* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download carbon cycle
Survey
Document related concepts
Transcript
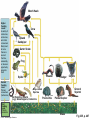
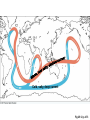
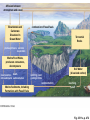
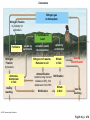
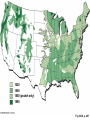
PowerLecture: Chapter 25 Ecology and Human Concerns Learning Objectives Understand how materials and energy enter, pass through, and exit an ecosystem. Describe how communities are organized, how they develop, and how they diversify. Understand the various trophic roles and levels. Diagram the principal biogeochemical cycles. Learn the language associated with the study of population ecology. Learning Objectives (cont’d) Understand the factors that affect population density, distribution, and change. Understand the meaning of logistic growth. Know the problems associated with the growth of human populations. Tell which factors have encouraged growth in some cultures and limited growth in others. Understand the magnitude of pollution problems in the United States. Learning Objectives (cont’d) Examine the effects modern agricultural techniques have on different ecosystems. Describe how our use of fossil fuels and nuclear energy affects ecosystems. Impacts/Issues The Human Touch The Human Touch At one time as many as 15,000 people lived on Easter Island. The tiny island could not support this many people. Crop yields declined; soil nutrients were depleted. Large statues were erected to appease the gods. The population dwindled and then disappeared as people turned against each other. Video: Easter Island CLICK TO PLAY From ABC News, Environmental Science in the Headlines, 2005 DVD. How Would You Vote? To conduct an instant in-class survey using a classroom response system, access the “Polls Clicker Questions” from the PowerLecture main menu. Are you willing to pay extra for “green” products? a. Yes, I would be willing to pay more for sustainable products. b. No, I would not be willing to pay more for “green” products. Section 1 Some Basic Principles of Ecology Some Basic Principles of Ecology The biosphere encompasses the earth’s crust, atmosphere, and waters that support life; a biome is one of the major realms of life, such as deserts or rain forests. Figure 25.1 Animation: Major Biomes CLICK TO PLAY Animation: Terrestrial Biomes CLICK TO PLAY Some Basic Principles of Ecology Ecology is the study of the interactions of organisms with one another and with the physical environment. A habitat is the place where a species normally lives; it is characterized by distinctive physical features and vegetation. Humans live in disturbed habitats, places we have modified to suit our own purposes. A community is the collection of all the populations in a given habitat. Some Basic Principles of Ecology The niche refers to a range of physical and biological conditions under which a species can live and reproduce. • • Specialist species have narrow niches. Generalists have broad ranges of habitats and niches. Some Basic Principles of Ecology An ecosystem consists of one or more communities interacting with one another and with the physical environment. Communities of organisms make up the biotic, or living, portions of an ecosystem. Figure 25.2 Some Basic Principles of Ecology Succession is the orderly progression of species changes that leads to a climax community. • • In primary succession, changes begin when pioneer species colonize a barren habitat. In secondary succession, a community reestablishes itself toward a climax state after a disturbance. Figure 25.3 Animation: Two Types of Ecological Succession CLICK TO PLAY Animation: Levels of Organization CLICK TO PLAY Video: Frogs Galore CLICK TO PLAY From ABC News, Biology in the Headlines, 2005 DVD. Section 2 Feeding Levels and Food Webs Feeding Levels and Food Webs Many ecosystems exist, but they are all similar in structure and function. Producers (autotrophs) capture sunlight energy and incorporate it into organic compounds. All other organisms in an ecosystem are consumers (heterotrophs) that depend on energy stored in the tissues of producers. energy input from sun Producers Nutrient Cycling Consumers energy lost (mainly heat) Fig 25.4, p. 456 Feeding Levels and Food Webs • • • • Herbivores eat plants (primary consumers). Carnivores eat animals (secondary or tertiary consumers). Omnivores eat a variety of organisms. Decomposers include fungi, bacteria, and small invertebrates that extract energy from the remains or products of organisms. Ecosystems require energy and nutrient input to continue to function. Energy is generally lost from the system as heat; some nutrients can also be lost. Animation: Energy Flow CLICK TO PLAY Animation: The Role of Organisms in an Ecosystem CLICK TO PLAY Feeding Levels and Food Webs Energy moves through a series of ecosystem feeding levels. Trophic levels (feeding levels) represent a hierarchy of energy transfers. Level 1 (closest to the energy source) consists of primary producers, level 2 is composed of herbivores, and levels 3 and above are carnivores. Decomposers and omnivores such as humans feed on organisms from all levels. Animation: Trophic Levels in a Simple Food Chain CLICK TO PLAY Animation: Prairie Trophic Levels CLICK TO PLAY Feeding Levels and Food Webs Food chains and webs show who eats whom. A linear sequence of who eats whom in an ecosystem is called a food chain; simple chains are rarely found in nature. Cross-connecting food chains make up food webs in which the same food resource is often part of more than one food chain. Marsh Hawk Upland Sandpiper Garter Snake Cutworm Plants In-text Fig, p. 466 Marsh Hawk Upland Sandpiper Garter Snake Cutworm Plants Stepped Art In-text Fig, p. 466 Animation: Categories of Food Webs CLICK TO PLAY Animation: Prairie Food Web CLICK TO PLAY Marsh Hawk Higher Feeding Levels A variety of carnivores, omnivores, and other consumers. Many feed at more than one level all the time, seasonally, or when an opportunity presents itself Crow Upland Sandpiper Garter Snake Frog Weasel Spider Second Feeding Level Primary consumers (herbivores) Badger Ground Squirrel Clay-colored Sparrow Earthworms, Insects (e.g.) Grasshoppers, Cutworms First Feeding Level Primary producers © 2007 Thomson Higher Education Coyote Prairie Vole Pocket Gopher Grass Fig 25.5, p. 467 Animation: Rainforest Food Web CLICK TO PLAY Section 3 Energy Flow through Ecosystems Energy Flow through Ecosystems Producers capture and store energy. Primary productivity is the total rate of photosynthesis (trapping of energy) for the ecosystem during a specified interval. How much energy is trapped depends on many factors. • • The number of individual plants and the relative balance between trapping energy and expending energy to produce new plants. Environmental factors such as availability of mineral nutrients, rain fall, and temperature. Earth’s Primary Productivity Figure 25.6 Energy Flow through Ecosystems Consumers subtract energy from ecosystems. An ecological pyramid describes the energy relationships in an ecosystem. • • Primary producers form the base. Successive tiers of consumers are found above them. Energy Flow through Ecosystems Ecological pyramids can be of two basic types: • • Biomass is the combined weight of all of an ecosystem’s organisms at each level of the pyramid; a biomass pyramid can be “right-side up,” with producers outnumbering consumers, or “upsidedown,” which is the opposite. An energy pyramid reflects the trophic structure more accurately because it is based on energy losses at each level; energy pyramids are always “right-side up.” Figure 25.7a 1.5 third-level carnivores (gar, large-mouth bass) 11 second-level consumers (fishes, invertebrates) 37 first-level consumers (herbivorous fishes, turtles, invertebrates) primary producers (algae, eelgrass, rooted plants) 5 809 decomposers (bacteria, crayfish) Fig 25.7a, p.468 1.5 third-level carnivores (gar, large-mouth bass) 11 second-level consumers (fishes, invertebrates) 37 first-level consumers (herbivorous fishes, turtles, invertebrates) primary producers (algae, eelgrass, rooted plants) 5 809 decomposers (bacteria, crayfish) Stepped Art Fig 25.7a, p.468 top carnivores carnivores herbivores producers 21 Decomposers = 5,060 383 3,368 20,810 Fig 25.7b Section 4 Biogeochemical Cycles— An Overview Biogeochemical Cycles Biogeochemical cycles describe the movement of nutrients from the environment to organisms and then back to the environment that serves as a reservoir for them. Cycling is slowest through the reservoir. The amount of nutrient being recycled through major ecosystems is greater than the amount entering or leaving in a given year. Inputs to an ecosystem’s nutrient reserves are by precipitation, metabolism, and rock weathering; outputs include losses by runoff. nutrient reservoirs in environment fraction available to ecosystem geochemical cycle primary producers consumers (herbivores, carnivores, parasites) decomposers Fig 25.8, p. 469 Biogeochemical Cycles There are three categories of biogeochemical cycles: In the global water cycle, oxygen and hydrogen move as water molecules. In the atmospheric cycles, elements such as carbon and nitrogen move in gaseous phase. In sedimentary cycles, solid, non-gaseous nutrients move from land to the seafloor and back to land through geological uplifting; this is a very slow cycle. Section 5 The Water Cycle The Water Cycle The hydrologic cycle (water cycle) encompasses water in the oceans, atmosphere, and land. The ocean serves as the main water reservoir. Evaporation moves water into the lower atmosphere where it returns to Earth as precipitation. Animation: Hydrologic Cycle CLICK TO PLAY atmosphere evaporation from ocean 425,000 wind-driven water vapor 40,000 evaporation from land plants precipitation (transpiration) into ocean 71,000 385,000 precipitation onto land 111,000 surface and groundwater flow 40,000 ocean land Fig 25.9, p. 470 The Water Cycle Water moves nutrients in or out of ecosystems. A watershed funnels rain or snow into a single river. Plants absorb nutrients to prevent their loss by leaching. Section 6 Cycling Chemicals from the Earth’s Crust Cycling Chemicals from the Earth’s Crust There are two phases in the phosphorus cycle: In the geochemical phase, phosphorus moves from land to sediments in the seas and back to the land over long periods of time. In the much more rapid ecosystem phase, plants take up the phosphorus from the soil; it is then transferred to herbivores and carnivores, which excrete it in wastes and their own decomposing bodies thus returning the phosphorus to plants. Cycling Chemicals from the Earth’s Crust Excessive phosphorus compounds in runoff water can lead to eutrophication of lakes and streams, characterized by explosive growth of algae and weeds. Animation: Phosphorus Cycle CLICK TO PLAY mining excretion Fertilizer Guano agriculture uptake by autotrophs Marine Food Webs weathering Dissolved in Ocean Water uptake by autotrophs weathering death, decomposition sedimentation Land Food Webs death, decomposition settling out Marine Sediments Dissolved in Soil Water, Lakes, Rivers leaching, runoff Rocks uplifting over geologic time © 2007 Thomson Higher Education Fig 25.10, p. 471 Section 7 The Carbon Cycle The Carbon Cycle In the carbon cycle, sediments and rocks hold most of the carbon; carbon moves also through the oceans, soil, atmosphere, and biomass. Carbon enters the atmosphere as CO2 produced by aerobic respiration, fossil-fuel burning, and volcanic eruptions. The Carbon Cycle Carbon in the ocean occurs as bicarbonate and carbonate; carbon dioxide in the ocean is carried to deep storage reservoirs on the seafloor. Figure 25.12 Cold, salty, deep current Fig 25.12, p. 473 The Carbon Cycle Carbon is removed from the atmosphere and the ocean by photosynthesizers and shelled organisms; carbon is held for different periods of time in different ecosystems. Decomposition of buried carbon compounds millions of years ago caused the formation of fossil fuels (natural gas, petroleum, and coal). Burning of fossil fuels puts extra amounts of carbon dioxide into the atmosphere, an occurrence that may lead to global warming. Animation: Carbon Cycle CLICK TO PLAY The Carbon Cycle Figure 25.11 diffusion between atmosphere and ocean Bicarbonate and Carbonate Dissolved in Ocean Water photosynthesis combustion of fossil fuels Terrestrial Rocks aerobic respiration Marine Food Webs, producers consumers, decomposers incorporation death, into sediments sedimentation uplifting over geologic time sedimentation Marine Sediments, Including Formations with Fossil Fuels Soil Water (dissolved carbon) leaching, runoff Fig. 25.11a, p. 472 Atmosphere combustion of fossil fuels photosynthesis aerobic respiration combustion of wood (for cleaning land; or for fuel) sedimentation Land Food Webs producers, consumers, decomposers death, burial, compaction over geologic time ©2007 Thomson Higher Education Peat, Fossil Fuels Fig. 25.11b, p. 473 Section 8 Global Warming Global Warming The greenhouse effect. Molecules of gases such as carbon dioxide, water, ozone, and others act like a pane of glass over the surface of the Earth. Figure 25.14 Animation: Greenhouse Gases CLICK TO PLAY Concentration (parts per billion) 380 Carbon dioxide 360 340 320 300 1960 1965 1970 1975 1980 1985 1990 1995 Time (years) © 2007 Thomson Higher Education Fig 25.14a2, p. 475 Concentration (parts per trillion) 1200 CFCs 1000 800 600 400 200 1976 1980 1985 1990 1995 1998 Time (years) © 2007 Thomson Higher Education Fig. 25.14b, p. 475 1.80 Concentration (parts per billion) Methane 1.70 1.60 1.50 1.40 1976 1980 1985 1990 1995 1998 Time (years) © 2007 Thomson Higher Education Fig. 25.14c, p. 475 Concentration (parts per billion) 320 Nitrous oxide (N2O) 310 300 290 280 270 260 1976 1980 1985 1990 1995 1998 Time (years) © 2007 Thomson Higher Education Fig. 25.14d, p. 475 Global Warming Wavelengths of visible light easily pass downward to Earth, but infrared wavelengths— heat—are impeded from passing back into space. The warming of the lower atmosphere is called the greenhouse effect. Figure 25.13 Rays of sunlight penetrate the lower atmosphere and warm the earth’s surface. a Surface radiates heat (infrared wavelengths) to the lower atmosphere. Some heat escapes into space. But greenhouse gases and water vapor absorb some infrared energy and radiate a portion of it back toward earth. b Increased concentrations of greenhouse gases trap more heat near Earth’s surface. Sea surface temperature rises, more water evaporates into the atmosphere, and Earth’s surface temperature rises. c Fig 25.13, p. 474 Animation: Greenhouse Effect CLICK TO PLAY Global Warming Global warming. Concentrations of greenhouse gases are increasing and may be at the highest levels they have been at for 420,000 to 20 million years. The result is a long-term rise in temperature— global warming; irreversible climate changes are already underway, such as melting of the polar ice caps and retreating of glaciers. Animation: Carbon Dioxide and Temperature CLICK TO PLAY Global Temperature Figure 25.15 Video: Kyoto Protocol This video clip is available in CNN Today Videos for Environmental Science, 2004, Volume VII. Instructors, contact your local sales representative to order this volume, while supplies last. Section 9 The Nitrogen Cycle The Nitrogen Cycle Gaseous nitrogen (N2) makes up about 80% of the atmosphere, which is the largest reservoir; this form of nitrogen can only be brought into the nitrogen cycle by certain species of bacteria. In nitrogen fixation, bacteria convert N2 to ammonia, which is then used in the synthesis of proteins and nucleic acids to be assimilated into plant, then animal, tissues. The Nitrogen Cycle Decomposition of dead nitrogen fixers releases nitrogen-containing compounds. Nitrification is a type of chemosynthesis where ammonia and ammonium ions are converted to nitrite; nitrite is turned to nitrates by bacteria for uptake by plants. Denitrification is the release of nitrogen gas to the atmosphere by the action of bacteria. Nitrogen can be lost from ecosystems through leaching. Animation: Nitrogen Cycle CLICK TO PLAY Consumers Nitrogen Fixation by industry for agriculture Nitrogen gas in Atmosphere Food Webs on Land Fertilizers Nitrogen Fixation by bacteria Ammonia, Ammonium in Soil loss by leaching uptake by autotrophs excretion, death, decomposition uptake by autotrophs Nitrogen-rich wastes, Remains in soil Nitrate in Soil Ammonification bacteria, fungi convert residues to NH3; this dissolves to form NH4 Nitrification Loss by Denitrification Nitrification Nitrate in Soil loss by leaching © 2007 Thomson Higher Education Fig 25.16, p. 476 Section 10 Biological Magnification Biological Magnification DDT is a synthetic organic pesticide that was first used during World War II in the fight against malaria and typhus; after the war it continued to be used as a pesticide to control agricultural and forest pests. DDT is insoluble in water, but it is fat soluble. Vapor forms and small particles in water can carry DDT through an environment; from the environment it can be absorbed into tissues. Biological Magnification Biological magnification describes the increased concentration of slowly degradable substances in organisms as it is passed upward in a food chain. Each organism in a chain essentially assumes the absorbed DDT in each organism it feeds on lower in the chain. With DDT, organisms at the very top of the food chain, such as bald eagles and other predatory birds, suffered the most and some were pushed almost to extinction. Biological Magnification DDT is banned in the US, but this is not true outside of the US. Figure 25.18 DDT Residues (ppm wet weight of whole live organism) Ring-billed gull fledgling (Larus delawarensis) Herring gull (Larus argentatus) Osprey (Pandion haliaetus) Green heron (Butorides virescens) Atlantic needlefish (Strongylura marina) Summer flounder (Paralychthys dentatus) Sheepshead minnow (Cyprinodon variegatus) Hard clam (Mercenaria mercenaria) Marsh grass shoots (Spartina patens) Flying insects (mostly flies) Mud snail (Nassarius obsoletus) Shrimps (composite of several samples) Green alga (Cladophora gracilis) Plankton (mostly zooplankton) Water 75.5 18.5 13.8 3.57 2.07 1.28 0.94 0.42 0.33 0.30 0.26 0.16 0.083 0.040 0.00005 Data for a Long Island, NY, estuary in 1967 Fig 25.17a, p. 477 Animation: Pesticide Examples CLICK TO PLAY Section 11 Human Population Growth Human Population Growth The human population is growing rapidly. The world population reached the 6.3 billion mark in 2004. • • It took 2.5 million years for the world’s human population to reach 1 billion. It took less than 200 years for it to reach 6 billion. The growth rate is determined mainly by the balance between births and deaths. • • The total fertility rate (TFR) is the average number of children born to a woman. Many developed countries have a TFR at or below 2.1 (replacement rate), but some developing countries have a TFR two or three times this rate. 6 Estimated size by 10,000 years ago 5 million 5 By 1904 By 1927 By 1960 By 1974 By 1987 By 1999 Projected for 2050 4 1 billion 2 billion 3 billion 4 billion 5 billion 6 billion 8.9 billion 3 2 domestication of plants, animals 9000B.C. (about 11,000 years ago) 14,000 12,000 10,000 8,000 beginning of industrial, scientific revolutions agriculturally based urban societies 6,000 4,000 2,000 BC AD 1 2,000 © 2007 Thomson Higher Education Fig. 25.19, p. 478 Animation: Exponential Growth CLICK TO PLAY Human Population Growth Population statistics help predict growth. Demographics influence a population’s growth and impact on ecosystems. • • • Population size is the number of individuals in the population’s gene pool. Population density is the number of individuals per unit of area or volume. Population distribution refers to the general pattern in which the population members are distributed, such as clustering in towns or cities. 292 million population in 2003 177 million 134 million population in 2050 (projected) 351 million 211 million 206 million population under age 15 population above age 65 21% 30% 44% 13% Gold: U.S. 6% Brown: Brazil 3% 2.0 2.2 total fertility rate Ivory: Nigeria 5.8 infant mortality rate 6.9 per 1,000 births 33 per 1,000 births 75 per 1,000 births 77 years 69 years life expectancy 52 years per capita income in 2001 $34,280 $7,070 $800 © 2007 Thomson Higher Education Fig. 25.20a, p. 479 Animation: Current and Projected Population Sizes by Region CLICK TO PLAY Human Population Growth Age structure defines the relative proportions of individuals of each age. • • The three categories are: prereproductive, reproductive, and postreproductive. The reproductive base (prereproductive and reproductive members) for a human population will determine the future growth rate of a population. Figure 25.21 UNITED STATES INDIA Fig 25.21, p. 479 Animation: Age Structure Diagrams CLICK TO PLAY Animation: U.S. Age Structure CLICK TO PLAY Section 12 Nature’s Controls on Population Growth Nature’s Controls on Population Growth The human population has been growing exponentially since the mid-1700s. There is a limit on how many people the Earth can sustain. The biotic potential of a population is its maximum rate of increase under ideal— nonlimiting—conditions. Limiting factors on population growth could include any essential resource that is in short supply such as food, water, or living space; predation and pathogens can also be limiting. Nature’s Controls on Population Growth The number of individuals that can be sustained by the resources in a given area is the carrying capacity. The carrying capacity can vary over time and is expressed graphically in the S-shaped curve pattern called logistic growth. Figure 25.22 Number of individuals initial carrying capacity TIME A new carrying capacity B C D E Fig 25.22, p. 480 Animation: Demographic Transition Model CLICK TO PLAY Nature’s Controls on Population Growth Some natural population controls are related to population density. Density-dependent controls (such as diseases) are limiting factors that exert their effects with respect to the number of individuals present. Density-independent controls, such as natural disasters, tend to increase the death rate without respect to the number of individuals present. Video: People Explosion CLICK TO PLAY From ABC News, Human Biology in the Headlines, 2006 DVD. Section 13 Assaults on Our Air Assaults on Our Air Pollutants are substances that adversely affect health, activities, or survival of a population. Air pollutants include oxides of carbon, sulfur, and nitrogen as well as CFCs. Over 700,000 metric tons of pollutants are released into the atmosphere every day in the United States alone. Assaults on Our Air Pollutants may be trapped in the atmosphere to produce two types of smog: • • Industrial smog is gray air found in industrial cities that burn fossil fuels. Photochemical smog is brown air found in large cities in warm climates; for example, gases from car exhaust. Burning of fossil fuels produces oxide particles that can fall to the earth as acid rain. Animation: Thermal Inversion and Smog CLICK TO PLAY Animation: Formation of Photochemical Smog CLICK TO PLAY Animation: Acid Deposition CLICK TO PLAY Animation: Effect of Air Pollution in Forests CLICK TO PLAY Assaults on Our Air The ozone layer has been damaged. Ozone (O3) in the lower stratosphere absorbs most of the ultraviolet radiation from the sun. • • Ozone thinning has produced an ozone hole over Antarctica; in 2001 an ozone hole appeared over the Arctic. Chlorofluorocarbons (CFCs) seem to be the cause— one chlorine atom can destroy 10,000 molecules of ozone. Assaults on Our Air While most CFC production is being phased out, it will take 100 to 200 years for the ozone layer to fully recover once all production and use ceases. Figure 25.23 Animation: How CFCs Destroy Ozone CLICK TO PLAY Video: Clean Air Act CLICK TO PLAY From ABC News, Environmental Science in the Headlines, 2005 DVD. Section 14 Water, Wastes, and Other Problems Water, Wastes, and Other Problems Problems with water are serious. Three out of four humans do not have enough clean water to meet basic needs. About one third of all food is raised on irrigated land, leading to salt buildup (salinization) and depletion of ground water systems. Figure 25.24 Water, Wastes, and Other Problems Humans waste limited water supplies and pollute much of the remaining water through agricultural and industrial runoff; even garbage and debris is dumped into our waterways. Animation: Stream Pollution CLICK TO PLAY Animation: Threats to Aquifers CLICK TO PLAY Water, Wastes, and Other Problems Where will we put solid wastes and produce food? Finding enough space to bury our wastes is becoming a problem, and the dump sites can leak toxic materials into the soil and water. Almost one quarter of all the land on Earth is used for agriculture. • • The green revolution has increased crop yields but uses many times more energy and mineral resources. Large-scale desertification is caused by overgrazing on marginal lands. Water, Wastes, and Other Problems Deforestation has global repercussions. Deforestation, the removal of all trees from large tracts of land, can reduce fertility, change rainfall patterns, increase temperatures, and increase carbon dioxide levels. Water, Wastes, and Other Problems Clearing large tracts of tropical forests may have global repercussions due to leaching and shifting rates of evaporation and sunlight penetration. Figure 25.25 Animation: Effects of Deforestation CLICK TO PLAY Video: Desertification in China CLICK TO PLAY From ABC News, Environmental Science in the Headlines, 2005 DVD. Section 15 Concerns about Energy Concerns about Energy The Earth has abundant energy, but the net amount of energy left after subtracting the energy it costs to find, process, and deliver this energy is relatively small. Some forms of energy are renewable, such as solar energy; coal and petroleum are examples of non-renewable energy. Figure 25.26a Concerns about Energy People in developed countries use far more energy per person than those in developing countries. Figure 25.26b Animation: Energy Use CLICK TO PLAY Concerns about Energy Fossil fuels are going fast. Fossil fuels include oil, coal, and natural gas; these sources represent the fossilized remains of ancient forests and organisms. Petroleum and natural gas reserves may be depleted during this century. Extraction and use of abundant reserves of coal are not environmentally attractive. Concerns about Energy Can other energy sources meet the need? Nuclear power can produce electricity at relatively low cost, but there are risks. • • Meltdowns may release large amounts of radioactivity to the environment. Waste is so radioactive that it must be isolated for 10,000 years. Solar energy can be converted to the mechanical energy of wind to run turbines; solar cells could be used to generate electricity for producing hydrogen gas. Concerns about Energy Hybrid cars are currently available, which work on a combination of gasoline and the electricity from batteries. Fusion power has potential, but many obstacles make the technology a distant possibility. Video: Nuclear Energy CLICK TO PLAY From ABC News, Environmental Science in the Headlines, 2005 DVD. Section 16 Loss of Biodiversity Loss of Biodiversity Humans have become a major factor in the premature extinction of more and more species. Extinction is irreversible and greatly decreases biodiversity. Speciation cannot balance rapid extinction. Animation: Humans Affect Biodiversity CLICK TO PLAY Animation: Biodiversity Hot Spots CLICK TO PLAY Loss of Biodiversity Tropical deforestation is the greatest source of extinction of species, followed closely by destruction of coral reefs. Loss of plant diversity directly hurts consumers by removing an important part of every food chain; plant loss also affects our sources of natural medicines. The underlying causes of such destruction are human population growth and poor economic policies. Animation: Habitat Loss and Fragmentation CLICK TO PLAY 1620 1850 1850 (pocket only) 1990 Fig 25.29, p. 487 Animation: Resource Depletion and Degradation CLICK TO PLAY Loss of Biodiversity To end the trend, we must collectively fight to reduce deforestation, global warming, ozone depletion, and poverty. Figures 25.27 and 25.28