* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Microbiotix has developed a pipeline of novel anti
Survey
Document related concepts
Antibiotics wikipedia , lookup
Traveler's diarrhea wikipedia , lookup
Middle East respiratory syndrome wikipedia , lookup
West Nile fever wikipedia , lookup
Orthohantavirus wikipedia , lookup
Ebola virus disease wikipedia , lookup
Marburg virus disease wikipedia , lookup
Neonatal infection wikipedia , lookup
Influenza A virus wikipedia , lookup
Hospital-acquired infection wikipedia , lookup
Hepatitis C wikipedia , lookup
Henipavirus wikipedia , lookup
Human cytomegalovirus wikipedia , lookup
Lymphocytic choriomeningitis wikipedia , lookup
Potato virus Y wikipedia , lookup
Transcript
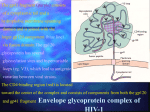
o VIROLOGY RESEARCH: Virus infection is a dynamic process involving a myriad of cellular and viral proteins. It starts with the attachment of the virus to the host cell and finishing with the release of the progeny virions from the cell. Microbiotix has two main targets for anti-viral drug discovery: (1). key viral enzymes polymerases and proteases that are required for the replication of viruses in host cells and (2) the viral entry process. Our pipeline includes programs for the treatment of human cytomegalovirus infection, respiratory syncytial virus, hepatitis C, and avian Influenza H5N1. Targeting of viral polymerase. Targeting the viral entry The first step, of a productive viral infection requires the binding of viral envelope proteins to specific cell surface receptors. This selective association between viral envelope proteins is a multistep process and requires highly specific sequential engagements between the viral envelope proteins and specific cell surface receptors and/or, coreceptors. Blocking viral entry into its target cell leads to suppression of viral infectivity, replication, and the cytotoxicity induced by virus-cell interaction. Respiratory Syncytial Virus (RSV) infections is the primary cause of hospitalisation in the first year of life for children in most parts of the world, and nearly 100% of children in the USA are infected with the virus by 2 to 3 years of age. The two viral envelope proteins include the fusion (F) protein and the attachment (G) protein which are essential for viral penetration and attachment to the host cells. Microbiotix has developed MBX300, sulfated sialyl lipid that inhibits RSV infection in vitro and in animal models. MBX300 acts on the G protein and inhibits the binding of virus to the cell membrane. MBX300 has also shown good activity against other RNA virus including hepatitis C virus in vitro. Therefore, the possibility exists that MBX300 may have broader activity and advantages against multiple viral pathogens. We are exploring the activity against other viruses. Microbiotix is planning to start IND enabling preclinical toxicology and safety pharmacology studies for MBX 300. Hepatitis C Virus (HCV) is recognized as a worldwide health problem affecting over 170 million people. HCV causes a spectrum of disease ranging from an asymptomatic carrier state to endstage liver disease; which includes cirrhosis and hepatocellular carcinoma. A vaccine for HCV is currently not available. The present therapies for HCV infection rarely result in viral clearance, are difficult to administer and cause serious toxic side effects. There are several drugs that have been developed and under trial for HCV infection. These drugs are targeting the HCV-encoded serine protease (NS3) and RNA polymerase (NS5B) that have emerged as a favourite target in the race for new anti-HCV drugs. Microbiotix has an active program to develop entry inhibitors against the HCV. However, unlike others we are targeting the viral entry process by attacking the selective interaction between viral envelope glycoproteins and specific cell-surface receptor(s). Since this virus is difficult to culture in vitro we have generated pseudotype virus expressing HCV envelope glycoproteins as surrogate model to screen for inhibitors that target the viral entry process. HCV, like HIV has a high mutation rate and therefore there will be high probability for the emergence of drug-resistant virus. Combination therapy, either with the existing treatments or with other specific antiviral drugs, will be needed to control the virus — a point that underscores the importance of continuing to develop inhibitors against various viral targets. Therefore the development of this entirely new class of inhibitors will have enormous impact on the treatment and management of chronic HCV infection. Furthermore, the FDA approval of the HIV fusion inhibitor T20 (enfuvirtide/Fuzeon) demonstrates the clinical feasibility of this anti-viral approach Recent outbreaks of highly pathogenic avian influenza A (H5N1) infections in poultry and in humans (through direct contact with infected birds), have highlighted the need to develop new anti-influenza therapeutics that will be active against all subtypes, including a newly emerged pandemic strain. Vaccines, the main strategy for protection against influenza epidemics, will not be effective against emerging strains not present in the vaccine. The currently available, antiinfluenza drugs, viral M2 ion-channel inhibitors (amantadine and its derivative) and neuraminidase (NA) inhibitors (oseltamivir and zanamivir) have limited success because of their strain specificity and emergence of drug resistance strains. We are developing new antiinfluenza therapeutics; that is targted against the highly conserved fusion and receptor binding domain of the envelope protein hemagglutinin (HA). A novel inhibitor targeting either of the conserved sites should be active against multiple subtypes, including a newly emerged pandemic strain. For drug discovery, we have generated a pseudotype virus expressing HA from pandemic H5N1 strain, as a surrogate model, to mimic HA mediated entry. This surrogate model system is being used to screen for inhibitors against HA of pathogenic avian influenza A (H5N1) virus under BSL2 conditions. BATERIOLOGY RESEARCH: Novel Antibiotic Targeting Bacterial DNA Polymerases The objective of this project is to develop novel antibiotic to treat antibiotic-resistant grampositive (Gr+) and gram-negative (Gr-) bacterial infections. The work utilizes the class III bacterial DNA polymerase (pol III), an unexploited target. The bacterial pol IIIs are highly conserved enzymes that are required for the synthesis of DNA during chromosomal replication. When an inhibitor of pol III is applied to a growing bacterium, it stops replication, and, thus, like the replication-specific quinolone antibiotics, it is bactericidal. Furthermore, the pol III-selective inhibitors are equally effective against clinically relevant antibiotic-resistant and antibioticsensitive pathogens. Using our proprietary antibiotic discovery platforms (Bacto-III and Replix) , we have developed a novel class of inhibitors active against DNA polymerase (pol) IIIC and E. The new family of compounds have displayed favorable properties (i.e. inhibition of Gr+ DNA polymerases as well as bacteria, low in vitro mammalian toxicity) and we are optimizing the most promising structure utilizing a rational drug design approach to develop antibacterial lead compounds Bacterial DNA Helicases: Targets for Novel Antibiotics We are developing new chemical classes of anti-bacterials that will target replicative DNA helicase (RDH), an essential target in the DNA replication pathway and use them to treat resistant organisms. For this purpose, we have developed a high throughput screen to identify inhibitors of the Staphylococcus aureus RDH, that will detect inhibitors of any of the multiple essential helicase functions, including strand unwinding, ATPase-coupled translocation, DNA binding, and protein-protein interactions. Novel Therapies for Staphylococcal BioFilm-Related Infections Biofilms are surface attached bacterial communities encased in a hydrated matrix of exopolysaccharide. In the body, infecting bacteria form biofilms on medical implants, such as indwelling catheters. In this biofilm mode of growth, they are resistant to antibiotics and attack by the body's immune system. Staphylococcal biofilms are the leading cause of hospital acquired implant-based infections, which result in approximately 30,000 deaths per year. S. epidermidis is the leading cause of these infections. The overall goal of this project is to discover drugs that selectively block the formation of staphylococcal biofilms. These drugs will be used to coat the surfaces of medical implants to prevent biofilm development when implants are placed in patients. Type III Secretion Inhibitors for Anti-Infective Therapy Pseudomonas aeruginosa is a common and extremely virulent cause of serious infections in immune- compromised/suppressed patients (e.g., HIV and cancer), cystic fibrosis patients, and those on mechanical ventilation or with burn wounds. Frequent antibiotic resistance and the highly virulent nature of P. aeruginosa make it deadlier than most other bacteria. New chemical classes of antibiotics acting on novel accessible targets are crucial for continued effective therapy against P. aeruginosa because such drugs will not be subject to existing resistance mechanisms. The strategy of this project is to develop new drugs by screening a diverse collection of synthetic and natural product compounds against extra-cellular targets that are critical for virulence. The type III secretion system (TTSS), dedicated to the secretion of toxins and their translocation into the cytoplasm of human cells has been validated as a clinically important target in P. aeruginosa. The goal of this project is to identify specific inhibitors of TTSS and to develop them into novel antibiotics for therapy against P. aeruginosa. BIODEFENSE RESEARCH: New anti-infectives based on novel chemical scaffolds are vital to the biodefense armory primarily because they will be effective against both natural and engineered resistant forms of bioterrorist microbes. Our research efforts are focused on the discovery and development of novel therapeutic agents for the treatment of Ebola Virus infection, and against Bacillus anthracis and Burkholderia pseudomalli Development of Entry Inhibitors Against Ebola Virus Infection: Ebola virus (EBOV) is an aggressive pathogen that causes highly lethal viral hemorrhagic fever syndrome. EBOV is believed to be indigenous to Africa and causes periodic outbreaks of severe viral hemorrhagic fevers in the continent, with a ~50-90% mortality rate in infected patients. It has been classified as a Category A bioweapons agent by the Centers for Disease Control and Prevention (CDC). Currently, there is no FDA approved vaccine or antiviral drug (not even an experimental one) that is effective against EBOV infections in humans. The rapid progression of EBOV infection also offers little opportunity to develop acquired immunity. Therefore, there is a critical need for development of effective therapies to respond to post-exposure prophylaxis during outbreaks of EBOV infection, and to counter potential acts of bioterrorism. We are trying to block the entry step of the EBOV infection. We have generated pseudotype virus expressing EBOV envelope glycoproteins as surrogate model to screen for inhibitors that target the viral entry process in BSL2 laboratory. Microbiotix is utilizing two main platforms for developing entry inhibitors against EBOV infection: 1. High-througput screening of library of small molecules to discover unique anti-EBOV drug candidate and 2. structure-based drug design targeting the cellular cysteine proteases Cathepsin B and Cathepsin L that are essential for EBOV infection. Sensing Biowarfare Agents by Surface Enhanced Raman The goal of this project is to develop a platform tool based on surface enhanced Raman scattering (SERS) microscopy for the simultaneous rapid detection and identification of a broad range of category A-C priority bacterial pathogens. The SERS microscopic diagnostic platform will provide rapid, reagentless, specific identification of species within minutes, and will be developed as a portable device for field use. We are developing a reference base for SERS spectra. We are also in process of developing rapid, robust methods of enriching bacteria from clinical samples for SERS and optimizing the SERS substrate performance. Development of Screens for Bacillus anthracis Targets The goal of this research is to discover and develop novel antibiotics effective against B. anthracis, including resistant forms of this organism, for biodefense. Our strategy is to screen for novel inhibitors against DNA polymerase III C (pol lII C) and topoisomerase IV (topo IV). Using non-pathogenic B. anthracis Sterne strain, we have developed a permeable-cell DNA replication pathway screen and is screening our library of small molecules to discover unique drug candidates against B. anthracis infections. Discovery of Burkholderia pseudomallei Therapeutics for Biodefense B. pseudomallei is a bioterrorist threat. With the best current therapies, lethality is typically as high as 40%. The overall goal of this application is the development of new drugs against this organism. We are exploiting the high sequence similarity between B. pseudomallei and its less virulent relative P. aeruginosa and is in process of develoing innovative screens for rapid, safe discovery of effective therapeutic agents. The two species are similar in genome size and composition, with nucleotide and amino acid sequence identities for many genes in the 50-70% range, and in their mechanisms of drug resistance.