* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Drosophila
Survey
Document related concepts
Transcript
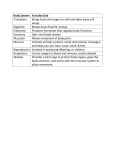
mRNA Regulation and Development Martine Simonelig Les organismes-modèles pour l’étude des dystrophies musculaires mRNA Regulation and Development Martine Simonelig Translational control of maternal mRNAs during early development in Drosophila Isabelle Busseau, Catherine Papin, Christel Rouget, Willy Joly, Bridlin Barckmann, Anne-Cécile Meunier Development of a Drosophila model of the human oculopharyngeal muscular dystrophy or OPMD Aymeric Chartier, Nicolas Barbezier, Cédric Soler Les organismes-modèles pour l’étude des maladies génétiques humaines Les organismes-modèles: levure crible à grande échelle (molécules) drosophile (Drosophila melanogaster) rapidité génétique/outils génétiques très développés nématode (Caenorhabditis elegans) souris génétique mammifère/plus proche de l’homme Quelles maladies génétiques humaines? Cancer, retards mentaux, épilepsie, diabète, infection (immunité innée)... Maladies neurodégénératives / Dystrophies musculaires Conservation des génomes et des fonctions moléculaires entre l’homme et les organismes modèles Gènes impliqués dans des maladies génétiques humaines 77% ont un homologue chez la drosophile 65% ont un homologue chez le nématode Two ways to produce an animal model of a human genetic disease By loss of function of the gene homologous to the human gene mutated in the disease recessive genetic disease due to a loss of function mutation requires that the animal model has the homologous gene affected in the disease e.g.: - Fragile X syndrome (mutation in FMR1: fragile X mental retardation 1) (Drosophila model) - Spinal muscular atrophy (mutation in SMN: survival motor neuron) (Drosophila model) - Duchenne Muscular Dystrophy ( mutation in dystrophin) (C. elegans model) By expressing or overexpressing the human mutant protein in the animal dominant genetic disease due to a gain of function mutation does not require the homologous gene in the animal model Neurodegenerative diseases / Oculopharyngeal Muscular Dystrophy Inducible expression in Drosophila using the UAS/Gal4 system (Brand & Perrimon 1993) Gal4 Fly stock GAL4 X UAS Fly stock cDNA-X UAS polyA Genomic regulation sequence UAS/Gal4 embryos, larvae or flies expression GAL4 GAL4 UAS cDNA-X Genomic regulation sequence Gal4 drivers: ubiquitous, neurons, photoreceptor neurons, muscles ... polyA Drosophila models of human neurodegenerative diseases Since 1998 Models of neurodegenerative diseases By expressing in Drosophila the human mutant protein Polyglutamine diseases: at least 9 neurodegenerative diseases due to expansion of a polyglutamine tract in different proteins: normal up to 35 glutamines / disease when 40 or more glutamines. - Huntington's (huntingtin) - Spinocerebellar ataxia type 3 (SCA3) - Spinocerebellar ataxia type 1 (ataxin 1) Non-polyglutamine diseases: - Parkinson's (a-synuclein) - Alzheimer's / Tauopathy (tau) Pathology: neurodegeneration, late onset, progressive memory loss, cognitive deficits, movement disorders At the cellular level: the mutant protein forms insoluble aggregates (protein conformation diseases) Expression of polyglutamine expanded protein in the eye: photoreceptor neurons (GMR-Gal4) Marsh & Thompson, 2004 Features of the human diseases that are conserved in Drosophila: polyglutamine diseases: - Neuronal degeneration - Late onset - Progressive - Onset and severity correlate with polyglutamine repeat length - Early death - Cellular level:Nuclear inclusions (NI) containing the polyglutamine-expanded mutant protein, or the polyglutamine alone with a tag - Control: no NI with 20/27 glutamines. Phenotypes reproduced in Drosophila with 127 glutamines out of the context of a protein: The polyglutamine is intrinsically toxic non-polyglutamine diseases: - Parkinson's: adult onset loss of dopaminergic neurons, filamentous inclusions in neurons (with a-synuclein), locomotor dysfunction. - Alzheimer's / Tauopathy: adult onset, progressive neurodegeneration, accumulation of abnormal tau. Identify suppressor genes of these human diseases in Drosophila, by a genetic approach identify genes involved in the disease process without a priopri of the molecular function to understand molecular mechanisms of the disease identify molecular pathways of the disease to find targets for possible therapies Identify suppressor genes of these human diseases in Drosophila, by a genetic approach (genetic screens) Mutagenesis, collections of mutants loss of function mutants: chemical mutagenesis, P element inserted randomly in the genome gain of fonction mutants: P-UAS element inserted randomly in the genome UAS P-UAS element gene X expression Screen Suppressors or enhancers of the phenotype induced by expression of the polyglutamine in the eye (by the UAS/Gal4 system) suppressor UAS-polyQ/+; GMR-Gal4/+ phenotype enhancer UAS-polyQ/+; GMR-Gal4/+ UAS-polyQ/+; GMR-Gal4/+ Suppressor genes of neurodegenerative diseases in Drosophila (by genetic screens) 1999/2007: genetic screens or test of candidates Suppressors of polyglutamine diseases: increased expression of - P35: viral anti-apoptotic apoptosis - Chaperone proteins/or pathway: HSP70 (human HSPA1L) HSP40/ HDJ1 (chaperone-related J domain) dTRP2 (chaperone-related J domain) HS response factor DnaJ1 64EF (chaperone) Major pathways: (N. Bonini, 2007) Protein folding Protein degradation - ubiquitin/proteasome pathway - proteins involved in autophagy (protein degradation by lysosomes) - CBP: histone acetyltransferase sequestration of transcription factors/or histone acetyltransferase by the poly-Q expanded protein Suppressors of Parkinson’s: increased expression of HSP70 New approaches to find suppressors of these diseases in the Drosophila model, based on the cellular pathways identified by genetics 1) Prevention of aggregation 2) Protein folding: HSP70 pathway 3) Transcription regulation: inhibitors of histone deacetylase New approaches to find suppressors of these diseases in the Drosophila model, based on the cellular pathways identified by genetics from 2002/2003 1) Prevention of aggregation Design of suppressor peptides: for Huntington’s disease 25Q 17 aa huntingtin spacer ahelice (54 to 67 aa) 25Q myc prevents polyglutamine aggregation suppresses the phenotype in vivo in the Drosophila model (L. Thompson, 2002) Screen of suppressor peptides and test in vivo in the Drosophila model: Polyglutamine Binding Peptide (QBP1) for polyglutamine diseases suppresses neurodegeneration and early death (T. Toda, 2003) Test of chemical or pharmacological compounds as suppressors - Congo red and cystamine: suppressors in vivo for polyglutamine disease (L. Thompson, 2003) New approaches to find suppressors of these diseases in the Drosophila model 2002/2003 2) Protein folding: HSP70 pathway Pharmacological compounds as suppressors Geldanamycin: antibiotic, increases the level of HSP70 suppressor in vivo of Parkinson’s disease (N. Bonini, 2002) 3) Transcription regulation: inhibitors of histone deacetylase Chemical compounds as suppressors Butyrate and SAHA: inhibitors of histone deacetylases suppressors in vivo of Huntington’s disease (L. Thompson 2001, Min 2003) From Drosophila models to mouse models Marsh &Thompson, 2004 New approaches to find suppressors: the Drosophila model as an in vivo test. High throughput test of molecules in Drosophila 2005/2006 Companies that test large collections of molecules (EnVivo Pharmaceuticals) -molecules are delivered in the food from the embryonic stage/ change of the food every day - e.g. 20 000 flies per week of a disease model possible test of collections up to 30 000 molecules Hits (or positive): their effects are analysed at cellular and molecular levels, for validation Test of the hits in mouse models 2006: at least one molecule in clinical trials in patients, identified thanks to a Drosophila model, in an academic lab (R. Cagan), for a cancer: Multiple Endocrine Neoplasia Type 2. The molecule stops metastasis. New approaches to find suppressors: the Drosophila model as an in vivo test. Test of intrabodies in Drosophila 2005 Ag Intrabodies : single chaine antibodies expressed within the cell VH VL linker Variable Light Variable Heavy IgG In Drosophila (Messer 2005): Cloning of intrabody DNA downstream of UAS: expression with Gal4, whithin the cells expressing polyQ-exon1Huntingtin: - screen for intrabodies specific to Huntingtin (yeast phage display) -optimisation of the intrabody (test in yeast model) intrabody UAS expression Reduction of neurodegeneration and formation of aggregates Increase survival to adulthood (23% without intrabody/ 100% with intrabody) Models for Muscular Dystrophies: in Drosophila or C. elegans Drosophila C. elegans Duchenne muscular Dystrophy dystrophin mutant yes yes Spinal muscular atrophy (SMA) survival motor neuron (SMN) mutant yes yes yes no yes yes Myotonic Dystrophy Oculopharyngeal muscular Dystrophy expression of noncoding CUG repeats expression of the human mutant protein Duchenne Muscular Dystrophy Loss of function mutation / recessive disease / X-linked The most common of muscular dystrophies: 1 boy / 3500 Extremely severe: wheelchair-bound at 12, respiratory failure in early twenties No treatment Mutation in the gene encoding Dystrophin (very big gene: 2.9 megabases, 79 exons) (sporadic cases: 1/10 000 sperm or eggs) Most DMD patients lack the Dystrophin Dystrophin: 3685 AA protein present in skeletal and cardiac muscles Duchenne Muscular Dystrophy Dystrophin bridges extracellular matrix and cytoskeleton inside muscle cells DGC: dystrophin glycoprotein complex Nowak & Davis 2004 Duchenne Muscular Dystrophy Potential mechanisms contributing to muscle degeneration in DMD: - Structural role of dystrophin: degradation of proteins of the Dystrophin-Glycoprotein complex in the absence of dystrophin: decrease in the amounts of the complex: Muscle fibers are more sensitive to mechanical damage: leads to muscle degeneration, chronic inflammation, susceptibility to oxidative stress - inappropriate location of membrane components leading to alteration of ionic canals - Role of the Dystrophin-Glycoprotein complex in the intracellular nitric oxide (NO) pathway: loss of association between DGC and the nitric oxide synthase (nNOS) leads to impaired nitric oxide production: role of NO in epigenetic regulations through the regulation of HDAC (histone deacethylase). Nitric oxide and HDAC have a role in DMD: rescue of nNOS expression ameliorates the dystrophic phenotype in the mouse model of DMD deacethylase inhibitors are beneficial in the mouse model Animal models for Duchenne Muscular Dystrophy Mouse model: mutation in the gene encoding dystrophin (stop codon in exon 23): mdx mice mild myopathy No cell model or in vitro model:need for a model useful for high-throughput screens Possible models in Drosophila or C. elegans: Dystrophin and the proteins of the DGC complex are conserved in Drosophila and C. elegans / in smaller number (dystrobrevin, sarcoglycans, syntrophins, dystroglycan, sarcospan) muscles with a sarcomeric structure and protein composition similar to mammalian striated muscles (but no satellite cells, and no fusion in C. elegans) C. elegans model of Duchenne muscular dystrophy (L. Ségalat, Lyon) mutation in the Ce gene encoding dystrophin: dys-1 (null mutation) phenotype: hyperactive locomotion, muscular hypercontraction, BUT... no muscle degeneration Double mutant in dys-1 and MyoD (myogenic factor) dys-1-; CeMyoDts : locomotion defects / adult onset / progressive over time protein homologous to a rat protein interacting with neural nitric oxide synthase nNOS WT CeMyoDts dys-1- dys-1-; CeMyoDts dyc-1 dys-1-; + CeMyoDts overexpression uncoordinated suppressor C. elegans model of Duchenne muscular dystrophy: analysis of muscle structure (optic microscopy) active molecule phalloidin staining: visualization of actine fibers WT dys-1-; CeMyoDts dys-1-; CeMyoDts + prednisone (0.5 mg/ml / steroid) Identification of prednisone from a test screen of 100 molecules (reduces muscle degeneration) Prednisone is used as a treatment for DMD boys Identification of this molecule in the C. elegans model in a blind screen indicates that this model can be used for the search of active molecules Suppressors of Duchenne muscular dystrophy in the C. elegans model Test of existing pharmaceutical compounds in the DMD C. elegans model : Serotonin (neuro-hormone) is a suppressor of muscle degeneration Reduction of serotonin levels leads to muscle degeneration in the CeMyoD mutant A function of serotonin in muscles Serotonin is beneficial to striated muscles (Ségalat 2006) - Mutation in the chn-1 gene decreases muscle degeneration in the DMD C. elegans model CHN-1 is the homologue of human CHIP: interacts with E3 ubiquitin ligase and E4 enzyme (ubiquitin-conjugating factor) - A proteasome inhibitor has the same effect (MG132) Implication of the ubiquitin/proteasome pathway in DMD (Baumeister 2007) Exon skipping therapy in Duchenne muscular dystrophy (Garcia, Danos /Généthon 2004) Knowledge of the disease in man, at the molecular level Test in the mouse model, mdx mouse Restoration of dystrophin production dystrophin spectrin-like repeats STOP Muscle regeneration Restoration of muscle capacity antisense RNA Stable over time possibly permanent U7 snRNA nonspliceosomal snRNA modified to be incorporated in spliceosome used to deliver antisense sequence during splicing in AAV vector: injected in mice, intramuscular or intra-arterial (adeno-associated virus) Drosophila model of oculopharyngeal muscular dystrophy: OPMD Aymeric Chartier, Béatrice Benoit & Martine Simonelig. A Drosophila model of oculopharyngeal muscular dystrophy reveals intrinsic toxicity of PABPN1. EMBO J. 2006, 25, 2253-2262. Chartier Aymeric, Raz Vered, Sterrenburg Ellen, Verrips Theo C., van der Maarel Silvère & Simonelig Martine. Prevention of oculopharyngeal muscular dystrophy by muscular expression of Llama single-chain intrabodies in vivo. Human Molecular Genetics 2009, 18, 1849-1859. Contact: [email protected] http://www.igh.cnrs.fr/equip/Martine.Simonelig/