* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Alkaloids Derived from Phenylalanine and Tyrosine
Pharmacogenomics wikipedia , lookup
Discovery and development of neuraminidase inhibitors wikipedia , lookup
Prescription costs wikipedia , lookup
Pharmaceutical industry wikipedia , lookup
Discovery and development of non-nucleoside reverse-transcriptase inhibitors wikipedia , lookup
Discovery and development of ACE inhibitors wikipedia , lookup
Discovery and development of proton pump inhibitors wikipedia , lookup
Drug interaction wikipedia , lookup
Neuropharmacology wikipedia , lookup
Zoopharmacognosy wikipedia , lookup
Neuropsychopharmacology wikipedia , lookup
Drug discovery wikipedia , lookup
Discovery and development of tubulin inhibitors wikipedia , lookup
Psychopharmacology wikipedia , lookup
Alkaloids Derived
from Phenylalanine and Tyrosine
İhsan ÇALIŞ
References
R. Hänsel&O. Sticher, Pharmakognosie, Phytopharmazie, Springer, Heidelberg 2007
J. Bruneton, Pharmacognosy: Phytochemistry - Medicinal Plants, Lavosier, Paris 1999, 2009
Alkaloids Derived
from Phenylalanine and Tyrosine
• GENERALITIES
• A very large number of alkaloid structures
arise from the metabolism of aromatic amino
acids (phenylalanine, tyrosine).
• These are always isoquinoline alkaloids.
Alkaloids Derived
from Phenylalanine and Tyrosine
• The alkaloids derived from phenylalanine and
tyrosine are compounds in which the basic
structural nucleus is an isoquinoline, or far
more often,
• a 1,2,3,4-tetrahydroisoquinoline.
NH
HO
COOH
N
NH2
HO
Quinoline Kinolein=Kinolin
Phenylalanin
Fenilalanin
N
COOH
isoquinoline
İzokinolein=İzokinolin
NH2
HO
Tyrosin
Tirozin
N H
Tetrahydroisoquinoline
Tetrahidroizokinolin
Alkaloids Derived
from Phenylalanine and Tyrosine
• Biosynthetically, these structures arise from the
reaction of the product of decarboxylation of the
amino acid (phenylethylamine, tyramine) or of
one of its homologs (dopamine) with another
molecule, most often a second molecule of
amino acid which has been deaminated
(aldehyde or equivalent, i.e., an α-ketoacid).
• In rare cases, an isoprene unit may be involved,
for example in Rubiaceae (Psychotria).
Alkaloids Derived
from Phenylalanine and Tyrosine
• Besides the phenethylamines, they are
classified under the five main groups as a
function of the nature of the precursor(s)
which react(s) with the aromatic amino acid
to form the final structure.
Alkaloids Derived
from Phenylalanine and Tyrosine
N
•
•
•
•
•
isoquinoline
İzokinolein=İzokinolin
1. Simple tetrahidroisoquinoleins
2. Benzyl-tetrahidro-isoquinoleins
3. Phenethyl-isoquinoleins
4. Amaryllidaceae Alkaloids
5. Monoterpenoid İsoquinoline alkaloids
N
N
Phenethylamines
N
N
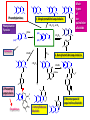
1. Simple tetrahidroisoquinoleins
+ C1, C2, or C5
Phenylalanine
Tyrosine
COOH
(HO)
N
+ C6C2
(HO)
NH 2
Betalains
NH 2
(HO)
+ C6C3
C10
2. Benzyltetrahidroisoquinoleins
+ C6C1
N
Main
types
of
isoquinoleine
alkaloids
+ C6C2
NH
N
O-Gluc
O
N
3. Phenethylisoquinoleins
N
5. Monoterpenoid
İsoquinoline alkaloids
N
Tropolones
O
4. Amaryllidaceae
Alkaloids
N
Alkaloids Derived
from Phenylalanine and Tyrosine
Simple Tetrahydroisoquinoline Alks.
Caryophyllales: Cactaceae, Chenopodiaceae
Benzyltetrahydroisoquinoline Alks.
Magnoliales, Laurales, or Papaverales (Annonaceae, Magnoliaceae,
Lauraceae, Monimiaceae, Papaveraceae, Fumariaceae). Ranunculales
(Berberidaceae, Menispermaceae, Ranunculaceae), (Euphorbiaceae,
Fabaceae).
Phenethylisoquinoline Alks.
Liliaceae (Colchicum)
Alkaloids of the Amaryllidaceae
Clivia, Crinum, Galanthus, Haemanthus, Leucojum, Sternbergia
Monoterpenoid Isoquinoline Alks.
Rubiaceae
1. Simple tetrahidroisoquinoleins
• Pyruvic acid or leucine reacts with
the arylalkylamine to form a
1-alkyltetrahydroisoquinoline.
• The reaction with formaldehyde (or
its equivalent) leads to a
tetrahydroisoquinoline.
• These compounds are relatively
rare, and are found in the
Caryophyllales:
– Cactaceae, Chenopodiaceae,
Fabaceae
N
N
N
+ C1, C2, or C5
(HO)
(HO)
NH 2
2. Benzyltetrahydroisoquinoline
Alkaloids
• Characterized by a C6C2-NC2C6 nucleus, this is the most
important subgroup from the
standpoint of size, and also
structural variety and
pharmacological potential.
(HO)
(HO)
NH 2
+ C6C2
N
• The basic nucleus arises from
the reaction of the
arylalkylamine with a second
amino acid, tyrosine.
2. Benzyltetrahydroisoquinoline
Alkaloids
• The different alkaloids in this group are
characteristic of a certain number of families
of the orders
– Magnoliales, Laurales, or
– Papaverales
• Annonaceae, Magnoliaceae, Lauraceae, Monimiaceae,
Papaveraceae, Fumariaceae
– Ranunculales
• Berberidaceae, Menispermaceae, Ranunculaceae
3. Phenethylisoquinoline Alkaloids
• A second molecule of
aromatic amino acid
participates in the
elaboration of a
C6C2-N-C3C6 nucleus, but
this time it is a
phenylpropanoic acid
(cinnamic acid).
• These compounds are
specific to the Liliaceae
(HO)
NH 2
(HO)
+ C6C3
N
– Androcymbium,
Bulbocodium [Colchicum],
Gloriosa, Kreysigia,
Schelhammera
N
O
Tropolones
3. Phenethylisoquinoleins
4. Alkaloids of the Amaryllidaceae
• Two aromatic amino acids
are required for the
formation of the alkaloids;
– one of the two loses one
carbon atom to form a C6C2N-C1C6 nucleus, which only
occurs in members of this
family, Amaryllidaceae.
(HO)
NH 2
(HO)
+ C6C1
N
Clivia, Crinum, Galanthus,
Haemanthus, Leucojum,
Sprekelia, Sternbergia
AMARYLLIDACEAE
ALKALOIDS
N
Alkaloids
AMARYLLIDACEAE
Activity Studies:
• Cholinesterase inhibitory (acetylcholinesterase
and butyrylcholinesterase) and tyrosinase
inhibitory, activities
Galanthus,
Pancratium
maritumum,
Sternbergia
species
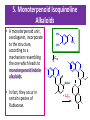
5. Monoterpenoid Isoquinoline
Alkaloids
• A monoterpenoid unit ,
secologanin, incorporate
to the structure,
according to a
mechanism resembling
the one which leads to
monoterpenoid indole
alkaloids.
(HO)
NH 2
(HO)
C10
N
NH
O-Gluc
• In fact, they occur in
certain species of
Rubiaceae.
O
+ C6C2
N
Phenethylamines
N
• Phenethylamine-containing Drugs
– Ephedras:
• Ephedra spp., Ephedraceae
– Khat:
• Catha edulis Forsk., Celastraceae
Phenethylamines
H
O
OH
NH-CH3
NH2
H
H
CH3
H3CO
NH2
CH3
H3CO
Ephedrine
Cathinone
OCH3
Mescaline
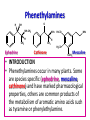
• INTRODUCTION
• Phenethylamines occur in many plants. Some
are species specific (ephedrine, mescaline,
cathinone) and have marked pharmacological
properties, others are common products of
the metabolism of aromatic amino acids such
as tyramine or phenylethylamine.
Phenethylamines
• INTRODUCTION
• Although the concentration of these decarboxylation
products in edible or medicinal plants is too low to induce
harmful effects, it is sometimes sufficient to play a role in
the onset of an attack of migraine.
• The effects of these amines, particularly tyramine, can
become serious in patients treated with MAO inhibitors:
tyramine is no longer metabolized in the intestine and liver,
and a risk of hypertensive crisis ensues.
• Therefore, it is necessary to monitor the consumption of
certain drugs by these patients (e.g., Genista spec.: broom
flowers), as well as certain vegetables (avocado, cabbage,
cucumber, spinach) and certain other foods (cheese).
PHENETHYLAMINE ALKS.
DRUGS: Ephedras
• Ephedra spp.,
Ephedraceae
• Although ephedrine is the
topic of a monograph in the
3rd edition of the European
Pharmacopoeia, ephedra has
long disappeared from most
pharmacopoeias: only
synthetic ephedrine still finds
some uses.
EPHEDRAS
Ephedra spp., Ephedraceae
• The Plants. Ephedras are
dioecious subshrubs with the
same habit as horsetails,
with slender, angular, and
striated branches, and with
leaves reduced to
membranous scales.
• The female flowers are
reduced to the ovule and
surrounded by bracts that
are red and fleshy at
maturity. The male flowers
are grouped in yellowish
catkins.
EPHEDRAS
Ephedra spp., Ephedraceae
• The species that contain substantial quantities of alkaloids
are mostly Asian:
– E. equisetina & E. sinica Stapf, from China,
– E. intermedia & E. gerardiana, from India and Pakistan.
• About ten species are found in North America, for example
Mormon tea, E. nevadensis.
• Ephedras are seldom found in Europe:
– E. major, E. procera, E. campylopoda, or E. distachya of the
Atlantic coast.
• Most of these species, except E. major, like the North American species,
are thought to contain alkaloids at a negligible concentration or no
alkaloids.
TIBBİ BİTKİLER HAKKINDA İLK ESERLER
• M.Ö. 4000
• “PEN TSAO” "herbal" ÇİN
İMPARATORU *Chi’en
NUNG
– 300’den fazla
Bitki Reçetesi
– “ma huang” Chinese Ephedra,
Efedrin (izolasyon: 1887)
Astım ve bronşit tedavisinde
kullanılmış en önemli drogtur
Ephedra - Ephedrine
• Ephedra- E. sinica,
known in Chinese as ma
huang has been used in
traditional Chinese
medicine for 5,000 years
for the treatment of
asthma and hay fever, as
well as for the common
cold.
Ephedra - Ephedrine
• Several additional species
belonging to the genus
Ephedra have traditionally
been used for a variety of
medicinal purposes, and are a
possible candidate for the
Soma plant of Indo-Iranian
religion.
• Native Americans and
Mormon pioneers drank a tea
brewed from an Ephedra,
called Mormon Tea.
Ephedra nevadensis
Soma (Sanskrit sóma), or Haoma (Avestan), from Proto-Indo-Iranian *sauma-, was a ritual
drink of importance among the early Indo-Iranians, and the subsequent Vedic and greater
Persian cultures.
EPHEDRAS
Chemical Composition.
(-)-Ephedrine
(+)-Pseudoephedrine
• Flavonoids and proanthocyanidins have been identified in the
drug, but nitrogen-containing substances—protoalkaloids—
are the focus of attention which are phenethylamine-type
derivatives (>2%).
• The main constituent is almost always (-)-ephedrine, which
represents from 40 to 90% of the total alkaloids.
• (-)-Ephedrine [= (1R,2S)-l-phenyl-2-methylaminopropan-l-ol]
occurs alongside (+)-pseudoephedrine (which has the 1S,2S
configuration) and the corresponding nor and N,N-dimethyl
derivatives.
EPHEDRAS,
Ephedra spp., Ephedraceae
• Chemical Composition.
• All of the Asian ephedras contain alkaloids, but their
concentration varies depending on the species: E. sinica
(1.3%), E. equisetina (2.2%), E. monosperma (2.8%), E.
intermedia (1.1-1.6%).
• Ephedrine is the major compound in most species, except
in E. intermedia where pseudoephedrine is dominant.
EPHEDRAS,
Ephedra spp., Ephedraceae
Ephedroxane
• The drug also contains traces of cyclic compounds: 5phenyloxazolidines and ephedroxane (a 3,4-dimethyl-5phenyloxazolidone).
• The stems also contain a small amount of an alkaloid derived from
spermidine, namely orantine, whose structure is very closely related
to that of the macrocycles described in the subterranean parts of
some species in the genus.
• The roots of several species contain an imidazole derivative (feruloyl
histamine), alkaloidal macrocycles derived from spermine
(ephedradines A-D), and dimeric flavonoids: bisflavanols
(mahuannines) and flavano-flavonols.
Ephedrine
Pharmacological Activity
• Pharmacological Activity.
• Ephedrine is an indirect sympathomimetic.
• Structurally very close to adrenaline, it triggers the
release of endogenous catecholamines from the postganglionic sympathetic fibers.
• Ephedrine
– stimulates cardiac automaticity and has a positive
inotropic activity;
– accelerates respiration and increases its intensity;
– is a bronchodilator and a stimulant of the brain stem
respiration center;
– decreases the contractility of the bladder.
Ephedrine
Pharmacological Activity
• Ephedrine
– is not metabolized much, can be used orally, and its duration of
action is longer than that of adrenaline.
– is well resorbed and highly lipophilic;
– crosses the blood-brain barrier and, by releasing mediators
centrally, has a stimulating psychic effect: stimulation of the
attention and ability to concentrate,
– decrease in the sensation of fatigue and the need for sleep.
• High doses can cause headaches, anxiety, tremors,
insomnia, and psychotic manifestations; redness of the
face; nausea; tachycardia and precordial pain; sweating;
urinary retention, and more.
• Ephedroxane and (+)-pseudoephedrine are experimental
anti-inflammatory agents.
USES OF EPHEDRAS,
Ephedra spp., Ephedraceae
• In France, its uses are very limited.
• In Germany, E. sinica can be used by the oral route, but only for a
short time.
• In Asia, the drug has been used for about 5 millenia. Mahuang
consists of the stems of E. sinica, E. intermedia, and E. equisetina,
and is official in the People's Republic of China where it is used as
an antiasthmatic, diuretic, and sudorific.
• The Chinese Pharmacopoeia also describes mahuanggen (ephedra
root), a drug reputed to be an antisudorific and used as such.
• In the United States, ephedras and ephedrine have been presented
for a few years as potential aids in weight loss, a claim based on a
hypothetical stimulating action on the combustion of fats.
USES OF EPHEDRAS,
Ephedra spp., Ephedraceae
• Ephedrine can be converted chemically into methcathinone and
metamphetamine, two illicit (illegal) substances.
• Naturally, this has led several states in the U.S. to enact restrictive
legislation.
• The proliferation of products based on mahuang and/or ephedrine
has caused an increase in the number of case reports of more or
less serious side effects and in 1997, the FDA proposed detailed
labeling requirements for ephedra-based dietary supplements:
warnings against prolonged used and against combinations with
products such as caffeine, limited claims, information on side
effects, and so forth.
• The directions for using these dietary supplements must not lead
the consumer to take more than 8 mg per unit dose and 24 mg/24
hours.
EPHEDRAS,
Ephedra spp., Ephedraceae
• Production of Ephedrine. Although ephedrine can be
extracted from Ephedra spp., it is also easy to
synthesize.
• The first step in the synthesis is a biological conversion
of benzaldehyde to (R)-1-phenyl-1-hydroxy-2propanone by a yeast {Saccharomyces sp.).
• The second step is a treatment with methylamine.
EPHEDRAS: EPHEDRINE
Uses of Ephedrine
• Ephedrine hydrochloride has long been used to treat the acute attack of
asthma.
• Its multiple activities,
– numerous contraindications (coronary insufficiency, arterial hypertension,
closed angle glaucoma, hyperthyroidism),
– drug interactions (MAO inhibitors, tricyclic antidepressants),
– the required precautions (prostatic hypertrophy, cardiac insufficiency,
diabetes),
– potential adverse effects (tachycardia, headaches, sweating, agitation,
insomnia, anxiety),
– as well as the fact that the effects wear out if the doses are repeated in close
time proximity (tachyphylaxis)
• have led to the virtual (almost) abandon of this compound as a
bronchodilator and analeptic.
•
However, it remains available in some European countries like France for this
indication, particularly in combinations (with theophylline, caffeine, and others).
EPHEDRAS: EPHEDRINE
Uses of Ephedrine
• Ephedrine was formerly used widely for its vasoconstrictive
properties, as an ingredient of nasal sprays or nasal drops, for the
antiseptic and vasoconstrictive treatment of the acute congestion
of rhinitis, sinusitis, and rhinopharyngitis.
• These solutions must not be used in children under three years of
age (risk of central excitation symptoms).
• Limiting the use of these solutions to a short time is important,
because they can induce iatrogenic rhinitis.
• Ephedrine is also an ingredient of syrups and other formulations
designed for the symptomatic treatment of non-productive coughs.
• Note that ephedrine is a banned stimulant in sports, so that its
use may result in a positive doping control test.
EPHEDRAS: PSEUDOEPHEDRINE
Uses of Pseudoephedrine
• Pseudoephedrine hydrochloride, alone or in combination (with
chlorphenamine, ibuprofen, paracetamol, or triprolidine), is an
ingredient of drugs designed for the symptomatic treatment of nasal
congestion and rhinorrhea (coryza).
• These products are contraindicated in children and in patients taking MAO
inhibitors.
• They must be avoided in pregnant or breast-feeding women.
• They must be used with caution in case of hypertension or urination
difficulties.
• Pseudoephedrine has a low toxicity, but it can cause dryness of the
mouth, insomnia, sweating, and anxiety. The onset of tachycardia requires
discontinuing the treatment.
• Pseudoephedrine, like its stereoisomer, is a banned stimulant in sport, so
that its use may result in a positive doping control test.
•
Other Compounds. Combinations of the same type as above contain a synthetic
analog of protoalkaloids: norephedrine (= phenypropanolamine).
KHAT
Catha edulis, Celastraceae
• Khat (or cath, chat, jat,
tschatt, and so forth) is a
shrub of modest size in
arid areas (1-2 m), but it
can reach 10 m in the
tropics.
• The leaves are highly
polymorphic and
indeciduous. Also known
as Abyssinian tea, it is
native to the horn of Africa
(but some think that it
originated in Yemen).
KHAT
Catha edulis, Celastraceae
• Commonly reproduced by
vegetative propagation, it is
cultivated in the south-east of
the Arabian peninsula (Ta'izz,
Yemen), in Somalia, Sudan,
Ethiopia (Harrar), and as far
as Kenya (Meru district) and
Madagascar.
• The leaves are harvested
from the tip of the branches
in the morning and carefully
wrapped (banana leaves,
damp paper, plastic) for
protection against drying and
wilting.
Khat in banana leaves
KHAT
Catha edulis Forsk., Celastraceae
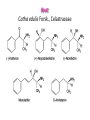
• Chemical Composition. Chemically, the leaf contains
flavonoids, some essential oil, complex polyesters of
polyhydroxylated dihydroagarofurans (cathedulines), and
arylalkylamines (the khatamines), which are responsible
for the activity of the drug.
• In the fresh and young leaves, the chief constituent is
cathinone, in other words (S)-α-aminopropiophenone. In
the dried drag and in older leaves, this (-)-cathinone has
been converted to an 80-20 mixture of (S,S)-(+)norpseudoephedrine and (R,S)-(-)-norephedrine.
KHAT
Catha edulis Forsk., Celastraceae
KHAT
Catha edulis Forsk., Celastraceae
• Chemical Composition. Chemically, the leaf contains flavonoids, some
essential oil, complex polyesters of polyhydroxylated dihydroagarofurans
(cathedulines), and arylalkylamines (the khatamines), which are
responsible for the activity of the drug. In the fresh and young leaves, the
chief constituent is cathinone, in other words (S)-α-aminopropiophenone.
In the dried drag and in older leaves, this (-)-cathinone has been converted
to an 80-20 mixture of (S,S)-(+)-norpseudoephedrine and (R,S)-(-)norephedrine.
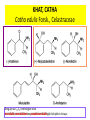
• Fresh drug from northern Kenya also contains the C6-C4 homologs of these
phenylpropylamines: (R,S)-(+)-merucathine, (S)-(+)-merucathinone, and
(S,S)-(-)-pseudomeracathine.
• The phenylpropylamine content is maximal in the young shoots and
appears to depend on the geographical origin: the cathinone level is
reported to range from 9 to 330 mg/100 g depending on the harvest
location. A range of variation just as wide has been observed for
norephedrine and norpseudoephedrine.
KHAT, CATHA
Catha edulis Forsk., Celastraceae
Drog ayrıca C6-C4 homologları olan
merukatin, merukatinon ve pseudomerukatin gibi bileşikleri de taşır.
KHAT
Catha edulis Forsk., Celastraceae
• The cathedulines (Kl, K2, K5,...El-6) are complex molecules whose
structure varies as a function of the geographical origin of the drug. Their
molecular weight ranges, depending on the acids that esterify the
polyhydroxylated sesquiterpene, from 600 to 1,200. Structurally, they are
quite close to the constituents with alkaloid-like behavior that occur in
spindle trees and in some species of Maytenus.
KHAT
Catha edulis Forsk., Celastraceae
• Pharmacological Activity - Toxicity. Pharmacologically, the activity
of (-)-cathinone is qualitatively quite comparable to that of Damphetamine: it causes anorexia, hyperthermia, respiratory
stimulation, mydriasis, arrhythmia, and hypertension.
• This amine induces the release of catecholamines from storage. Its
effects on the central nervous system depend in part on the
subject's environment; they are characterized by a subjective and
euphoric sensation of increased energy, well-being, self-confidence,
mental acuity, and ease in thought formation. Objectively, slight
euphoria can be observed in a talkative and sometimes hyperactive
subject. Later on, undesired effects can appear: insomnia,
nervousness, and nightmares.
• In very rare cases, khat can induce a toxic psychosis, probably by
potentiating a prepsychotic condition. Depression is then observed,
and even schizophreniform or paranoid symptoms.
KHAT
Catha edulis Forsk., Celastraceae
• Use of Khat. The fresh leaves, sold within 24 hours of
harvest, constitute a masticatory known for its
stimulating properties. In some countries (Yemen), khat
use is an ancient custom and it is practiced at social
events, thereby strengthening social bonds. In other
countries, khat is used mainly to seek the
pharmacological effects of the alkaloids (to suppress
the appetite and to combat fatigue).
• Traditionally, the leaves (50-200 g) are chewed one by
one, kept in the mouth for a while, then most often
spit out.
KHAT
Catha edulis Forsk., Celastraceae
• In the early 1990s, Brenneisen and El Sohly estimated the
number of daily users of khat leaves to be between two
and eight million (northeast Africa, Yemen).
• Khat use is officially forbidden in some countries (Saudi
Arabia, Sudan, Somalia) and more or less tolerated in other
countries. Tolerated in Yemen where the authorities ban
alcohol, khat is thought to be consumed daily by 50% of
adult men.
• Cultivated without restriction in Ethiopia, it makes a
massive contribution to the national economy: it is widely
consumed there, and it is also exported to Djibouti where
90% of men and 10% of women are thought to be regular
or occasional users.
Isoquinoline Alkaloids
Simple Tetrahydroisoquinolines
• PEYOTE
• Lophophora williamsii –
Cactaceae
• Considered a divine plant by the
Aztecs, this cactus is a
particularly potent hallucinogen.
This is the "plant that makes the
eyes amazed", in other words
causes visual hallucinations, due
to the CNS activity of a
phenethylamine alkaloid,
mescaline.
Simple Tetrahydroisoquinolines
PEYOTE: Lophophora williamsii – Cactaceae
• The drug contains a large amount of mucilage and about
fifty nitrogen-containing compounds: phenethylamines and
tetrahydroisoquinolines (fresh plant: 0.5-1%, mescal
buttons: 6%).
• The phenethylamines include mescaline (= 3,4,5-trimethoxy-phenethylamine) and its derivatives (N-formyl, Nacetyl, N-methyl), hordenine, 3-demethyl- and 3,4demethylmescaline, tyramine and its derivatives
methylated on the nitrogen atom, and dopamine.
• The tetrahydroisoquinoline alkaloids are anhalamine,
anhalonidine, anhalidine, pellotine, and lophophorine.
• They arise from the condensation of a phenethylamine
with an a-ketoacid (glyoxylic acid, pyruvic acid).
Phenylethylamines and Alkaloids of Peyote
Simple Tetrahydroisoquinolines
PEYOTE: Lophophora williamsii – Cactaceae
• The ingestion of peyote essentially causes psychic effects.
• Mescaline has clinical effects resembling those of LSD (lysergic
acid diethylamide): psychic, cognitive, and physical.
– Note in particular a distortion of the perception of shapes, an
intensification of colors, auditory hallucinations, and a slowing in the
perception of time; the intensity and the nature of the effects are highly
dependent on the environment and the intellect of the subject (for
example, his or her artistic sensitivity).
• The physical symptoms that accompany these hallucinations
are mydriasis, tachycardia, bradypnea, a sensation of change in
temperature, nausea, and possibly agitation and anxiety. At
high doses, memory loss, hypertensive encephalopathy, and
intracranial hemorrhage may be observed.
Isoquinoline Alkaloids
Benzyltetrahydroisoquinolines
• I. Simple Benzylisoquinolines
– Papaverine
• II. Bisbenzyltetrahydroisoquinolines
– Curare
– Other Naturally-occurring Substances with Curare-like Activity
• III. Aporphinoids
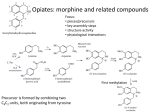
• IV. Protoberberines and Derivatives
• V. Morphinan Alkaloids
– Introduction: Biosynthetic Origin
– Opium Poppy: Opium
• Chemical Composition
–
–
–
–
–
Opium
Morphine
Codeine
Other Alkaloids
Semisynthetic Alkaloids
Isoquinoline Alkaloids
Benzyltetrahydroisoquinolines
• Introduction: The alkaloids derived from 1benzylisoquinoline are surpassed, in structural
diversity.
• Oxidative Coupling
• The above structural diversity results from the
broad reactivity of phenolics, particularly their
coupling reactions via radical intermediates:
this is the classic oxidative coupling of phenols.
Isoquinoline Alkaloids
Benzyltetrahydroisoquinolines
Example of
Oxidative
coupling
Formation of the
phenoxy radical,
mesomerization,
• The phenoxy radical, formed upon oxidation of the phenate
ion, and stabilized by resonance, is highly reactive:
depending on whether the coupling involves the phenoxy
radical and its mesomers, or only the latter, the result is
either the formation of
– a biphenylether bond (Ar-O-Ar), or a
– biphenyl carbon-carbon bond (HO-Ar-Ar-OH).
Isoquinoline Alkaloids
Benzyltetrahydroisoquinolines
• Although this coupling is generally
intramolecular, as in the biosynthesis of
morphine, aporphinoids, or cularines, it can also
be intermolecular: this explains the formation of
the
– bisbenzyltetrahydroisoquinoline alkaloids and the
– binary aporphine-benzyltetrahydroisoquinoline
alkaloids.
• These coupling reactions are normally followed
by rearomatization (aporphines).
Isoquinoline Alkaloids
Benzyltetrahydroisoquinolines
Alkyl substitution at the coupling site leads to a
rearrangment, after which rearomatization is possible
• In a few cases, however, there is no simple pathway for
rearomatization, and rearrangements take place instead,
which can lead to new, more or less profound structural
variations (morphinan and erythrinane alkaloids).
I. Simple benzylisoquinolines
• The quasi totality of these simple compounds are
1,2,3,4-tetrahydro derivatives, in other words
benzyltetrahydroisoquinolines.
• In a few exceptional cases, they are aromatic: one
example is papaverine.
• All of these compounds have, for biogenetic reasons, a
6,7-disubstituted isoquinoline nucleus and a mono-,
di-, or trisubstituted benzyl moiety: the most common
derivatives are of the coclaurine type (12-monosubstituted) and reticuline type (11,12-disubstituted).
• They are the precursors of all other isoquinoline
alkaloids.
I. Simple benzylisoquinolines
• Some of these compounds have an interesting
pharmacological potential, for example higenamine
from Annona squamosa and Aconitum japonicum
which is a cardiac stimulant.
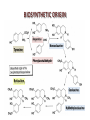
BIOSYNTHETIC ORIGIN
• Benzyltetrahydroisoquinolines are pivotal intermediates in
the metabolism of isoquinoline alkaloids, and are formed
by a Mannich-type condensation between two metabolites
of phenylalanine: for a long time, it was thought that the
condensation of dopamine and 3,4-dihydroxyphenylpyruvic
acid led, via norlaudanosoline, to reticuline, a central
intermediate in the metabolism of isoquinoline alkaloids.
• The condensation of these two molecules leads to (S)-6demethylcoclaurine, which is subsequently methylated (on
the 6-position of the phenol and on the nitrogen atom)
before being hydroxylated at C-12 and finally, methylated
to (5)-reticuline.
BIOSYNTHETIC ORIGIN
Dopamine
Tyrosine
Norcoclaurine
Phenylacetaldehyde
Reticuline
Coclaurine
N-Methylcoclaurine
I. Simple benzylisoquinolines
PAPAVERINE
H3CO
N
H3CO
OCH3
OCH3
• Pharmacological Activity.
• Papaverine is a musculotropic
spasmolytic which relaxes smooth
muscle fibers, especially those of
cerebral, pulmonary, and systemic
peripheral blood vessels, but also those
of the bronchia, intestines, ureters, and
biliary ducts.
• The spasmolytic activity is more pronounced in the case of a preexisting spastic condition.
• Papaverine has an effect on the heart muscle: it decreases
conductibility and excitability, prolongs the refractory period, and
increases coronary blood flow.
• Its activity is linked to its ability to inhibit the phosphodiesterase
which hydrolyzes cAMP, and to decrease the intracellular calcium
concentration (by inhibiting its entry into the cell or increasing its
uptake by the reticulum).
I. Simple benzylisoquinolines
PAPAVERINE: Uses
• Uses. It is still fairly widely used as"vasodilator and antiischemic" in the curative or preventive treatment of
cerebral circulatory insufficiency.
• In addition to being indicated as a smooth muscle relaxant
(injectable solution at 4%) and for the symptomatic
treatment of the intermittent claudication due to chronic
occlusive arterial disease of the lower limbs, it is proposed:
– 1. to improve certain symptoms of senility (e.g., loss of
attention and memory);
– 2. for the symptoms of ischemia in the eye. It is also used
for vertigo in the elderly and to treat the sequelae of
cerebrovascular accidents.
I. Simple benzylisoquinolines
PAPAVERINE: Uses
• Uses.
• Contraindications include intracranial hypertension, parkinsonism,
and intracardiac conductibility alterations, but papaverine is not a
hypotensive agent and only rarely has side effects (potential
tachycardia, constipation, altered transaminases, phosphatases,
and bilirubinemia).
• For the same indications as above, papaverine is sometimes
combined with other compounds (e.g., butalamine). This alkaloid is
also an ingredient of combinations designed to treat capillary
fragility (e.g., combinations with hesperidin methyl chalcone,
ascorbic acid, and ethoxazorutin).
• As an antispasmodic, it is a component of proprietary drugs
designed to relieve the symptoms of functional colopathy,
particularly flatulence and diarrhea.