* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Metal polymer multiscale material formed by arranging
Survey
Document related concepts
Transcript
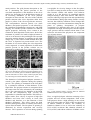
Recent Researches in Communications, Automation, Signal Processing, Nanotechnology, Astronomy and Nuclear Physics Metal polymer multiscale material formed by arranging metal nanoparticles in self patterned spin-on films P. MIKE GÜNTHER, J. MICHAEL KÖHLER Techn. Univ. Ilmenau, Institute of Micro- und Nanotechnologies/ Institute for Physics, Dept. Phys. Chem. and Microreaction Technology, Ilmenau, GERMANY corresponding author: Prof. Dr. J. Michael Köhler, Techn. University Ilmenau, Institute of Micro- und Nanotechnologies/ Institute for Physics, PF 10 05 65, D- 98684 Ilmenau, GERMANY Abstract: Self-patterned polymer films formed by PEGA/PMMA and PVP/PMMA mixtures were generated during spin coating of silicon chips. PVP/PMMA films were applied for arranging gold/silver-core/shell nanoparticles on the film surfaces. The method of spontaneous demixing during spin coating was applied for formation of self-patterned polymer films. A fast demixing is caused by the fast evaporation of ethanol in the final phase of spin-on deposition. It results in the formation of polymer domains with a typical size in the lower micrometer range. The application of aqueous colloidal solution of metal nanoparticles leads to a preferential bonding of the hydrophilic nanoparticles in the PVP domains of self patterned PMMA/PVP films. By this method patterned surface films of metals could be generated without application of lithographic techniques Keywords: nanotechnology, particle manipulation, multi-scale material, self-organization, nanoparticles, gold, silver, PMMA, polyvinylpyrrolidone 1 Introduction A specific feature of metal nanoparticles is their interaction with light. The specific intrinsic energy states of the particles results in a characteristic light absorption connected with the plasmonic states of the particles. The wavelength and intensity of this absorption as well as light scattering and reemission are dependent of the nanoparticle material and their size and shape [2]. Gold and silver are of particular interest for the application of plasmonic nanoparticles. On one hand, the noble metal particles are stable in aqueous colloidal solutions on the other hand the plasmon absorptions can cover different parts of the visible spectrum. So, the gold plasmon absorption is in the range of green light (526 nm) resulting in a red color of the colloidal solution. Silver nanoparticles show a characteristic plasmon absorption band at about 410 nm (violet light) resulting in a yellow color of colloidal solution. Alloy particles as well as Au/Ag core/shell particles show plasmon absorption bands in the whole range between this both values, whereby the position and the shape of the band can be controlled by the composition and size of the particles [3]. Reduced energy gaps result from larger particles and particle aggregates with electronic interactions. This can be used for detection of interactions between nanoparticle-labeled molecules [4]. This longer wavelength absorption - red or purpur - leads to blue or violet color of the colloidal solutions. The plasmonic excitation of silver particles and core/shell The development of nanoparticle-based composite materials can be used for realization of new functions in dependence on their specific mechanical, electrical, chemical or optical properties. These properties are not only determined by the quality of the components but are mainly controlled by the distribution of particles and phases. A high effect of distribution can be expected if the components differ in their properties significantly. This is particular the case in systems consisting of conductors and isolators as metal/polymer composites. Metal nanoparticles are of scientific and practical interest due to their specific electronic and optical properties as well as their catalytic activity. Metal nanoparticle materials combine the high electrical conductivity of metals with the possibility of controlled electron transfer between them. Isolated metal nanoparticles can be act as electron confinements and can be addressed by capacitive coupled nanoelectrodes. Ultrathin oxide films or molecular surface films can be used for controlling electron tunneling between nanoparticles or between a nanoparticle and a conductive substrate. So, metal nanoparticles and in particular arrangements of nanoparticles and polymer molecules are of interest for the development of nanoelectronic devices [1]. ISBN: 978-960-474-276-9 361 Recent Researches in Communications, Automation, Signal Processing, Nanotechnology, Astronomy and Nuclear Physics particles with a silver surface came to the focus of analytical techniques due to the possibility of surface enhanced Raman scattering [5]. By this method, small concentrations of organic molecules can be detected by a non-invasive method if these substances are adsorbed at the surface of the optically active metal nanoparticles and modulate their plasmonic behavior [6]. transparent and non-conductive materials are more interesting if the plasmonic properties of the nanoparticles are addressed. So, polymer self patterning seems to be a better principle of lithographic-free definition of binding areas for nanoparticles. Such structures can be generated by mechanic stress or by precipitation and demixing effects during the evaporation of solvents [13]. The application of polymer films with thicknesses in the order of magnitude of 1 micron is suited for the generation of polymer patterns with typical length at the scale of a few microns. Therefore, the possibility of generation of such self-micro patterned polymer films and their applicability for modulating the local binding density of Au/Ag core/shell nanoparticles was investigated. For technical purposes, the metal nanoparticles must be brought into a suited micro environment. This could either be given by the application of metal nanoparticles at solid surfaces [7], by incorporation in micro particles or by integration in a carrier matrix under formation of a composite material. Polymers are particular interesting for such incorporation. The properties of the polymer matrix and in particular the electron and energy transfer properties can be used for modulate the optical properties of the incorporated metal nanoparticles. Vice versa, the integration of metal nanoparticles into the polymer matrix can be used for tuning their physical properties [8, 9]. 2 Experimental Synthesis of monodisperse Au/Ag nanoparticles Metal nanoparticles can be integrated in polymer matrices if their surface properties are adapted to the chemical properties of the polymers. Molecular surface capping of metal nanoparticles [10] is a suited way for achieving compatibility with the desired polymer matrix. Alternatively, hydrophilic metal nanoparticles can be introduced into a polymer matrix by application of water-soluble polymers [11]. This method is well suited for generation of particle-doped polymer thin films made by spinning. The metal nanoparticles were synthesized in aqueous solution by application of ascorbic acid as reducing agent. In a first step gold nanoparticles were prepared by reduction of tetrachloroauric acid at room temperature. Therefore, 1 mM solution of HAuCl4 was fast mixed with a 20 mM solution of ascorbic acid. The gold nanoparticles are formed in about one second. The formation is indicated by the appearance of the typical purple color of the colloidal solution of metallic gold particles. A more or less statistical distribution of nanoparticles is expected if they are immobilized or adsorbed at a smooth solid surface. Self-patterning is only observed by particle aggregation during the adsorption process. Irregular islands of particle aggregates or irregular network structures are mainly observed in these cases. Alternatively, the density of immobilized particles can be controlled by application of lithographically patterned surfaces. Therefore, polymer films can be used or functionalized micro areas are applied. So, gold nanoparticles capped by oligonucleotides of DNA can be arranged in microsquares by application of patterned films of complementary oligonucleotides [12]. For preparation of nanoparticles with silver shell, the colloidal gold solution was diluted and the final concentration of ascorbic acid was chosen in the range between1 and 5 mM. The silver nitrate solution (in the concentration range between 2 mM and 10 mM) was added by fast mixing. The comparatively small concentrations of the reducing agent and the silver salt results in a slow deposition of silver. The silver nanoparticle formation is very slowly in the absence of any metal surfaces. Gold nanoparticles act as catalytic species for silver deposition. Metallic silver is formed at the surface of the gold nanoparticles. The deposition rate is dependent on silver salt concentration, ascorbic acid concentration, pH, temperature and addition of effectors adsorbing at the particle surface [14]. Despite microlithography, the self patterning of thin films could open a possibility for microlocal modulation of the binding density of nanoparticles. Deposition of islands of a certain thin film material by vacuum methods as evaporation or sputtering as well as galvanic deposition can be applied for generation of metal spots on a smooth surface of a substrate. But, ISBN: 978-960-474-276-9 The complete preparation scheme is shown in fig. 1. The procedure includes both the preparation of the core shell metal nanoparticles and the formation of self-patterned polymer films. The core/shell nanoparticles and the self-patterned polymer films are 362 Recent Researches in Communications, Automation, Signal Processing, Nanotechnology, Astronomy and Nuclear Physics attached to the PMMA film elements is reduced quickly. In contrast, a certain swelling of the PVP matrix and the stronger interaction between the hydrophilic nanoparticles and the hydrophilic PVP result in an enrichment of the nanoparticles at the surface of the PVP film elements. prepared in two separate process chains. In a final step, the particles are brought on to the surface of the patterned polymer film. The chips with self-patterned polymer films with and without metal nanoparticles were inspected by light microscopy and by scanning electron microscopy. Strong optical contrasts were obtained by dark field microscopy in cause of the high light scattering efficiency of the metal particles. In SEM graphs (secondary electron image), the metal nanoparticles appear as bright spots on the darker polymer background due to the higher electron density and the higher yield of secondary electrons of the heavy metals. Fig. 1 Scheme for formation of nanoparticle arrangements on self-patterned polymer films Self-patterning of polymer films were achieved by demixing of solutions of two polymers with two different solvents. The phase behavior and pattern formation was earlier described in polymer systems [15, 16]. Here, solutions of polymethylmethacrylate (PMMA) in toluene (1%), polyethyleneglycol (PEG) in ethanol (10%) and polyvinylpyrrolidone (PVP) in ethanol (5%) were used. The mixed polymer solutions were prepared by mixing the solutions of the pure polymer by vortexing in an Eppendorf cup. 3 Results and Discussion The thickness of silver shell on gold cores can be controlled by the ratio of concentration of colloidal gold particles and the applied silver ion concentration. Simply, higher shell thicknesses can be achieved by application of higher silver salt content in relation to the applied concentration of gold nanoparticles. The reducing agent is applied in a certain excess. The combination of fast nucleation (gold core formation) and low rate of silver deposition lead to a rather homogeneous silver deposition and a homogeneous growth of silver shells. So, Au/Ag core/shell nanoparticles with comparatively narrow size distribution can be synthesized. The ratio of gold cores to silver content determines the final mean diameter of the obtained core/shell particles (Fig. 2). The distribution of particle diameter can be kept low even for high shell thickness. The polymer films were formed by spin-coating of silicon chips. Therefore, the mixed polymer solution was dispensed on to the chip surface. The film was formed by a final rotation rate of about 3500 rpm. The process of pattern formation could be monitored visually by the changing reflection from the chip surface. During the reduction of film thickness and starting solvent evaporation the polymer film remained transparent in the first phase. After a few seconds, the film became opaque what could be seen by an increase of intensity of scattered light. In addition, the final reduction of film thickness, drying and stabilization of film can be observed by changing interference patterns during the last phase of spinning. The dried chips with self-patterned polymer films of PMMA/PVP mixtures were incubated with the suspension of Au/Ag core/shell nanoparticles. After an incubation time of about 15 minutes, the chips were quickly rinsed by cold water. In case of PEG, the water rinsing is not applicable, because it leads to a fast dissolution of the PEG matrix. Fig. 2 Formation of Au/Ag core/shell nanoparticles with different shell thickness The water rinsing causes a fast remove of weakly adsorbed nanoparticles from the surface of the hydrophobic polymer. So, the number of particles The size of the core/shell particles can be tuned between about 20 nm and 400 nm. The thickness of the shell is influencing the optical properties of the ISBN: 978-960-474-276-9 363 Recent Researches in Communications, Automation, Signal Processing, Nanotechnology, Astronomy and Nuclear Physics is negligible for viscosity changes in the first phase, but causes a strong increase in film viscosity when the film becomes smaller and smaller. This behavior results in to a fast decrease of centrifugal material transport during a short time interval. This effect is used for achieving a high precision and reproducibility in film thickness during spin coating. Beside viscosity increase, the solubility conditions can be changed due to the change of solvent concentrations during the last phase of spinning. Precipitation of polymer components or a demixing takes place if one solvent component with high solvation power has the highest vapor pressure and the residual solvent alone is not suited for solvation of the polymer or one component of a polymer mixture. metal particles. The gold plasmon absorption at 526 nm is dominating the optical spectrum of the core/shell nanoparticles if the thickness of the silver shell is low. Increasing silver shell thickness results in an increasing dominance of the silver plasmon absorption at about 410 nm. The color of the colloidal product solutions after silver deposition shifts from purple to yellow if the silver-to-gold-ratio increases. The correspondence between particle size (shell thickness) and optical properties is well reflected by the centrifugal sedimentation spectroscopy and SEM imaging for colloidal product solutions of different optical properties. Increasing silver content in the solution for shell deposition results into a shift of the maximum of particle size band to higher diameter as well as to an increase in the peak height. The expected increasing particle diameter is confirmed by the SEM images (fig. 3). The results of both methods show that the silver is deposited on present metal cores. The formation of new nuclei during the silver deposition can be neglected. A certain population of small metal particles present in the product solution can be explained by residual gold cores without silver shell. Fig. 3 Examples of Au/Ag core/shell nanoparticles with narrow size distribution: particles possessing different silver shell size but constant gold core diameter (SEM images of particles adsorbed on silicon chips (without polymer film); a-f) increasing silver shell thickness (scale bars: 1 µm) The formation of self-organized polymer structures is caused by the phase separation during the spinning process. The spinning is dominated in the first phase by a continuous reduction of film thickness of the liquid film. The polymer solution is transported from the center to the rim of the substrate by the centrifugal forces. The comparatively low thickness of the liquid film and the comparatively high viscosity are responsible for a laminar liquid motion. This leads to a higher surface flow velocity in regions of high film thickness and lower flow velocity at lower film thickness and, therefore, to a fast reduction of local thickness differences. This effect causes the high thickness homogeneity which is normally achieved by spin-on film formation. The last phase of spinning is dominated by solvent evaporation and viscosity increase. The evaporation of solvent during spinning ISBN: 978-960-474-276-9 Fig. 4 Self patterned binary polymer films for different PMMA/PEG mass ratios (optical images) Here, we applied polymer solutions consisting of two polymer and two solvent components. Each of the both polymers shows a high solubility in one solvent and a low in the other one: PVP or PEG was dissolved in ethanol, PMMA in toluene. The solubility of PMMA in ethanol is low as well as the solubility of PVP or PEG in toluene. But, ethanolic solutions of PVP (or PEG) and toluene solutions of PMMA can be 364 Recent Researches in Communications, Automation, Signal Processing, Nanotechnology, Astronomy and Nuclear Physics mixed and form a homogenous liquid. This viscose liquid is applied on silicon chips for spin-on film formation. During the spinning, the concentration of ethanol is faster reduced than the concentration of toluene due to the higher ethanol vapor pressure. The passing of limit concentration of ethanol for miscibility leads to a phase separation, which can be observed during the spinning process by an increase of scattering of light from the film. A toluene-rich phase containing the PMMA is formed beside a PVP-rich phase or a PEG-rich phase containing the majority of the residual ethanol. network consisting of a phase appearing dark in SEM and a second phase appearing bright. The feature sizes of the networks structures are about 10 microns. The dot-like structures have sizes of about 1 to 2 microns. A high number of bright dots was found in the darker matrix area. This pattern could be interpreted as a twophase demixing process. It is assumed that the low ethanol content leads to an early separation into a ethanol-rich phase (A) mainly containing PVP and a toluene-rich phase mainly containing PMMA (B). But both phases contain a certain part of the other solvent and the second polymer. The consequence is a second demixing step inside the previously formed phases if the residual ethanol is evaporated. The phase separation takes place in a time interval in the order of magnitude of a second or less. This time is much to short for a long-range phase separation. The already reduced film thickness and the fast further increase of viscosity by solvent evaporation result in a microlocal character of the separation process. Consequently, more or less regular micro pattern with characteristic dimensions of the single features in the lower micrometer range and characteristic shapes are obtained. Examples of typical demixing patterns in the system PMMA/PEG are given in fig. 4. Therefore different ratios of PMMA-solution in toluene and PEG-solution in ethanol were mixed. The SEM images show bright areas corresponding with the PEG phases and dark areas corresponding with the PMMA phase. Dark islands with an extension of several microns separated by a network of bright lines were found at low PEGto-PMMA-ratio. The principle character of this network structure was also found if the PEG-toPMMA-ratio was slightly increased. But, in this case the size of islands was reduced. The network became denser. A further increase of PEG-to-PMMA-ratio resulted in a transition of bright network into a island structure of bright areas. AFM images show that the polymer film pattern is also connected with a certain surface topography. The roughness of the film surface caused by the demixing process is in the range of about 15 - 25 nm. Fig. 5 Self patterned binary polymer PVP/PMMA film (SEM images): a) polymer mass ratio 3:1, b) polymer mass ratio 4:1 (formation of substructures) Similar network structures were also obtained in case of PMMA/PVP films generated from ethanol/toluene solutions (fig. 5 A). The transition between a close network of PVP (coherent bright areas) into isolated islands (incoherent phase) of PVP was observed, if the PVP/PMMA-ratio was reduced. Metal nanoparticles can be brought into polymer solutions if their surfaces are compatible with the polymer and the solvent. In this case organic colloidal solutions of the metal nanoparticle can be formed in analogy to the colloidal aqueous solutions. The obtained solutions contain the dissolved polymer molecules as molecular disperse system forming the coherent phase and the metal particles forming the incoherent phase. In some cases of reduced ethanol content, the formation of a more complex pattern was found. In this case a structure hierarchy was formed in the selfpatterned polymer films (fig. 5 B): Small dot-like structures appear inside the features of the polymer ISBN: 978-960-474-276-9 The polymer phase separation can be combined with 365 Recent Researches in Communications, Automation, Signal Processing, Nanotechnology, Astronomy and Nuclear Physics groups and functional groups of high affinity to the nanoparticle surface. Such surfactants allow moving the metal nanoparticles from the aqueous phase into a non polar solvent as toluene. Then, the obtained colloidal solution of metal nanoparticles in toluene can be mixed with the PMMA solution and the PVP solution and used for film formation by spinning. The introduction of nanoparticles and surfactants influences the conditions of pattern formation in the phase separation during the spinning process. Any additive substance influences the viscosity of the involved liquids, the partial vapor pressures, miscibilities and interface tensions. So, the obtained self-organized micro pattern differs with changing concentrations of solvents and nanoparticles. matrix deposition of metal nanoparticles if they are introduced into a solution containing two polymers with different solubility properties and a suited solvent mixture. Gold nanoparticles with a polar surface state can be generated in ethanolic solution. So, they can be introduced into the PMMA/PVP system by mixing the ethanolic colloidal solution with an ethanolic solution of PVP and further mixing with the PMMA solution in toluene. A selective deposition of nanoparticles at selforganized micro spots is due to the hydrophilicity of the metal nanoparticles. A high asymmetry between PVP and PMMA areas could be realized if suited rinsing conditions were applied (fig. 7). It was found that the gold/silver core/shell nanoparticles prefer the attachment to the more hydrophilic PVP areas and are removed from the PMMA areas as expected. High densities of metal nanoparticles on the PVP phase and lower density on the PMMA phase can be achieved. Fig. 6 Nanoparticle aggregation and preferential surface binding of metal nanoparticles (Au/Ag) on a self-patterned polymer spin-on films of PVA/PMMA (SEM image) Figure 6 shows the preferential deposition of Au/Ag core/shell particles (mean diameter: 240 nm) on a selfpatterned PVP/PMMA film (3.75:1). The highest density of nanoparticles was found in the central regions of the PVP areas. 4 Conclusions The investigations show that self-organized microstructures in thin polymer films can be applied for arranging metal nanoparticles on chip surfaces. The preferential bonding of gold/silver core/shell nanoparticles is obviously due to the hydrophilic character of these metal nanoparticles and their affinity to the hydrophilic phase of the self-patterned polymer film. The distribution of the metal nanoparticles on the chip surface can be controlled by the type of realized polymer patterns. This depends on the ratio of applied polymers as well as on the applied solvent mixture. The preferential bonding of metal nanoparticles is not restricted to small nanoparticles, but can also be applied for particles up to diameters of several hundred nanometers. Fig. 7 Selective surface binding of Au/Ag core/shell nanoparticles on self-patterned polymer spin-on films (PMMA/PVP) References An introduction of metal nanoparticles into the toluene phase demands for a conversion of the surface state of nanoparticle and a phase transfer process. This can be realized by application of surfactants with alkyl ISBN: 978-960-474-276-9 [1] [2] 366 G. Schmidt (ed) (2004) Nanoparticles. WileyVCH, Weinheim A.N. Shipway, E. Katz, I. Willner, ChemPhysChem 1, 2010, 18-52 Recent Researches in Communications, Automation, Signal Processing, Nanotechnology, Astronomy and Nuclear Physics [3] [4] [5] [6] [7] [8] [9] [10] [11] [12] M. Moskovitch, I. Srnouvá-Sloufová, B. Vlskova, J. Chem. Phys. 116, 2002, 1043510446 W. Fritzsche, Rev. Mol. Biotechnol. 82, 2001, 37 N.H. Kim, S.J. Lee, K. Kim, Chem. Commun., 2003, 724-725 K. Strehle, D. Cialla, P. Rösch, T. Henkel, J.M. Köhler, J. Popp, Anal. Chem. 79, 2007, 1542-1547 R.M. Bright, M.D. Musick, M.J. Natan, Langmuir 14, 1989, 5695-5701 R. Jordan, N. West, A. Ulman, Y.-M. Chao, O. Nuyken, Macromolecules 34, 2001, 16061611 J. Bae, E. Glogowski, S. Gupta, W. Chen, T. Emrick T, T.P. Russel, Macromolecules 41, 2008, 2722-2726 S. Link, Z.L. Wang, M.A. El-Sayed, J. Phys. Chem. 103:, 1999, 3529-3533 W. Fritzsche, H. Porwol, A. Wiegand, S. Bornmann, J.M. Köhler, Nanostructured Materials 10:. 1998, 89-97 J. Reichert A. Csaki, J.M. Köhler, W. ISBN: 978-960-474-276-9 [13] [14] [15] [16] [17] 367 Fritzsche, Analytical Chemistry 72, 2000, 6025-6029 J.M. Köhler, L. Weh, Makromol. Chem. 192:, 1991, 1147 K. Sun, J.X. Qui, J.W. Liu, Y.Q. Miao, J. Mater. Sci. 44:, 2009, 754-758 J.M. Köhler, M. Held, U. Hübner, J. Wagner, Chem. Eng. Technol. 30, 2007, 347-354 J.S. Gutmann, P. Müller-Buschbaum, M. Stamm (1999), Faraday Discuss. 112: 285297 L.V. Govor, J. Parisi, G.H. Bauer, G. Reiter, Phys. Rev. E 71, 2005, No 051603