* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download 2016 Q7 - Loreto Balbriggan
Survey
Document related concepts
Radiator (engine cooling) wikipedia , lookup
Building insulation materials wikipedia , lookup
Cutting fluid wikipedia , lookup
Underfloor heating wikipedia , lookup
Dynamic insulation wikipedia , lookup
Heat exchanger wikipedia , lookup
Heat equation wikipedia , lookup
Copper in heat exchangers wikipedia , lookup
Solar water heating wikipedia , lookup
R-value (insulation) wikipedia , lookup
Thermoregulation wikipedia , lookup
Cogeneration wikipedia , lookup
Intercooler wikipedia , lookup
Thermal conduction wikipedia , lookup
Transcript
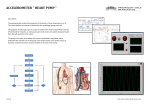
State Examination Commission – Physics Higher Level, 2016 Question 7 At a lecture in Cork in 1843, James Joule, while describing his work on heat and temperature, suggested the principle of conservation of energy. Later in the nineteenth century, the work of Joule and Lord Kelvin led to the invention of the heat pump. Distinguish between heat and temperature. State the principle of conservation of energy. (10) As part of his presentation, Joule proposed that the temperature of the water at the bottom of the Niagara Falls would be 0.12 °C greater than that at the top, due to gravitational potential energy being converted into heat energy. Calculate the height of the Niagara Falls. In reality the increase in temperature will be much smaller. Suggest a reason for this. (12) In a heat pump, a fluid is used to transfer energy from a cold body to a warmer body. Describe the operation of a heat pump and explain how a heat pump can be used to reduce the temperature of a cold region, for example the interior of a refrigerator. State two desirable physical properties of the fluid used in a heat pump. (18) The fluid in the heat pump of a refrigerator has a specific latent heat of vaporisation of 4.6 MJ kg–1. The internal volume of the refrigerator is 0.6 m3. The heat pump removes 12 kJ of energy from the air in the refrigerator as the fluid evaporates. Calculate (i) the mass of fluid that has evaporated (ii) the fall in temperature of the air in the refrigerator. (16) (specific heat capacity of water = 4200 J kg–1 K–1; acceleration due to gravity = 9.8 m s–2; density of air = 1.23 kg m–3; specific heat capacity of air = 1005 J kg–1 K–1) _______________________________________________________________ Distinguish between heat and temperature. State the principle of conservation of energy. (10) Textbook As part of his presentation, Joule proposed that the temperature of the water at the bottom of the Niagara Falls would be 0.12 °C greater than that at the top, due to gravitational potential energy being converted into heat energy. Calculate the height of the Niagara Falls. loss in potential energy of water = gain in heat energy m gh = mc Δ θ gh = c Δ θ h = cΔθ 4200×0.12 = = 51.4 m g 9.8 In reality the increase in temperature will be much smaller. Suggest a reason for this. (12) A lot of the water will vaporise on falling In a heat pump, a fluid is used to transfer energy from a cold body to a warmer body. Describe the operation of a heat pump and explain how a heat pump can be used to reduce the temperature of a cold region, for example the interior of a refrigerator. Textbook State two desirable physical properties of the fluid used in a heat pump. (18) A high specific latent heat of vaporization and low boiling point. C. Garvey 2016 The fluid in the heat pump of a refrigerator has a specific latent heat of vaporisation of 4.6 MJ kg–1. The internal volume of the refrigerator is 0.6 m3. The heat pump removes 12 kJ of energy from the air in the refrigerator as the fluid evaporates. Calculate (i) the mass of fluid that has evaporated E = ml 4 m = E 1.2×10 = = 2.6×10−3 kg l 4.6×10 6 (ii) the fall in temperature of the air in the refrigerator. Mass of air in the refrigerator = ρ V = 1.23×0.6 = 0.738 kg If 12 kJ is removed from this mass of air it will drop in temperature by Δθ = E 12,000 0 = = 16.2 C mc 0.738×1,005 (16)