* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Atomic Theory Notes
Grand Unified Theory wikipedia , lookup
Renormalization group wikipedia , lookup
Compact Muon Solenoid wikipedia , lookup
Electric charge wikipedia , lookup
Quantum electrodynamics wikipedia , lookup
Renormalization wikipedia , lookup
Standard Model wikipedia , lookup
Theory of everything wikipedia , lookup
Electron scattering wikipedia , lookup
Introduction to quantum mechanics wikipedia , lookup
Elementary particle wikipedia , lookup
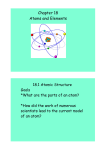
Chemistry Atomic Theory Ms. Mavi/ Ms. Gogolin Atomic Theory Notes Atoms Matter is anything that takes up space and has mass. All matter is made of atoms. Atoms are the basic building blocks of matter. They make up everything around us; Your desk, the board, your body, everything is made of atoms! Atoms are too small to see without powerful microscopes. Subatomic Particles Subatomic Particle Charge Location Proton Positive (+) Nucleus or “Core” Neutron No Charge (0) Nucleus or “Core” Electron Negative (-) Electron Cloud Atomic Structure Electron Cloud: • Electrons orbit the nucleus. Nucleus or “Core”: • Protons and Neutrons are found in the nucleus. Chemistry Atomic Theory Ms. Mavi/ Ms. Gogolin Atomic Theory Over Time Because we cannot see atoms, we use models to teach and learn about atoms. The atomic theory has changed over time as new technologies have become available. o Remember: Scientific knowledge builds on past research and experimentation. Scientist Discovery/ Theory John Dalton All matter is made of atoms. Atoms are too small to see, indivisible and indestructible. All atoms of a given element are identical. J.J. Thompson Discovered the negative electron, and predicted that there also must be a positive particle to hold the electrons in place. Ernest Rutherford Discovered the nucleus of an atom and named the positive particles in the nucleus “protons”. Concluded that electrons are scattered in empty space around the nucleus. Model