* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Myths and Barriers - Jacobi Medical Center
Survey
Document related concepts
Public health genomics wikipedia , lookup
Compartmental models in epidemiology wikipedia , lookup
Transmission (medicine) wikipedia , lookup
Eradication of infectious diseases wikipedia , lookup
Human mortality from H5N1 wikipedia , lookup
Infection control wikipedia , lookup
Herd immunity wikipedia , lookup
Influenza A virus subtype H5N1 wikipedia , lookup
Viral phylodynamics wikipedia , lookup
Transmission and infection of H5N1 wikipedia , lookup
Swine influenza wikipedia , lookup
Avian influenza wikipedia , lookup
Transcript
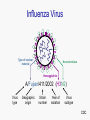
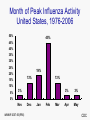
Influenza Immunization Update 2007-2008 Sheila L. Palevsky, MD MPH Bureau of Immunization New York City Department of Health and Mental Hygiene October 17, 2007 Impact of Influenza • It is estimated that 10-20% of all adults and up • to 40% of children in the U.S. are infected with influenza each year ~226,000 hospitalizations annually – About half among those in persons >65 years of age – Rates of influenza hospitalization in children <5 years similar to rates in adults >65 years • >36,000 deaths during each influenza season – More than 90% are in adults >65 years Influenza Virus • Orthomyxoviridae family • 3 types: A, B, C • Only influenza A and B cause epidemic human disease • Influenza A viruses categorized into subtypes on the basis of two surface antigens: – hemagglutinin – neuraminidase Influenza Virus Type of nuclear material Neuraminidase Hemagglutinin A/Fujian/411/2002 (H3N2) Virus type Geographic origin Strain number Year of isolation Virus subtype CDC Influenza Antigenic Changes Influenza viruses undergo genetic changes Drift Shift • Minor changes, same subtype • Major change,new subtype • Genetic point mutations • Exchange of gene segments • Occurs in A and B subtypes • Occurs only in A subtypes • May cause yearly epidemics t • May cause periodic pandemics ⇛ Occurs frequently ⇛ Occurs infrequently The Next Antigenic Shift The question is not if, but when …nor do we know what kind it will be Immunity to Influenza Antigens • Immunity to the surface antigens, especially the hemagglutinin, reduces the likelihood of infection • Antibody against one influenza virus type or subtype confers limited or no protection against another type or subtype of influenza • Antibody to one antigenic type or subtype might not protect against infection with a new antigenic variant of the same type or subtype Influenza – Clinical Features • • • • Respiratory transmission of virus Contagious prior to the onset of symptoms Incubation period 1-5 days (average 2 days) Abrupt onset of fever, myalgia, sore throat, nonproductive cough, headache (vomiting and diarrhea may be seen in childhood illness) • Severity of illness depends on prior experience with antigenically related variants All that is called the “flu” is not influenza Influenza - Complications • Pneumonia – Primary influenza – Secondary bacterial • • • • Otitis media (children) CNS complications including Reye Syndrome Myocarditis / myositis / rhabdomyolysis Exacerbations of underlying medical condition(s) • Death 0.5 – 1 per 1,000 cases (for all ages) Important to Remember • Influenza is spread by coughing, sneezing – through droplet spread • Individuals are contagious for 1-4 days before the onset of symptoms and for 5-10 days after the first symptoms • Children may shed virus for >10 days after onset of symptoms • About 50% of infected individuals do not have any symptoms but are still contagious Differentiating Respiratory Illnesses • Difficult to distinguish influenza from other respiratory illnesses on the basis of signs and symptoms – Young children less likely to have typical symptoms of cough and fever – Older adults may not have typical symptoms • The absence of symptoms of influenza-like illness (ILI) does not effectively rule out influenza Use laboratory tests to confirm the diagnosis Influenza Vaccine Efficacy • Dependent upon: – Age and immunocompetence of the vaccine recipient – The degree of similarity between virus in the vaccine and those in circulation – Outcomes being measured • • • • • Medically attended acute respiratory illness (MAARI) Laboratory confirmed influenza Prevention of influenza-associated hospitalization or death Seroconversion to circulating vaccine strains Seroconversionto circlutaing virus strains Influenza Vaccine Efficacy Healthy Adults • For healthy adults <65 years of age: – 70%-90% effective in preventing clinical illness in healthy adults when vaccine matches circulating strains of virus – 50%-77% against laboratory-confirmed illness when vaccine strains were antigenically dissimilar to circulating strains – Protection against healthy adults against influenza-related hospitalization was 90% You cannot get “the flu” from the flu shot Influenza Vaccine Efficacy Adults With Chronic Disease • For adults <65 years of age with chronic disease: – Vaccine generally less effective than in healthy adults – In adults 50-64 years of age, when circulating strain and vaccine were not well matched (2003-2004), vaccine efficacy: • 60% for healthy adults vs. 48% among high-risk adults in preventing laboratory confirmed illness • 90% for healthy adults vs. 36% among high-risk adults in preventing hospitalization You cannot get “the flu” from the flu shot Influenza Vaccine Efficacy Older Adults • For adults >65 years, the goal is to prevent secondary complications and reduce the risk for influenza-related hospitalization and death – Among non-institutionalized persons >60 years, ~60% effective in preventing clinical illness (lower efficacy in those >70 years) – 30%-70% effective in preventing hospitalization in noninstitutionalized seniors – In older nursing home residents: • 20-40% effective in preventing MAARI • ~80% effective in preventing death You cannot get “the flu” from the flu shot Flu Vaccine • Although the efficacy of flu vaccine in the elderly may be less than desired, it is the best protection against influenza and its complications • The risk of hospitalization and death due to flu is high in the elderly (about 5% of deaths) • Preventing spread of the flu is important ! Flu Vaccine in Children • Influenza vaccine is effective in children >6 months of age • Children 6 months <9 years of age should receive 2 doses of flu vaccine in the first season they receive vaccine – Among children <9 years of age who have never received influenza vaccine before and who received only 1 dose of vaccine in their first year of vaccination, vaccine is less effective as compared with children who received 2 doses in their first year of being vaccinated. Influenza Vaccine (TIV) Adverse Reactions Local reactions* 15% - 20% Fever, malaise* uncommon Allergic reactions Neurological reactions rare very rare You cannot get “the flu” from the flu shot *Local rxns and fever may be more common in young children The Flu Shot (TIV) • Contraindications - anaphylactic reaction to a previous dose or to egg protein, gelatin, or other vaccine component • Precaution – moderate to severe illness – Vaccinate if minor illness: diarrhea, upper respiratory tract illness (including otitis media) with or without fever or on current antimicrobial therapy • Precaution – history of GBS within 6 weeks of receipt of a dose of flu vaccine Month of Peak Influenza Activity United States, 1976-2006 50% 45% 45% 40% 35% 30% 25% 19% 20% 13% 15% 13% 10% 5% 3% 3% 3% Apr May 0% Nov MMWR 2007:56(RR6) Dec Jan Feb Mar CDC 2007-2008 Influenza Vaccine • A/Solomon Islands/3/2006(H1N1) - like • A/Wisconsin/67/2005 (H3N2) - like • B/Malaysia/2506/2004 - like Strains included in the vaccine change every year based on surveillance data You need this year’s flu vaccine to protect against this year’s flu!! Vaccine supply information will be available at www.cdc.gov/nip/flu Flu Vaccine Availability and Timing of Vaccination • Influenza vaccine is being received in offices now! • Vaccine is usually most plentiful later in the calendar year – in November and December – prior to the peak of influenza activity • Influenza peaks in late winter; vaccine should continue to be given well into the spring • Keep vaccinating so long as supply is available • If indicated, give pneumococcal (and any other indicated) vaccine(s) along with flu vaccine 1 Recommendations for Influenza Vaccine: 2007-2008 • All persons, including school-aged children, who want to reduce the risk of becoming ill with influenza or of transmitting influenza to others • All children 6-59 months of age • All persons >50 years • Children and teens (6 months -18 years) on long-term aspirin therapy • Women who will be pregnant during influenza season • Children and adults with chronic pulmonary (asthma), cardiovascular, renal, hepatic, hematological or metabolic disorders (diabetes mellitus) • Children and adults with immunosuppression (medication, therapy, human immunodeficiency virus) CDC 2 Recommendations for Influenza Vaccine: 2007-2008 • Children and adults who have any condition that compromise respiratory function or handling of secretions (cognitive dysfunction, spinal cord injuries, seizures, neuromuscular disorders) • Residents of nursing homes or other chronic care facilities who are at least 6 months of age • Health care personnel • Healthy contacts including children and caregivers of children <5 years and adults >50 years, with emphasis on contacts of children <6 months of age • Healthy household contacts (including children) and caregivers of persons with medical conditions that predispose to complications from influenza CDC Influenza Vaccine Recommendations Chronic Illness* • • • • • • Pulmonary disorders (includes emphysema and asthma) Heart disease Metabolic disease (e.g., diabetes) Renal dysfunction Hemoglobinopathies Immunosupression – HIV/AIDS – Medications and other treatments • Conditions that compromise respiratory function or the the handling of secretions or increase the risk of aspiration * >6 months of age LAIV Indications • Healthy* persons 2 - 49 years of age – Health care workers, except those who have direct contact with severely immunocompromised patients who require protective isolation – Contacts of those at high risk (including contacts of infants and young children) – Persons who wish to reduce their own risk of influenza * Persons who do not have medical conditions that increase their risk for complications of influenza Administration of LAIV • Persons at increased risk for influenza complications* may administer LAIV • Gloves and masks are not required *e.g., pregnant women, persons with asthma and persons 50 years of age or older Pregnancy and Influenza • Risk of hospitalization due to complications of the flu is 4 times higher than in non-pregnant women • Risk of complications comparable to nonpregnant women with high-risk medical conditions • Vaccination (with TIV) is recommended if pregnant during influenza season • Vaccination can occur during any trimester • Vaccinate all contacts of pregnant women HIV Infection and Influenza • Persons with HIV at increased risk of complications of influenza • TIV induces protective antibody titers in many HIV infected persons • Transient increase in HIV replication reported • TIV will benefit many HIV-infected persons • Vaccinate all contacts of HIV-infected persons Influenza Vaccines 2007-2008 Vaccine Dose / Presentation Age Doses Route INACTIVATED VACCINE (TIV) 0.25 ml pre-filled syringe* 6-35 mos 1 or 2† IM 0.5 ml pre-filled syringe* >36 mos 1 or 2† IM 0.5 ml vial* >36 mos 1 or 2† IM 5.0 ml multi-dose vial* > 6 mos 1 or 2† IM Fluvirin (Novartis) 5.0 ml multi-dose vial >4 yrs 1 or 2† IM Fluarix (GSK) 0.5 ml pre-filled syringe >18 yrs 1 IM FluLuval (GSK) 5.0 ml multi-dose vial >18 yrs 1 IM Afluria (CSL Limited) 0.5 ml pre-filled syringe >18 yrs 1 IM 5.0 ml multi-dose vial >18 yrs 1 IM 2-49 yrs 1 or 2¶ Intra-nasal Fluzone (sanofi pasteur) LIVE, ATTENUATED (LAIV) FluMist (LAIV) 0.1 ml spray in each nostril *vaccines approved for children younger than 4 years † two doses administered at least one month apart for children 6 months <9 yrs who are receiving influenza vaccine for the first time; children <9 years who received only one dose in their first season of vaccination should receive 2 doses in the next year ¶ two doses administered at least one month apart for children 2 yrs <9 yrs who are receiving influenza vaccine for the first time; children 2 yrs <9 yrs who received only one dose in their first season of vaccination should receive 2 doses in the next year Vaccination of Health Care Personnel • All physicians, nurses, other health care personnel in all hospital and out-patient settings, including emergency rooms, and all home care attendants should receive vaccine annually • But - - - • in the 2004 National Health Interview Survey, only • 42% of health care workers reported receiving influenza vaccine in the previous 12 months; and in 2005, only 33% of health care workers in NYC reported they got a flu vaccine! What about you ??? Benefits of Influenza Vaccination of Health Care Workers • Reduction in nosocomial influenza and influenza-related deaths • Reduction in staff illness and illnessrelated absenteeism • Reduction of direct medical costs and indirect costs from work absenteeism MMWR. 2006;55 (RR-2) Reasons Health Care Workers Do Not Receive Influenza Vaccine • Concern about vaccine adverse events • Perception of a low personal risk of influenza virus infection • Insufficient time or inconvenience • Reliance on homeopathic medications • Avoidance of all medications • Fear of needles • Mistrust of government recommendations MMWR. 2006;55 (RR-2) Factors Facilitating Health Care Worker Influenza Vaccination • • • • • Desire for self-protection Previous receipt of influenza vaccine Desire to protect patients Perceived effectiveness of the vaccine Legislative and regulatory mandates – Effective January 2007, JCAHO requires facilities to offer flu vaccine on site to staff as part of employee health programs MMWR. 2006;55 (RR-2) Barriers to Vaccination • Health care workers’ misconceptions about influenza as a disease • Lack of understanding by health care workers that influenza can be deadly for patients and contacts “flu is not a serious disease” Barriers to Vaccination • Perception of a low personal risk of contracting influenza “I don’t get sick; I don’t need it” “absenteeism vs. presenteeism” Barriers to Vaccination • Failure of health care workers to explain vaccine efficacy and safety “the flu vaccine will make me sick” “I don’t think the vaccine really works” Barriers to Vaccination • Health care workers are poor role models Did you have your flu vaccine last year? Influenza in Healthcare Workers • Healthcare workers – Implicated in introducing influenza into and causing outbreaks among patients in health care settings • ICU, neonatal intensive care units, nursing homes – Often work while ill, exposing vulnerable patients and their coworkers to influenza – May be able to spread influenza if infected, but not symptomatic • Can shed virus before symptoms develop • About half of all influenza infections asymptomatic Barriers to Vaccination • Need for annual vaccination “I had the flu shot before, I don’t need it again” Barriers to Vaccination • Need to address popular myths – taking of vitamins, never get sick, etc. “home remedies and preventives are better” “I don’t need foreign substances in my body” Barriers to Vaccination • Fear of needles LAIV is a good alternative, if eligible Barriers to Vaccination • There are many excuses offered to avoid vaccine – don’t give in to them “I don’t eat eggs” “I am taking medications” “now isn’t convenient -I’ll get it later” Barriers to Vaccination • Low reimbursement Reimbursement • Medicare covers flu vaccine and includes an administration fee • All Medicaid managed care plans will cover influenza vaccine for at-risk adults 19-64 years • Commercial insurance plans should provide coverage for at-risk patients • Commercial insurance must cover children with an indication for flu vaccine • Influenza vaccine is available through the Vaccines for Children program for eligible children and teens Barriers to Vaccination • Missed opportunities Use every encounter as an opportunity to vaccinate Cultural and Ethnic Issues • Understand and address different cultural beliefs of health and sickness • Address socio-political issues such as distrust of governmental recommendations and the memory of Tuskegee Getting Ready for Flu Season • Order sufficient vaccine • Vaccinate all office staff • Develop protocols within the health care setting – – – – – – Have patient information available (posters, flyers) Flag charts of high-risk patients Use standing orders Have special designated vaccination hours Make every encounter an opportunity to vaccinate Use signed declination forms if is vaccine refused • Offer vaccine once available: continue vaccinating into the spring Influenza Vaccine: Strategies to Increase Coverage • Expand use of standing orders • For employees: – Vaccination sessions • • • • Institute vaccination days Use traveling teams – take the vaccine to the employee Use incentives Monitor and report influenza vaccination levels in the facility – Education • Education, education, education, education, education Get documentation if vaccine was given at another location Pneumococcal Disease • Pneumococcal pneumonia is a common complication of influenza • Rates of severe pneumococcal infection and death are highest in the elderly Incidence of Invasive Pneumococcal Disease in Adults >18 yrs, US 1999-2000 Category Incidence rate (cases/100,000 persons) Healthy 8.8 Diabetes 51.4 Chronic heart disease 93.7 Chronic lung disease 62.9 Solid cancer 300.4 HIV/AIDS 422.9 Hematological cancer 503.1 Alcohol abuse 100.4 Adapted from Kyaw, et al., JID. 2005;192:377-386. Age-Specific Incidence, by Race, of Invasive Pneumococcal Disease in Adults (>18 yrs) with No Reported Underlying Illness United States, 1999-2000 Kyaw, et al., JID. 2005;192:377-382. Rates of Death or Intensive Care Unit (ICU) Admission, by Pneumococcal Vaccination Status and Age Johnstone, J. et al. Arch Intern Med 2007;167:1938-1943. PPV23 Recommendations • All adults >65 years of age • Persons >2 years with – certain chronic illness – functional or anatomic asplenia – immunocompromised (disease, chemotherapy, radiation therapy, high-dose steroids) – HIV infection – cerebrospinal fluid leaks and cochlear implants – environments or settings with increased risk PPV 23 Vaccination: Chronic Illness* • • • • • • • Diabetes or other metabolic disease Chronic heart disease Chronic respiratory disorders Chronic liver disease including alcoholism Kidney failure, ESRD, hemodialysis Hemoglobinopathies Immunosupression – HIV/AIDS – cancer – medications and other treatments * >2 years of age PPV23 Revaccination • Routine revaccination of immunocompetent persons is not recommended • Revaccination recommended for persons age >2 years at highest risk of serious pneumococcal infection • Single revaccination dose >5 years after first dose PPV23 Candidates for Revaccination • Persons >2 years of age with: – the highest risk of complications from IPD – persons whose antibodies decline rapidly • functional or anatomic asplenia • immunosuppression • chronic renal failure • nephrotic syndrome • transplant recipients • Persons vaccinated at <65 years of age PPV23 Revaccination • Revaccination once, 5 years after 1st dose for those aged >65 years: – if received vaccine >5 years ago, and – were aged <65 years at the time of vaccination PPV23 Vaccination and Revaccination • If previous vaccination is uncertain – no available record or date of vaccine VACCINATE do not miss the opportunity There are no contraindications to simultaneous administration of any vaccines Give influenza and pneumococcal vaccines at the same visit if both are indicated VPD Surveillance Report suspected cases of vaccine preventable diseases to 212-676-2284/88 after hours: 212-POISONS Vaccine Adverse Events Report suspected vaccine adverse events (VAERS) 800-822 7967 or www.vaers.org or call 212-676-2284 A vaccine not given is 100% not effective Sheila L. Palevsky, MD MPH Provider Liaison Chief, Professional Education Unit New York City Department of Health and Mental Hygiene 2 Lafayette Street – 19th floor - CN21 New York, NY 10007 Phone: 212-676-2264 Fax: 212-442-8091 Email: [email protected]