* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Tn917 insertion site in the 2C4 mutant
Survey
Document related concepts
Transcript
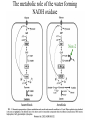
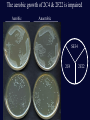
Fast Killing of C. elegans by Enterococcus faecium is mediated by Hydrogen Peroxide Terence Moy Lab meeting #2 February 25, 2003 What are Enterococci? -Gram-positive enteric bacteria -they are considered a “hardy” bacteria -2nd or 3rd leading source of nosocomial infections -causes endocarditis, bacteremia, urinary-tract infections, surgical wound infections -are intrinsically resistant to many antibiotics & they are capable of acquiring antibiotic resistance -increasingly, enterococci are becoming vancomycin-resistant - 80% of infections are caused by E. faecalis and 15-20% are caused by E. faecium - VREF (Vancomycin Resistant E. faecium): a larger % of clinical isolates are VRE compared to faecalis E. faecium that is grown anaerobically quickly kills C. elegans Killing Assay Protocol: 1. Grow E. faecium in liquid BHI media at 37° (5h-O/N) 2. Spread culture onto BHI plates ] ANAEROBIC Conditions 3. Incubate plates at 37° O/N in a GasPack 4. Remove plates from GasPack, cool to RT 5. Add C. elegans. Watch’em die. Atmospheric Air ] Atmospheric Air grown anaerobically Killing of C. elegans by E. faecium strain E7 100 90 80 Percent Killing 70 60 E7-1 50 E7-2 OG1RF-1 40 30 20 10 0 0 100 200 300 400 Time (minutes) Mobility decreased 500 600 Characteristics of E. faecium Fast Killing Growth under Anaerobic conditions leads to toxicity Killing efficiency depends on bacterial cell density, the bacterial strain, & the age of the nematode Toxicity can be inactivated by autoclaving the plate Toxicity does not required direct bacteria to worm contact; The toxin is diffusible. The toxin decreases worm mobility. Most worms die within 24 hours upon incubation with the toxin. Goal of my project: Identify the mechanism of fast killing. Approach: Transposon mutagenize E. faecium. Identify and characterize mutants produce more toxin or less toxin. Transposon Mutagenesis of E. faecium SE34 • plasmid pTV1OK was transformed into SE34 • pTV1OK contains Tn917 marked w/ Ermr, kanr on vector backbone, and a temperature sensitive origin of replication • SE34 w/ pTV1OK is maintained at 28° on BHI-kan 2 mg/ml, erm 50 µg/ml • Transposition is selected at 46-47° on BHI-erm 2 µg/ml • Transposition frequency is 4 10-4 • Qbot transferred approx. 13,000 colonies to 35- 382 well plates containing BHI-erm50+15% glycerol, cultures were grown O/N at 45°, and then stored at -80° Screening Method Bacteria: 1. liquid cultures were inculated by frogging frozen stocks and grown at 37° O/N under semi-aerobic conditions 2. saturated cultures were either frogged to 96 well plates or 20 µl of culture was spotted onto 24 well plates c BHI-erm50-agar, 3. Each strain was spotted onto two plates, one was incubated aerobically and the other anaerobically (in a gaspak) Worms: approx 500,000 L2 worms and 100,000 L4/young adults were used to screen a 382 well plate. Worms were resuspended in M9 buffer and pipetted onto each well. After ~ 2 hours, I examined the Anaerobically grown plates and I identified the bacteria that did not impair worm mobility. After ~ 5 hours, I examined the aerobically grown plates and identified the bacteria that impaired worm movement Results of the Screen 3,120 E. faecium clones were screened 3 strains were less toxic to C. elegans 2F22 3C23 11M12 1 strain was more toxic to C. elegans 2C4 Mutants are altered in C. elegans killing Strain Percent of Worms Alive after 24 hrs (number of worms) Bacterial Growth Condition Anaerobic Aerobic SE34 0% (n=102) 100% (92) 2C4 1% (102) 35%* (95) 2F22 31%* (99) 100% (95) E. faecalis VS583 100% (109) 99% (98) Tn917 insertion site in the 2C4 mutant Tn917 p1955 is an amino acid aminotransferase p1954 NADH peroxidase (npr), a 1379 bp gene Tn917 is inserted 16 bp upstream of the npr start codon p1953 is similar to the transposase of IS1077E, ISSmu1, & IS861 p1952 is a conserved hypothetical ORF of unknown function p1947 thru p1951 are ORFs encoded in the opposite direction of p1954 NADH peroxidase reduces hydrogen peroxide to water NADH+H+ + H2O2 Npr NAD+ + 2H2O Npr is a FAD containing reductase with a sulfonic acid derivative of cysteine at its active site In contrast, heme containing Catalase converts 2H2O2 to 2H2O and O2 (gas). E. faecalis possess a catalase while E. faecium does Not In E. faecalis, Npr activity is induced by aerobic growth and a putative OxyR binding site upstream of npr may regulate its expression Tn917 insertion site in the 2F22 mutant = 3C23 Tn917 p172 Fur family (Ferric Uptake Regulator) similar to B. subtilis PerR (peroxide stress response regulator) p173 NADH oxidase (nox) nox is a 1305 bp gene, Tn917 is inserted at position +925 bp in 2F22 p174 is a 300 bp hypothetical ORF with no homology to known genes p175 is a 567 bp ORF w/ homology to MarR & NapB p176 & p177 are putative tetracycline efflux pumps NADH Oxidase reduces oxygen to form water or hydrogen peroxide 2NADH+H+ + O2 NADH+H+ + O2 Nox-2 Nox-1 2NAD+ + 2H2O NAD+ + H2O2 Nox contains a cysteine-sulfenic acid redox center & a FAD A S. pneumoniae nox-2 mutant grows poorly in aerobic conditions unless glucose is present. This mutant & a S. mutans mutant are attenuated in mouse infection models. E. faecalis V583 has 3 nox genes. E. faecium DO has 7 nox-like genes The metabolic role of the water forming NADH oxidase Nox-2 The aerobic growth of 2C4 & 2F22 is impaired Aerobic Anaerobic SE34 2C4 2F22 Growth of the mutants under semi-aerobic conditions 1.00E+10 1.00E+09 CFU/ml 1.00E+08 1.00E+07 SE34 2C4 1.00E+06 2F22 1.00E+05 0 10 20 30 40 Time (hours) 50 60 70 80 Hydrogen Peroxide levels in culture supernatants Strain Culture conditions- (culture density in CFU/ml) Anaerobic to semi-aerobic aerobic Anaerobic aerobic (2hrs) SE34 BDL<30µM (1.1E9) 0.62 mM (8.8E8) BDL 0.35 mM (1.1E9) 2C4 0.22 mM (9.4E8) 1.71 mM (3.8E8) BDL 0.59 mM (1.5E9) 2F22 BDL 0.04 mM (2.2E8) BDL 0.12 mM (1.4E9) E. fs VS583 BDL BDL (3.8E9) BDL BDL (2.8E9) Killing of C. elegans by Hydrogen Peroxide killing curve from Jansen et al. (2002) Infect Immun 70:5202. Sources of Hydrogen Peroxide 1. NADH oxidase-1 2. Dismutation of superoxide 3. oxidoreductases of glucose, alcohols, malate, choline, amino acids, sugars, nucleosides, lactate, etc 4. -glycerophosphate oxidase (GlpO) Glycerol metabolism in E. faecalis M. Huycke Physiology of Enterococci Addition of Glucose to E. faecium cultures lowers Hydrogen peroxide concentrations Strain Liquid Culture conditions aerobic Anaerobic (o/n) to aerobic (2h) no glu +0.5% glu no glu +0.2% glu SE34 0.63 mM BDL (<50µM) 2C4 (npr) 1.8 mM BDL 2F22 (nox) BDL BDL 1.17 mM BDL (<0.3mM) Effect of Glycerol and Glucose of the toxicity of E. faecium towards C. elegans Glucose decreases the toxicity of E. faecium 0.5-1% glucose prevents the toxicity of E7 and 2C4 grown anaerobically & delays 2C4’s effect when grown aerobically Glycerol increases the toxicity of E. faecium (mobility assay): E7 and 2F22 become toxic when grown aerobically in the presence of 0.2% glycerol The onset of the toxicity from 2C4 grown aerobically occurs earlier in the presence of 0.02% glycerol Sucrose (1.7%) prevents toxicity of E7, delays 2C4’s effect In contrast, addition of sorbitol, DTT, DMSO, succinate, formate, or NaCl does not effect toxicity Catalase rescues E. faecium Toxicity *Culture supernatants from SE34 is toxic to C. elegans when the bacteria are grown aerobically but not when grown semi-aerobically Addition of 150 U/ml catalase rescues effect of faecium toxicity **qualification: fresh, untreated BHI liquid can kill C. elegans *** Fast killing assay on plates ± 1,000 U/ml catalase strain SE34 2C4 2F22 Percent of live worms after 26.5 hrs (number of worms) reg AN plates + catalase 62% (21) 100% (43) 14% (22) 100% (24) 94% (31) 100% (39) The 11M12 mutant is less toxic to C. elegans In mutant screen- mobility assay: > 75% of L2 worms retain normal mobility in presence of 11M12 > 50% of adult worms retain normal mobility “ “ Versus WT toxin producing strain which causes complete immobility in >99% L2 worms and ~70% of adult worms, the adult worms continue to move do so at a much slower pace • Sequencing the Tn917 insertion site in the 11M12 mutant was problematic. Tn917 insertion site in 11M12 is near a copA-like gene 11M12 copA tblastx using NCBI produced one hit: S. epidermidis copA score = 126 bits expect = 10-27 The translated 11M12 ORF aligns to the N terminus of a E. faecalis putative cation transporter 11M12 EF0758 CopA is a Copper influx pump Copper can generate hydroxyl radicals from hydrogen peroxide O2- + Cu2+ H2O2 + Cu+ O2 + Cu+ •OH + OH- + Cu2+ Hydrogen peroxide production is decreased in the 11M12 mutant strain SE34 11M12 SE34 11M12 concentration of H2O2 17 hrs aerobic culture 1.5-1.6 mM BDL ( < 0.25 mM) 15 hr Anaerobic culture followed by 2 hrs of aeration 0.8 mM 0.5-0.6 mM Summary of the Evidence that E. faecium kills Worms with Hydrogen Peroxide 1. The E. faecium toxin is diffusible and does not require direct contact with the bacteria. 2. The NADH peroxidase mutant produces more H2O2 and has enhanced C. elegans killing activity 3. The NADH oxidase and the potential Copper transport mutants produce less H2O2 and have decreased C. elegans killing activity 4. The concentration of H2O2 produced by WT faecium is sufficient to kill C. elegans. The timing of the toxicity and effect on worm mobility by the faecium toxin can be duplicated with purified H2O2. 5. Glycerol stimulataes faecium killing while Glucose inhibits faecium killing. 6. Changes in Oxygen levels leads to faecium toxin production 7. Addition of Catalase completely rescues faecium toxicity Possible Reasons for Hydrogen Peroxide Production 1. Byproduct of oxidase reactions 2. Deleterious consequence of aerobiosis 3. Inhibition the growth of competing bacteria 4. Promotes pathogenicity: host tissue damage, induce an inflammatory response or interfere with an immune response 5. The H2O2 forming NADH oxidase (nox-1) may protect against reactive oxygen species. Alternatively, its function may be to actively produce hydrogen peroxide. 6. Acts as a signaling molecule? Young Larval C. elegans feeding off of Enterococci appear to be Starving Adult C. elegans feeding off of E. faecalis or E. faecium does not give rise to viable progeny. Progeny does not develop past the L2-L3 stage. I plated E. faecium strains SE34, 2C4, & 2F22 E. faecalis strain VS583 B. subtilis (didn’t grow well because of antibiotics) I added either eggs, synchronized L1, L2, or young adults Development of the worms Eggs grown on E. coli OP50 or B. subtilis at 25 degress after 24 hrs, reach L2-L3 stage after 48 hrs, become young adults laying eggs and L1 progeny are present after 72 hrs, they are old adults, progeny are L1-L3 stage Eggs grown on Enterococcus resemble eggs grown on bacteria free media after 1 day, reach L1 after 3 days, mostly L2 stay L2-L3 for up to 20 days There is some killing of young larval worms on E. faecalis but the number of worms alive on E faecium is similar to the number on bare media Future Plans 1. Repeat killing assays w/ catalase, the 11M12 mutant, other mut. 2. Rescue the 2C4 and 2F22 mutant phenotypes by expressing plasmid encoded npr and nox genes. 3. Clone the SE34 copA locus. 4. Testing the 2C4 and 2F22 mutants in a mouse sepsis model in collaboration with Joahnnes Huebner 5? Produce recombinant NADH oxidase. Determine if it produces water or hydrogen peroxide? 6? Determine the effect of changing oxygen levels on the expression of npr & nox? Acknowledgments Fred Ausubel Lefterios Mylonakis Danielle Garsin Joahnnes Huebner Markus Stephanie Ausubelites