* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download SCIENCE
Negative mass wikipedia , lookup
Standard Model wikipedia , lookup
Fundamental interaction wikipedia , lookup
Electrical resistivity and conductivity wikipedia , lookup
Electric charge wikipedia , lookup
Nuclear structure wikipedia , lookup
Condensed matter physics wikipedia , lookup
Elementary particle wikipedia , lookup
History of subatomic physics wikipedia , lookup
Nuclear physics wikipedia , lookup
State of matter wikipedia , lookup
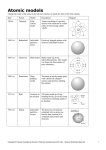
EVERYTHING YOU NEED TO ACE SCIENCE IN ONE BIG FAT NOTEBOOK Flexibound paperback 5⅞" x 8" • 528 pages $14.95 U.S. • Higher in Canada 978-0-7611-6095-3 • No. 16095 Coming August 2016 Chapter 6 ATOMIC MODELS Atoms are made of smaller particles: MATTER, PROPERTIES, AND PHASES MATTER and ATOMS MATTER describes everything that we can see, touch, smell, or feel. In other words, mat ter is anything that has mass and takes up space (including air and almost everything else). The smallest unit of matter is called an ATOM. If you chop a piece of metal into a bajillion pieces, the smallest bit you are left with that still has the properties of PROTONS (positively charged particles) NEUTRONS (electrically neutral particles, which means it has no charge), ELECTRONS (negatively charged particles with almost no mass) matter anything that has mass and takes up space atom nucleus the smallest unit of matter The word "a tom" is derived from a Greek word that means “cannot be divided.” the metal is called an atom. the center of an atom, formed by protons and neutrons Protons and neutrons stick together to form the center of an atom, called the NUCLEUS, which has a net positive charge. Electrons (and the Greeks didn't even have a particle accelerator!) 60 (Remember a “model” is a way to represent something th at we can’t easily see.) orbit, or circle around, the nucleus, but too quickly to The MODERN ATOMIC MODEL shows an ELECTRON CLOUD rather than individual electrons like the model above to demonstrate where you’re most likely to find an orbiting electron. Denser area of the cloud means higher probability of electrons. pinpoint their exact locations. 61 Brief History of the Atomic Models COLOR JOHN DALTON was the first scientist to propose that elements SIZE DENSITY MALLEABILITY (how easily something are composed of indestructible atoms. He thought that there can be flattened, shaped, or pressed) were particles so small that we could not see them. He called MAGNETISM (whether or not something is magnetic) these particles atoms, and his theory on matter was known as the ATOMIC THEORY OF MATTER. BOILING POINT and MELTING POINT (the temperature at which something boils or melts) SIR JOSEPH JOHN (J. J.) THOMSON discovered the presence of negatively charged particles SOLUBILITY (how easily something (electrons) in atoms and pictured them dissolves into another substance) embedded with positively charged particles, kind of like raisins in oatmeal-raisin cookies. IS: HELLO MY NAME ERN EST HELLO MY NAM E IS: J. J. A PHYSICAL CHANGE is any change to the physical properties of mat ter such as its size, shape, or state (solid, liquid, or ERNEST RUTHERFORD worked out that atoms had small and gas/vapor). The final product of any physical change is still heavy positively charged centers, which he called a nucleus. He composed of the same matter. For example, you can revert ice, figured out that electrons were orbiting the nucleus in mostly snow, or vapor back to water by either heating or cooling them. empty space. He called the positive particles in the nucleus Ice, vapor, and water are all H 20-just in different states. protons. Rutherford’s student SIR JAMES CHADWICK proposed the existence of uncharged particles in the nucleus, which he CHEMICAL PROPERTIES describe the ability of something called neutrons. to undergo different chemical changes. PHYSICAL and CHEMICAL PROPERTIES and CHANGES The way something looks, feels, smells, and tastes are all Some examples of chemical properties: FLAMMABILITY (how easily something lights on fire) PHYSICAL PROPERTIES. It's easy to classify matter by these characteristics. Some common physical properties used to REACTIVITY (how reactive something differentiate matter are: is to oxygen, water, light, etc.) 62 63 1 1 1 H Hydrogen 1.0078 3 2 3 4 5 6 7 Li period group 2 4 Be 3 Li 11 12 Sodium 22.990 Ma g nesium 24.305 3 4 5 6 19 20 21 22 23 Potassium 39.098 Calcium 40.078 Scandium 44.956 Titanium 47.867 37 38 39 Rubidium 85.468 Strontium 87.62 Yttrium 88.906 55 56 Caesium 132.91 Barium 137.33 87 Fr group period Francium ( 223 ) Lithium 6.941 72 16 17 18 S Cl Ar Aluminum 26.982 Silicon 28.084 Phos phorus 30.974 Sulfur 32.059 Chlorine 35.446 Arg on 39.948 34 35 36 Gallium 69.723 Germanium 72.63 Arsenic 74.922 Selenium 78.96 Bromine 79.904 Kr ypton 83.798 45 46 47 48 49 50 51 52 53 Rhodium 102.91 Palladium 106.42 Silver 107.87 Cadmium 112.41 Indium 114.82 Tin 118.71 Antimony 121.76 Tellurium 127.60 I 54 Iodine 126.90 Xe 77 78 79 80 81 82 83 84 85 86 Iridium 1.0078 Platinum 195.08 Gold 196.97 Mercur y 200.59 Thallium 204.38 Lead 207.2 Bismuth 208.98 Polonium ( 209 ) Astatine ( 210 ) Radon ( 222 ) 108 109 110 111 112 113 114 115 116 117 118 Bohrium ( 264 ) Hassium ( 269 ) Meitnerium ( 226 ) Ununtrium ( 268 ) Flerovium ( 268 ) 60 61 62 Promethium ( 145 ) 44 Zirconium 91.224 Niobium 92.906 Moly bdenum 95.96 Technetium 98.9062 Ruthenium 101.07 72 73 74 75 76 Hafnium 178.49 Tantalum 180.95 Tungsten 183.84 Rhenium 186.21 Osmium 190.23 88 104 105 106 107 Radium ( 226 ) Rutherfordium ( 261 ) Dubnium ( 262 ) Seaborg ium ( 266 ) 57 58 59 Lanthanum 138.91 Cerium 140.12 89 90 Actinium ( 226 ) Thorium 1.0078 Zr Nb Mo Tc Ta W Re Ru Os Co Rh Ir 92 U Uranium 238.03 Pd A g Cd In Pt Au Hg Tl Sn Sb Pb Bi Se Te Br Kr Xenon 131.29 Po At Rn Fl Uup Lv Uus Uuo Rg Cn Uut 63 64 65 66 67 68 69 70 Samarium 150.36 Euro p ium 151.96 Gadolinium 157.25 Terbium 158.93 Dys p rosium 162.50 Holmium 164.93 Erbium 167.26 Thulium 168.93 Ytterbium 173.04 Lutetium 174.97 93 94 95 96 97 98 99 100 101 102 103 Neptunium ( 237 ) Plutonium ( 244 ) Americium ( 243 ) Curium ( 247 ) Berkelium ( 247 ) Fermium ( 257 ) Mendelevium ( 258 ) Nobelium ( 259 ) Lawrencium ( 262 ) Pr Nd Pm Sm Praseody mium Neody mium 140.91 144.24 Ni Cu Zn Ga Ge As Ds Rf Db S g Bh Hs Mt Pa 15 Zinc 65.38 43 Protactinium 231.04 14 Co pp er 63.546 42 Ac Th 13 Neon 20.180 Nickel 58.693 Cr Mn Fe 91 Ne Cobalt 58.933 41 La Ce F 33 40 Ra Fluorine 18.998 32 Iron 55.845 Hf Ox yg en 15.999 31 Man g anese 54.938 Y Nitrog en 14.006 P O 10 Carbon 12.009 Si N Helium 4.0026 30 Chromium 51.996 V 17 9 29 Vanadium 50.942 Ti 16 8 Boron 10.806 Al C 15 7 28 24 Ca Sc B 14 6 He 12 9 27 10 13 5 18 2 11 8 26 Cs Ba → NEW AN D PEN DING DISCOVERIES → OTHER NONMETALS 7 25 Rb Sr → LANTHANIDES → ACTINIDES → POST-TRANSITION METALS → METALLOIDS Ber y llium 9.0122 K → HALOGENS → NOBLE GASES → TRANSITION METALS → UNKNOWN PROPERTIES Lithium 6.941 Na M g → ALKALAI METALS → ALKALINE EARTH METALS Np Pu Darmstadtium Roentg enium Co p ernicium ( 268 ) ( 268 ) ( 268 ) Unun p entium Livermorium Ununseptium ( 268 ) ( 268 ) ( 268 ) Eu Gd Tb Dy Ho Er Tm Yb Am Cm Bk Cf Californium ( 251 ) Es Einsteinium ( 252 ) Ununoctium ( 268 ) 71 Lu Fm Md No Lr 73 solution There are two kinds of mixtures: a homogenous mixture 1. Heterogeneous mixture: a mixture where the substances aren’t evenly mixed. A salad is an example of a heterogeneous mixture; every bite of a salad is different no matter how many times you mix the salad. HETERO is Greek for “ different,” so the mixture has different parts; it’s not all the same. 2. Homogenous mixture: a mixture where the molecules of each substance are equally mixed, and you can’t see the different parts of the mixture. Sugar that has been dissolved in water creates a homogenous mixture; you can’t see the sugar and the water, just a liquid that contains molecules of both. HOMO is Greek for “same,” so the mixture is the same throu g hout. solvent the substance into which the solute dissolves SOLUTIONS. A solution is made of a SOLUTE and a SOLVENT. A solute is the substance SOLUBILITY is the ability of a substance to dissolve into another substance. Lots of things affect solubility: Temperature is one factor: Usually solid solutes are more soluble in water at higher temperatures, which is why it is easier to dissolve sugar in hot water. SOLUTE SOLV ENT that gets dissolved, and the solvent is the substance that dissolves the solute. For example, some sports drinks are a solution that is made of water and powdered sports drink mix. 84 the substance that is dissolved into the other substance SOLUBILITY Sometimes homogenous mixtures are called solute PRESSURE and the CONCENTRATION of other solvents in a solution also affect solubility. Gases can be dissolved into liq uid, too! Gas solutes, like carbonation, are the opposite. Gases are more soluble in liquids at colder temperatures. Carbonated beverages remain fizzy longer when they are cold because gas is more soluble in cold liquid. 85 Electrical Circuit Batteries An electric current will continuously flow if the charges can Bat teries provide the energy source that pushes the electric travel in a closed conducting loop, called a CIRCUIT. The charges around a circuit. When connected to a circuit, electric field keeps the charge moving. a bat tery creates an electric field with a positive and negative terminal at each end of the bat tery (which is The components of a circuit are: electrical conductor, such as a wire, connects to the power source to form a closed loop (a connection with no openings or breaks) load (not necessary, but usually there), a device that the circuit is powering, like a lightbulb, fan, or speaker. the + or - sign you see on different sides of the bat tery). The electrons, which are the moving charges in a current, are at tracted to the positive terminal and repelled by the negative terminal. They travel like traffic on a road (as long as the circuit is a closed loop). Voltage The energy of flowing electrons in a circuit is called VOLTAGE. Voltage, measured in volts (V), is voltage the amount of potential energy an electron in a circuit can gain the electric potential difference between two points in a circuit, such as the positive and negative terminals of a battery. Voltage provides potential energy to an electron, just like gravity provides potential energy to a ball held above the ground The higher the voltage, the greater the potential difference, and the more energy power source of electrical energy, such as a battery the current can supply. So a 9-volt battery will make a small switch (not necessary, but frequently there) is a device to open and close a circuit lightbulb glow much brighter than a AA battery would (which is 1.5 volts). IT’S LIKE A DRAWBRID G E ON A ROAD. 164 165