* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download E - Purdue Physics
Renormalization wikipedia , lookup
Particle in a box wikipedia , lookup
Matter wave wikipedia , lookup
Atomic orbital wikipedia , lookup
Quantum key distribution wikipedia , lookup
Bohr–Einstein debates wikipedia , lookup
Double-slit experiment wikipedia , lookup
Tight binding wikipedia , lookup
Quantum electrodynamics wikipedia , lookup
X-ray photoelectron spectroscopy wikipedia , lookup
Rutherford backscattering spectrometry wikipedia , lookup
Franck–Condon principle wikipedia , lookup
Wheeler's delayed choice experiment wikipedia , lookup
Electron configuration wikipedia , lookup
Hydrogen atom wikipedia , lookup
Electron scattering wikipedia , lookup
Ultrafast laser spectroscopy wikipedia , lookup
Delayed choice quantum eraser wikipedia , lookup
Wave–particle duality wikipedia , lookup
Atomic theory wikipedia , lookup
Theoretical and experimental justification for the Schrödinger equation wikipedia , lookup
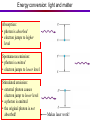
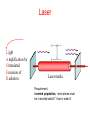
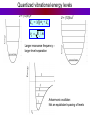
PHYS 172: Modern Mechanics Lecture 14 – Energy Quantization Summer 2012 Read 8.1-8.8 Quantization •Classical Physics: quantities are continuous. • Quantum Physics: Some quantities are limited to a discrete set of values. Example: charge, Q = N.e Quantum means quantized Answers come in whole numbers Example: The number of unopened Coke cans in your refrigerator is quantized. Quantum Waves are Quantized There are discrete vibrational modes (normal modes) 1D: One Dimension Violin string, jumprope 2D: Two Dimensions Modes of a drumhead, coffee sloshing in your mug http://demonstrations.wolfram.com/NormalModesOfACircularDrumHead/ 3D: Three Dimensions Electron Waves around Atomic Nuclei! http://www.daugerresearch.com/orbitals/index.shtml Higher Frequency = Higher Energy Photons Photons come in discrete particles, or packets of energy. One PHOTON = One packet of light And yet it's still a wave: = Wavelength (crest to crest) wavelength [m] frequency [1/s] speed of light [m/s] Number of wavelengths which go by per second Photons Photons come in discrete particles, or packets of energy. One PHOTON = One packet of light Photon energy and wavelength: E photon = hn light hc = llight Visible light Electromagnetic spectrum E = 3.1 eV ν = 7.5x1014 s-1 Wavelength 400 450 E = 1.8 eV ν = 4.2x1014 s-1 500 550 600 650 700 750 nm Atoms and Light Absorb a Photon 3S Absorb a Photon 2S 1S Adding a photon increases the energy of the atom Atoms and Light Release a Photon 3S 2S Releasing a photon decreases the energy of the atom Release a Photon 1S Atoms and Light QUANTUM MECHANICS says each ELEMENT (type of atom) can only have specific, QUANTIZED energies. Each atomic transition has a CHARACTERISTIC COLOR Photon Energy = Frequency = Color The Sun Dark lines correspond to specific atomic transitions, such as “1s to 2s in Hydrogen”, or “1s to 2p in Helium”. Hydrogen atom: electron energy 3S 2S 1S Emission spectra Hydrogen atom: Energy of emitted photon: 3S EN = K + U e = - hn = E 4 - E1 13.6 eV N2 2S Line spectrum – light is emitted at fixed frequencies 1S Absorption spectra Hydrogen atom: Energy of absorbed photon: EN = K + U e = 3S 13.6 eV N2 hn = E 2 - E1 Line spectrum – absorption at fixed frequencies 2S Different atoms – different energies Atomic spectra – signature of element Example: He was discovered on Sun first 1S Clicker Question Suppose that these are the quantized energy levels (K+U) for an atom. Initially the atom is in its ground state (symbolized by a dot). An electron with kinetic energy 6 eV collides with the atom and excites it. What is the remaining kinetic energy of the electron? A) 9 eV B) 6 eV C) 5 eV D) 3 eV E) 2 eV Effect of temperature Boltzmann constant: k=1.4×10-23J/K Population of level: ~ exp (- E / kT ) Temperature, K Energy of the level above the “ground state”, EN – E1 ~ exp (- E / kT ) = 10 -33 Population of levels for visible light transition (E = 2eV): At Room Temperature, 300K: On the Sun, 6000K: Energy conversion: light and matter Absorption: • photon is absorbed • electron jumps to higher level Spontaneous emission: • photon is emitted • electron jumps to lower level Stimulated emission: • external photon causes electron jump to lower level • a photon is emitted • the original photon is not absorbed! Makes laser work! Laser L ight A mplification by S timulated E mission of R adiation Laser media Requirement: inverted population, more atoms must be in excited state E’ than in state E. Quantizing two interacting atoms E Spring (harmonic oscillator) r U for two atoms If atoms don’t move too far from equilibrium, U looks like Uspring. Thus, energy levels should correspond to a quantized spring . . . Quantizing two interacting atoms Classical harmonic oscillator: Quantum harmonic oscillator: U = (1/2)kss2 U = (1/2)kss2 w0 = E2 = 2 w0 + E0 E1 = w0 + E0 E0 = 12 w 0 ks m ground state equidistant spacing 2 E = 12 mv 2 + 12 kx 2 = 12 kAmax Any value of A is allowed and any E is possible. w0 = k s / m h = h = 1.05 X10 34 J s 2 Energy levels: EN = N w 0 + 12 w 0 Yes, Tiny Harmonic Oscillators are Quantized Quantum harmonic oscillator: U = (1/2)kss2 w0 = E2 = 2 w0 + E0 E1 = w0 + E0 E0 = 12 w 0 ks m ground state equidistant spacing w0 = k s / m = h = 1.05 エ10 34 J s 2p Energy levels: EN = N w 0 + 12 w 0 WEB DEMO: http://web.ift.uib.no/AMOS/MOV/HO/ Quantized vibrational energy levels U = (1/2)ksx2 U = (1/2)ksx2 EN = N w 0 + E0 w0 = k s / m Larger resonance frequency – larger level separation Anharmonic oscillator: Not an equidistant spacing of levels Home study: Rotational energy levels (8.5,page 338) Nuclear & Hadronic energy levels (8.6) Comparison of energy level spacing (8.7) Laser Ruby: aluminum oxide crystal (sapphire) where some Al were replaced by Cr



























![L 35 Modern Physics [1]](http://s1.studyres.com/store/data/000572764_1-c4bf5ed66474525e3cf4981a43e1bbe1-150x150.png)


