly increases S/N Epi-illumination separates light source, Fluorescence signal First barrier filter Selects excitation Arc lamp Second barrier filter Selects signal From background dichroic mirror objective lens specimen General Jablonski Diagram Typical molecular timescales: S2 Absorption: instantaneous (10-17 s) Vibronic Relaxation ~ 10-12 s S1 T1 Fluorescence: 10-6 -10-12 s 10-9 most typical Singlet to a singlet (strong) S0 Fluorescence always from relaxed level of S1 Phosphorescence: 10-3 -10-6 s Triplet to singlet (weak) Magnitude of Extinction Coefficients Extinction coefficient ε: Beer’s Law A= εcl Strong absorbers (dyes) have ε between 20,000-100,000 Absorption cross section is also used: 1x 10-16 cm2 = 23,000 ε Brightness= absorption coefficient* QY Oscillator strength is integral of the absorption band Sum rule: oscillator strength, f, for one electron over all transitions is: 1x 10-16 cm2 eV Emission spectrum is independent of of path excitation (both spectral wavelength and width): always from S1 Emission intensities will be different Due to different absorption probability: Franck-Condon Principle Franck-Condon Principle Consider electronic states anharmonic oscillators: bond length Most probable transitions are “vertical” Big geometry change=broad spectrum (smaller maximum absorption) Both for absorption and emission: conserve oscillator strength Geometry and Absorption and Emission Spectra Bigger Stokes shifts provide better signal to noise In fluorescence because of filter efficiencies, (dichroics, low pass, high pass) But usually involve large geometry change: lower intensities Spreading out oscillator strength Fluorescence Quantum Yield φ: important for dyes Ratio of emitted to absorbed photons Quantum Yield: kf k f k isc (k is rate, Inverse of time) kf k f k isc k nonrad 1 k f kisc Measured lifetime is sum of natural lifetime and non radiative decay paths Lifetime is useful contrast Probe of environment B. Fluorescent Probes • Molecular Probes (Invitrogen) – www.probes.com – Catalog contains thousands of fluorescent probes, with valuable technical information. Organelle Probes Target, Name Mitochondria MitoTracker dyes Golgi BODIPY FL and TR C5-ceramide Lysosomes LysoTracker LysoSensor Endoplasmic reticulum DiOC6, DiIC6 Fluorescent Brefeldin A Excitation Emission 490/578/551 516/599/576 505/589 511, 620 /617 Various Various Various Various 484/549 501/565 Comments MT Green accumulates in mitochondria regardless of MB potential, Red and Orange in active mitochondria; aldehyde fixable TR is better for double labeling, since no green emission DiO and DiI also stain membranes generally Inhibitor of protein transport Immunofluorescence Imaging – Detect Proteins IgG Fab Fc Fluorescence in situ Hybridization (FISH) – Detecting Nucleic Acids Fluorescein: most common dye for microscopy Blue green Xanthene family “green fluorophore” e80,000, φ~0.9 Brightness ~ ef Many functionalized forms for cell imaging: pH, ion sensing •High quantum yield •General purpose •But degrades quickly •Small Stokes shift (filter bleedthrough) Rhodamine 6G Green Red Xanthene family “red fluorophore” Internal Donor-acceptor pair Red-shifts the spectra relative to fluorescein Many functionalized forms for cell imaging •High quantum yield •General purpose •Good stability •Also Small Stokes shift Green Fluorescent Protein (GFP) Fluorophore made of Ser65, Tyr66 and Gly67 • • • • Requires no co-factor or substrate. Works in almost any organism. Easy to quantify. Genetically modifiable. Tsien, Ann.Rev. Biochem. 67, 509 (1998) Many fluorescent proteins: Jellyfish, Coral Reefs Colored Proteins allow labeling of multiple specific organelles Variants of Fluorescent Proteins BFP EBFP, Sapphire, T-sapphire CFP ECFP, mCFP, Cerulean, CyPet, AmCyan, Midoriishi Cyan GFP EGFP, Azami Green, TurboGFP, ZsGreen, Emerald YFP EYFP, Topaz, Venus, mCitrine, YPet, ZsYellow1, PhiYFP OFP mBanana, Kusabira Orange, mOrange RFP dsRed, tangerine, dTamato, mStrawberry, AsRed2, mRFP, mCherry, mRasberry, mPlum, JRed, HcRed GFP Chromophore Aequorea: FSYGVQ Renilla: p-hydroxybenzylidene-imidazolidone Ser - dehydroTyr - Gly FSYGDR Chromophore Maturation Takes ~ 30 min for wild type GFP. GREEN FLUORESCENCE PROTEIN GFP Jelly fish isolate DNA encoding GFP cellular protein cellular protein couple gene for GFP with gene for protein of interest GFP transform cell with altered protein Completely general and versatile Problems with Fluorescent Protein • Size comparable to the target. Might interfere with the function of the target protein • Maturation time • Probably not 100% fluorescent • PH dependence • Many variants mis-fold when fused to another protein Linearly Polarized Light s= horizontal p= vertical For propagation Parallel to floor Polarizer is device that selects polarization Can be crystal or film (Polaroid) Operation of Analyzer (Birefringent) Light transmitted at angle relative to angle of 2 Crossed polarizers 2 I I 0 cos Combining linear polarized light IN PHASE Linear Polarization OUT OF PHASE Elliptical Polarization Circularly Polarized Light • Decompose to linear polarized light with 1/4 phase shift. • No direction (always pass 50% through polarizer in dependent of polarizer orientation) • NOT the same as unpolarized light. • Can be converted back to linear polarized light with birefringent materials (1/4 wave plate). Half wave plate rotate the polarization direction of light Absorption is polarized Fluorescence is also polarized GFP Crystal Anisotropic sample - Fluorescent intensity is dependent on the polarization _and_ the orientation of the molecules Isotropic sample - Fluorescent intensity is independent of excitation polarization - Fluorescence is polarized if the excitation is polarized. Fluorescence anisotropy r < 0.4 Microscopic Measurements of Anisotropy r = r0 / ( 1 + / ) Use Small Numerical Aperture Fluorescence Resonance Energy Transfer (FRET) Förster Radius The distance at which energy transfer is 50% efficient (i.e. 50% of excited donors are deactivated by FRET) is defined by the Förster Radius (R0). Applications of FRET in Biology Survey of FRET-Based Assays • • • • • • • Protease activity Calcium Ion measurements cAMP Protein tyrosine kinase activity Phospholipase C activity Protein kinase C activity Membrane potential FRET probes conformational changes Different conformation gives Different FRET signature Inter and Intramolecular Forms of FRET with Proteins CFP-YFP good combo FRET increases In both cases Protein-Protein Interactions In cytoplasm and membranes When FRET Occurs No FRET for No overlap of donor emission, acceptor absorption No FRET for Orthogonal dipole orientation No FRET for molecules more than 10 nm apart Number of FRET Publications since 1989 Fluorescence Resonance Energy Transfer Detection of Probe Proximity F FD FA F D FRET Max 0 0 FD FA FA D0 0 D 0 A 0 D R0 typically 40-50 Angstroms 50% transfer R06 FRET 6 6 R R0 Typical Values of Ro green Donor Fluorescein Acceptor Ro (Å) Tetramethylrhodamine 55 IAEDANS EDANS Fluorescein Fluorescein DABCYL Fluorescein 46 33 44 BODIPY FL Fluorescein Cy3 CFP BODIPY FL QSY 7 dye Cy5 YFP 57 61 53 50 red GFPs and other colored “FPs have transformed FRET microscopy FRET Considerations: 1. Spectral overlap 2. Chromophore orientations 3. Distance dependence (Eff. 1/R6) 4. How to quantify? E E f acceptor f donor f acceptor f baseline f A spillover f donor f D spillover Practical Challenges to FRET Quantitation • • • • Emission from A contaminates D channel (filters) Emission from D contaminates A channel Unknown labeling levels for D and A Signal variation due to bleaching – Complicates kinetic studies – Bleaching rate of D can actually be slowed by FRET Solutions: • Separately labeled D and A controls to define bleedthrough 0 F • Acceptor destruction by photobleaching to establish D • Dual wavelength ratio imaging to normalize away variations in label levels and bleaching effects Ca2+ Release During Shrimp Egg Activation • From Lindsay et al. (1992). Extracellular Mg2+ Induces an Intracellular Ca2+ Wave During Oocyte Activation in the Marine Shrimp Sicyonia ingentis. Dev. Biol. 152:94102. Low quantum yield with no Ca2+, big increase When binds Ca2+: up to 50 fold increase Not absolute concentration of ions, measure relative changes: easier Fluo- dyes By Tsien Choose depending on desired Range of sensing Blue Ca2+ Indicators: Fluo-3 has single Ex and Em wavelengths • A visible light excitable dye (488 nm), so Argon laser can be used. • Emission at 525 nm. • OK for qualitative detection but not quantitative. Calcium Sensing Indo-1 Ratiometric using single excitation, dual emission Excite 338 nm, collect 405, 485 nm fluorescence Determine absolute calcium concentration by imaging Free Ca2+ Concentration in a Purkinje Neuron from Embryonic Mouse Cerebellum • Neurons were loaded with fura-2. • Neurons were stimulated with glutamate receptor agonist. • The composite image represents the ratio of images obtained with excitation at 340 nm and 380 nm. Membrane Potential Membrane Potential Is Due to Charge Imbalance Depolarized Bis-oxonol Very sensitive: 1%/mV Anionic dye crosses into Mitochondria when depolarized (high potassium) Resting potential (~-300 mV) Voltage Sensitive Styryl Chromophore Fast dye Stains membrane e Max~30,000 Large geometry change, Charge shift upon absorption: Makes spectra sensitive to electric fields Charge shift in styryl dyes Excited state - + + - Ground state Voltage sensitivity of membrane potential dyes (electrochromism / Stark Effect) E S1 d E E 0 d d d E0 d S0 d Depolarized Blue shift intra extracellular d d d d d Hyper-polarized Red shift d 1 h G E E G E E 2 2 depends upon dipole moment, polarizability, field strength and orientation Mechanism of Voltage-dependent Spectral Shifts in Styryl Dyes ‘Electrochromism’ Red shift Blue shift Ratiometric approach at Inflection points for Highest sensitivity 1) Single excitation, dual emission: Laser excitation 2) Dual excitation, same emission: Arc lamp excitation F/F: normalizes Also more sensitive, Changes are small-10%/100 mV F/F: normalizes for geometrical factors, bleaching: Ratiometric approach used for many types of dyes