* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Electron beam computed tomography
Remote ischemic conditioning wikipedia , lookup
Antihypertensive drug wikipedia , lookup
Quantium Medical Cardiac Output wikipedia , lookup
Saturated fat and cardiovascular disease wikipedia , lookup
Cardiovascular disease wikipedia , lookup
Cardiac surgery wikipedia , lookup
History of invasive and interventional cardiology wikipedia , lookup
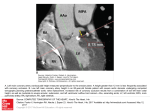
European Heart Journal Supplements (2002) 4 (Supplement F), F47–F53 Understanding the pathophysiology of the arterial wall: which method should we choose? Electron beam computed tomography R. Erbel1, T. Budde2, G. Kerkhoff2, S. Möhlenkamp1 and A. Schmermund1 1Department of Cardiology, University Essen, and 2Department of Internal Medicine and Cardiology, Alfried Krupp Hospital, Essen, Germany Coronary imaging techniques may be direct or indirect. Indirect methods permit visualization of the coronary artery lumen but not the diseased coronary artery vessel wall. In the early stages of coronary arteriosclerosis, vessel size enlargement compensates for up to 50% stenosis from plaque formation. Indirect methods are therefore unable to visualize early signs of coronary arteriosclerosis. Direct techniques are those that permit visualization of non-calcified and calcified plaque deposits within the coronary artery vessel wall. Electron beam computed tomography (EBCT) is currently the standard technique for assessing calcified lesions. It allows direct visualization of the coronary arteries in a non-invasive manner. Coronary calcium within a plaque is accurately detected and quantified by EBCT. This modality therefore holds promise as a method for studying the natural history of coronary artery disease, irrespective of symptomatic status. Studies have shown that the presence of calcification almost invariably indicates the presence of coronary artery disease, and that the absence of calcification can almost rule out significant coronary artery disease. Moreover, a close correlation exists between the degree of calcification and severity of coronary artery disease. EBCT may also be used in young patients with risk factors in order to detect disease in pre-clinical stage, and to provide new information on the natural history of coronary artery disease. (Eur Heart J Supplements 2002; 4 (Suppl F): F47–F53) © 2002 The European Society of Cardiology Introduction therefore important, in particular because the degree of calcification has been shown to correlate with plaque load[3–5] and provides a means of verifying the presence of coronary arteriosclerosis. Coronary imaging techniques can be either indirect or direct. Indirect techniques permit visualization of the artery lumen but not the diseased artery wall, and include selective coronary angiography, magnetic resonance angiography, contrast-enhanced electron beam tomography angiography and synchrotron radiation angiography[6]. Direct techniques permit visualization of both non-calcified and calcified plaque deposits within the vessel wall. Fluoroscopy and, more recently, digital fluoroscopy have been used to detect coronary calcification[7,8]. However, it is virtually impossible to localize and quantify calcification using fluoroscopy. Therefore, other techniques have been developed that also allow non-invasive diagnosis of coronary arteriosclerosis, including electron beam computed tomography (EBCT). This modality is now the technique of choice for assessing calcified lesions, and has become the The development of arteriosclerosis is divided into five stages (Stary lesion types I–V), according to the recommendations of the American Heart Association[1]. Lesions of types I and II are regarded as normal degenerative disease. Type III is an intermediate stage, which is characterized by increasing numbers of lipid droplets that coalesce to form a lipid core, leading to the formation of a type IV lesion – an atheroma. If the plaque contains collagen, then the lesion is classified as type V (fibroatheroma)[1]. Calcification does not necessarily indicate advanced disease because it may be detected in type III lesions, first intra-cellularly and then extra-cellularly[2]. Detection of coronary calcification is Correspondence: Raimund Erbel,MD, FESC, FACC, FAHA, University Professor of Medicine/Cardiology, European Cardiologist, Department of Cardiology, Division of Internal Medicine, University Essen, Hufelandstr. 55, D-45122 Essen, Germany. 1520-765X/02/0F0047 + 07 $35.00/0 Key Words: Arteriosclerosis, atherosclerosis, calcification, coronary artery disease, electron beam computed tomography, non-invasive imaging. © 2002 The European Society of Cardiology F48 R. Erbel et al. ECG Echocardiography Noninvasive methods Scintigraphy PET EBCT, cardiac CT Invasive methods 0% Intracoronary ultrasound Coronary angiography 20% 45% 60% 70% 90% Remodeling Figure 1 Overview of the diagnosis of coronary atherosclerosis. Current non-invasive techniques are focused on detecting myocardial ischaemia that occurs as a consequence of flow-obstructing coronary stenoses. Therefore, nonstenotic coronary atherosclerotic lesions are not detected. In contrast, electron beam computed tomography (EBCT), or other CT methods with sufficiently rapid image acquisition, visualize coronary plaque formation directly. Accordingly, non-stenotic plaques can be identified. Whereas coronary angiography (the standard method for invasive diagnosis) is best suited to analyzing highly stenotic lesions, intra-vascular ultrasound (IVUS) allows visualization of calcified and non-calcified coronary plaques of all degrees of stenosis. However, because of its invasive nature and the associated costs, the use of IVUS is limited to a small subgroup of patients. PET=positron emission tomography. ‘gold standard’ of non-invasive direct coronary imaging techniques (Fig. 1)[6]. Technique of electron beam computed tomography EBCT uses an electron sweep of stationary tungsten target rings in order to generate X-ray images that can accurately detect small amounts of calcium[9]. The EBCT scanner can be operated in various scanning modes, using up to four target rings. For coronary artery imaging, the ‘single-slice mode’, which employs only one target ring, gives optimal spatial resolution[6]. The EBCT examination is performed using a C100, C150 or C300 scanner (GE Imatron, South San Francisco, California, U.S.A.), with contiguous, non-overlapping 3-mm slices, and an acquisition time of 100 ms. Patients are supine and, after localization of the main pulmonary artery, 36–40 slices in the craniocaudal direction through the apex of the heart are obtained, with ECG triggering at 80% of the R–R interval. The presence of coronary calcium is evaluated at each level. The threshold for calcific lesions is usually set at a CT density of 130 Hounsfield units (HU), in an area of at least 0·52 mm2 (two pixels, 512 × 512 pixel matrix and a 26-cm field of view). This threshold has been shown to provide sufficient differentiation from surrounding tissue and blood, and has been widely accepted as signifying calcification[10,11]. The total examination time is 3–4 min. In addition, 5–10 min are needed for evaluation, quantification of calcification and generation of a report; ideal presumption for a screening technique. Eur Heart J Supplements, Vol. 4 (Suppl F) September 2002 Detection of coronary calcification EBCT permits non-invasive visualization of coronary artery calcification[10–17]. In the different scan fields, all lesions exceeding 130 HU (representing coronary calcification with the image intensities similar to bone structures) are imaged (Figs 2–4). Figure 4 is a typical example, showing calcification not only in the proximal coronary arteries but also in the distal parts of the left and right coronary arteries. Distribution of calcification All lesions exceeding 130 HU are regarded as calcified lesions and the distribution of calcification in the left and right coronary arteries can therefore be analyzed (Figs 3 and 4). The distribution of calcification is semi-quantitatively described, in accordance with the 16-segment model of the American Heart Association[18]. The referring physician must be aware of the vessel segments involved, in order to correlate coronary calcification with wall motion, or location of myocardial infarction[5,11,19]. Quantification of calcification Traditionally, the scoring algorithm introduced by Agatston et al.[10] has been used to quantify coronary calcium. The calcium score is defined as the product of the area of coronary artery calcium (at least two contiguous pixels, with a CT density ≥130 HU) and a factor (rated 1–4) that is Electron beam computed tomography Figure 2 Quantification of calcification by electron beam computed tomography (EBCT). Quantification of coronary calcification according to the Agatston score of a calcification in segment 6 of the left anterior descending coronary artery. The area of pixels with Hounsfield units exceeding 130 are summed up and multiplied by the maximum computed tomographic number (MCTN), according to a given score. In this example the number is 3 based on 313 HU and the area 8 mm2, giving a score of 24. AO=aorta; C=calcification. dictated by the maximum CT density within that lesion (1 = 130–200 HU; 2 = 201–300 HU; 3 = 301–400 HU; 4 = >400 HU). In the example shown in Fig. 2 the area is 8 mm2 and the density is 313 HU, giving a calcium grade of 3; the Agatston score is therefore 24. Calcification can be analyzed for separate coronary segments, such as those classified by the American Heart Association, and summed F49 Figure 3 (A) Electron beam computed tomography (EBCT) and (B) angiography of a healthy patient with normal coronary arteries. Coronary calcification is absent in the left main and in segment 6/7 of the left anterior descending coronary artery. Ao=aorta; LAD=left anterior descending; RVOT=right ventricular outflow tract. for each artery to yield a total score[5]. As in the example shown in Fig. 2, calcification is typically found in segment 6 of the left anterior descending coronary artery. Figure 4 shows a lesion with an Agatston score greater than 2000. Accuracy of electron beam computed tomography In order to evaluate its accuracy, EBCT has been compared with intra-coronary ultrasound, which is able to visualize not only calcified but also non-calcified plaques at high resolution (100–150 µm)[5,11]. It has been demonstrated that even single spots of calcification detected by intra-coronary Figure 4 (A) Electron beam computed tomography (EBCT) and (B) angiography of a patient with coronary artery disease. Shown are EBCT images in four slices of a patient with severe coronary calcification, confirmed by coronary angiography with main stem stenosis, and severe disease in left circumflex and right coronary arteries. In the different slices, the highlighted areas are those in which the pixels exceed 130 Hounsfield units. AO=aorta; RVOT=right ventricular outflow tract. Eur Heart J Supplements, Vol. 4 (Suppl F) September 2002 F50 R. Erbel et al. ultrasound can also be visualized using EBCT. These findings are particularly significant because the comparison was conducted in individuals with no or minimal luminal narrowing. EBCT is therefore a highly accurate method for detecting coronary calcification non-invasively. Moreover, EBCT shows a strong relationship between the number of coronary segments with plaque formation and the number of calcified coronary segments, confirming in vivo the high correlation found at autopsy between plaque load and amount of calcification[3,4]. EBCT has been compared with coronary angiography in many studies[19–23]. Those comparisons were unable to evaluate the accuracy of EBCT because coronary angiography only visualizes the lumen of coronary arteries whereas EBCT visualizes the arterial wall. However, new developments in EBCT and particularly multislice computed tomography (MSCT) have shown that, when contrast is injected in order to discriminate the arterial wall from the coronary lumen, not only calcified but also non-calcified lesions can be visualized[24]. Prevalence of coronary calcium Pathological studies, conducted in soldiers and individuals who have died from causes other than cardiac disease (such as car accidents), have demonstrated the presence of advanced stages of coronary plaque formation (Stary lesions types IV/V). More than 15% of young adults have advanced coronary lesions (atheroma and fibroatheroma)[2] that can be detected by fluoroscopy[25]. Janowitz et al.[26] demonstrated that the prevalence of calcification as visualized by EBCT is similar to that seen in pathological studies. Calcified plaques have been detected in 20% of patients aged 30–39 years, in 40% of those aged 40–49 years, in 50–60% of those aged 50–59 years, and in 70–80% of those aged 60–69 years[23]. In the Muscatine study[8], conducted in people aged 29–37 years, coronary calcification was detected in 31% of men and 10% of women. The Coronary Artery Risk Development in Young Adults (CARDIA) study[27], which analyzed persons aged 28–40 years, detected coronary calcification in 17% of men and 9% of women. Those studies demonstrate that coronary calcification is not only present in the advanced stages of the disease but can also be detected quite early in a high proportion of young adults in the general population. Risk factors for coronary calcification Risk factors for development of coronary arteriosclerosis are well known and have been classified according to cardiovascular risk in the Framingham study[28]. The following risk factors for coronary calcification were identified by multivariate regression analysis[5,14]: age, fibrinogen, the total cholesterol/high-density lipoprotein cholesterol ratio, hypertension, male sex and positive familial history. Lowdensity lipoprotein (LDL)-cholesterol, diabetes and current Eur Heart J Supplements, Vol. 4 (Suppl F) September 2002 smoking were positively correlated with frequency of calcified and non-calcified plaques[5]. Topography of coronary calcification In order to elucidate the natural history of coronary calcification, EBCT was used to investigate the distribution and prevalence of calcification in various coronary segments of young persons with or without coronary luminal narrowing[29]. Calcification was first observed in segment 6 of the left anterior descending coronary artery – a finding consistent with pathological anatomy reports that coronary arteriosclerosis is first found in the proximal 2 cm of the left anterior descending coronary artery[30]. Later, calcification is also found in the middle and proximal right coronary arteries, as well as in the proximal left circumflex coronary artery. In more advanced stages, the middle part of the left anterior descending and the left main arteries are involved. Only in the most advanced stages of the disease is calcification also found in distal segments of the left anterior descending and left circumflex arteries. The distribution in the right coronary artery is more even[29]. Coronary calcification in acute coronary syndromes Because coronary calcification is found in the early stages of the disease, it is not surprising that it is found in a high percentage of people suffering sudden cardiac death[31–34]. In a study of 50 cases of sudden death due to coronary thrombosis, plaque rupture was found in 28 out of 50 individuals and plaque erosion in 22 out of 50 individuals[31]; 69% of the ruptured plaques and 23% of the eroded plaques were calcified. In another study, the plaques responsible for sudden cardiac death in 108 individuals (mean age 50 years) were classified as ‘stable’ (n = 20), ‘erosion’ (n = 33), ‘acute rupture’ (n = 37), or ‘healed rupture’ (n = 18), and it was found that 80% of ruptured plaques were calcified[34]. Similarly, in a study of 79 adults (mean age 49 ± 11 years, 71% male) who suffered sudden cardiac death, those with stable (n = 27) or ruptured (n = 30) plaques showed significantly more coronary calcification than did those with eroded plaques (n = 22)[32]. Recently, in a study of 28 individuals under 50 years old who suffered sudden cardiac death, Schmermund et al.[33] reported that calcification was present either at or close to the site of plaque rupture in a high percentage of cases. This indicates a close association between coronary calcification and plaque rupture. The presence of calcium appears to influence the mechanics of the arterial wall, increasing wall and shear stresses, and thereby increasing the likelihood of plaque rupture. Prognostic value of coronary calcification determination In six studies, coronary calcification (as determined using EBCT) and mortality were analyzed in order to calculate the Electron beam computed tomography relative risk ratio[35–40]. In one follow-up study 288 patients who had undergone EBCT scanning at the same time as coronary angiography were contacted after a mean of 6·9 years[35]. It was found that there was a significant relative risk ratio (1·88; P < 0·05) associated with the extent of coronary calcification at baseline and the incidence of future hard cardiac events (cardiac death and non-fatal myocardial infarction). In a multicentre prospective study, 491 patients (mostly symptomatic, with suspected coronary artery disease) underwent both EBCT and coronary angiography[36]. The data showed that patients who suffered a coronary event had calcification scores of 100 or greater, whereas those who did not have an event had calcification scores below 100. In a study of over 1000 asymptomatic individuals (baseline age 53 years, 71% men), over an average followup of 3·6 years, it was found that a baseline coronary calcification score of 160 or greater was associated with an odds ratio of 15·8 for all coronary events and of 22·2 for cardiac death or non-fatal myocardial infarction[38]. In a study of 172 patients with a first acute myocardial infarction (mean age 53 ± 8 years), 165 patients (96%) showed coronary calcification by EBCT, and in 87% of the 172 patients the degree of calcification was greater than expected for their age and sex[39]. The prognostic value of EBCT-determined coronary calcification was confirmed in a recent prospective study of 221 patients (mean age 53 ± 9 years, 54% men)[40]. Patients underwent EBCT scanning, in addition to usual care, on admission to the emergency department of a large tertiary care hospital for chest pain syndromes. When calcification was present, the relative risk ratio for coronary events was found to be 28 within an average follow-up time of 4·2 years. Implementation of detection and grading of coronary calcification According to the Prevention Conference V proceedings, individuals may be classified as having low, intermediate, or high risk for cardiovascular events[41]. High risk is defined as a 20% chance of suffering a cardiovascular event within 10 years. Risk factors are subdivided into causal risk factors (hypertension, hypercholesterolaemia, diabetes and smoking) and conditional risk factors (lipoprotein[a], high triglyceride levels and elevated fibrinogen). If carotid stenosis, coronary artery disease, aortic aneurysm, or peripheral vascular disease is present, then the individual is considered to be at high risk. Also at high risk are those individuals with more than two causal risk factors or more than four conditional risk factors. Individuals with two causal risk factors or three to four conditional risk factors are considered to be at intermediate risk. The Prevention Conference V participants considered the role of several measures of subclinical disease in order to determine whether individuals at intermediate risk should be reclassified as high risk. Currently, an intima–media thickness greater than 1 mm, an ankle brachial index below F51 0·9, or an elevated level of C-reactive protein are the criteria used to redefine such individuals. It is anticipated that EBCT will provide an additional method of risk assessment. The advantage of EBCT is that it measures a direct sign of coronary arteriosclerosis (coronary calcification) rather than an indirect sign, such as intima–media thickness or peripheral artery disease. Progression of coronary calcification In order to demonstrate spontaneous progression of coronary artery disease, EBCT was used to visualize changes in calcification over time in 102 asymptomatic persons[29]. Over a mean period of 18 months, the area of calcification as well as the Agatston score were measured, because it has been suggested that the former provides the most reproducible results for serial EBCT studies[42]. Calcification was analyzed in the 12 major coronary segments, as classified by the American Heart Association. The median relative annual progression in the total area of calcification and in total Agatston score were 27% and 32%, respectively. In the near future, it is likely that, because of lower interobserver variability, the calcium volume score will be used to calculate total coronary calcium load. Thus far, all prognostic long-term studies have used the Agatston score. Other studies have also analyzed the progression of coronary calcification over time. Maher et al.[43] measured the change in coronary calcification in 88 individuals over a 3·5-year period, and showed an average increase of 24% per year in calcification score. Möhlenkamp et al.[44] observed an increase of the Agatston score in non-obstructive coronary artery disease of 8 ± 2 and in obstructive coronary artery disease of 46 ± 9 per year[44]. Janowitz et al.[26] reported an increase of the Agatston score in unknown coronary artery disease of 20 ± 8 and in obstructive coronary artery disease of 159 ± 10. The data indicate a linear relationship between increases in Agatstons score and scores measured at the first EBCT study time. The progression mainly occurred in areas where most calcium was already detected. Measurement of coronary calcification to study the treatment of arteriosclerosis The detection of coronary calcification, and its quantification by EBCT, is an ideal pharmacodynamic method for studying the effects of treatment on arteriosclerosis. It has recently been shown that statins influence the development of coronary calcification[45]. A retrospective study was conducted in 149 patients (age range 32–75 years, 61% men) with no history of coronary artery disease, who were referred for EBCT screening. All patients underwent screening at baseline and at a follow-up assessment 12–15 months later. Calcium volume was calculated as an estimate of plaque burden. Statin treatment was started at Eur Heart J Supplements, Vol. 4 (Suppl F) September 2002 F52 R. Erbel et al. the discretion of the referring physician, and serial measurements of LDL-cholesterol were correlated with change in calcium volume over time. The 44 (30%) patients who did not receive treatment had an average LDLcholesterol level of 120 mg . dl – 1 or greater at follow-up, and a significant increase (52%) in calcium volume. Of the 105 (75%) patients who received statin treatment, a net reduction (–7%) in calcium volume was observed only in those patients (n = 65) whose LDL-cholesterol levels at follow-up were below 120 mg . dl – 1; the treated patients whose LDL-cholesterol levels were 120 mg/dl or greater at follow-up (n = 40) had a significant increase (25%) in mean calcium volume, although the increase was smaller than that in the untreated group. In addition, Achenbach et al.[46] recently reported a reduction in coronary calcification progression with cerivastatin therapy. Whereas a first follow-up study showed an increase in calcium volume score of 28%, it was reduced by cerivastatin to 9%. In order to study the effect of lipid-lowering therapy on coronary calcification in more detail, several studies have been initiated. The Beyond Endorsed Lipid Lowering with EBT Scanning (BELLES) study was designed to compare aggressive lipid-lowering therapy using atorvastatin with a more moderate regimen using pravastatin, in postmenopausal women with hypercholesterolaemia[47]. In the EBCT Assessment of Coronary Calcification in High Risk Patients with Minimal or Moderate Coronary Arteriosclerosis (EBEAT), patients received intensified lipidlowering therapy with low and high doses of atorvastatin. The Treat-to-Target study uses a new phosphate-binding drug in haemodialysis patients. Future developments In the future, studies of coronary calcification will not only utilize EBCT but also MSCT[48]. In addition to allowing visualization of calcified plaques, MSCT is showing promise for the visualization of non-calcified plaques when contrast material is used in addition to native scanning[49,50]. Similar results have been obtained with EBCT, allowing the concurrent visualization of coronary calcification and luminal narrowing. Conclusion The detection of coronary calcification in asymptomatic persons allows us to step beyond secondary prevention and to identify the high-risk patient for primary prevention. Prevention will therefore not be based on risk factor assessment alone but also on direct visualization of coronary arteriosclerosis, allowing treatment to be targeted only at those who require it, reducing cost and risk. EBCT is an ideal technique for assessment of risk and it can be used in emergency units. It may be expected that this technique will be included in future coronary heart disease guidelines, in order to classify patients at intermediate risk Eur Heart J Supplements, Vol. 4 (Suppl F) September 2002 more accurately. EBCT is already in use as a new tool in pharmacodynamic studies of arteriosclerosis. References [1] Stary HC, Chandler AB, Dinsmore RE, et al. A definition of advanced types of atherosclerotic lesions and a histological classification of atherosclerosis. A report from the Committee on Vascular Lesions of the Council on Arteriosclerosis, American Heart Association. Circulation 1995; 92: 1355–74. [2] Stary HC. The sequence of cell and matrix changes in atherosclerotic lesions of coronary arteries in the first forty years of life. Eur Heart J 1990; 11: E3–19. [3] Sangiorgi G, Rumberger JA, Severson A, et al. Arterial calcification and not lumen stenosis is highly correlated with atherosclerotic plaque burden in humans: a histologic study of 723 coronary artery segments using nondecalcifying methodology. J Am Coll Cardiol 1998; 31: 126–33. [4] Rumberger JA, Simons DB, Fitzpatrick LA, Sheedy PF, Schwartz RS. Coronary artery calcium area by electron-beam computed tomography and coronary atherosclerotic plaque area. A histopathologic correlative study. Circulation 1995; 92: 2157–62. [5] Schmermund A, Baumgart D, Adamzik M, et al. Comparison of electron-beam computed tomography and intracoronary ultrasound in detecting calcified and noncalcified plaques in patients with acute coronary syndromes and no or minimal to moderate angiographic coronary artery disease. Am J Cardiol 1998; 81: 141–6. [6] Erbel R, Schmermund A, Möhlenkamp S, Sack S, Baumgart D. Electron-beam computed tomography for detection of early signs of coronary arteriosclerosis. Eur Heart J 2000; 21: 720–32. [7] Detrano R, Salcedo EE, Hobbs RE, Yiannikas J. Cardiac cinefluoroscopy as an inexpensive aid in the diagnosis of coronary artery disease. Am J Cardiol 1986; 57: 1041–6. [8] Mahoney LT, Burns TL, Stanford W, et al. Coronary risk factors measured in childhood and young adult life are associated with coronary artery calcification in young adults: the Muscatine Study. J Am Coll Cardiol 1996; 27: 277–84. [9] Stanford W, Thompson BH, Weiss RM. Coronary artery calcification: clinical significance and current methods of detection. Am J Roentgenol 1993; 161: 1139–46. [10] Agatston AS, Janowitz WR, Hildner FJ, Zusmer NR, Viamonte M Jr, Detrano R. Quantification of coronary artery calcium using ultrafast computed tomography. J Am Coll Cardiol 1990; 15: 827–32. [11] Baumgart D, Schmermund A, Görge G, et al. Comparison of electron beam computed tomography with intracoronary ultrasound and coronary angiography for detection of coronary atherosclerosis. J Am Coll Cardiol 1997; 30: 57–64. [12] Achenbach S, Ropers D, Mohlenkamp S, et al. Variability of repeated coronary artery calcium measurements by electron beam tomography. Am J Cardiol 2001; 87: 210–3. [13] Adamzik M, Schmermund A, Reed JE, Adamzik S, Behrenbeck T, Sheedy PF II. Comparison of two different software systems for electron-beam CT-derived quantification of coronary calcification. Invest Radiol 1999; 34: 767–73. [14] Schmermund A, Baumgart D, Görge G, et al. Measuring the effect of risk factors on coronary atherosclerosis: coronary calcium score versus angiographic disease severity. J Am Coll Cardiol 1998; 31: 1267–73. [15] Schmermund A, Baumgart D, Görge G, et al. Coronary artery calcium in acute coronary syndromes: a comparative study of electron-beam computed tomography, coronary angiography, and intracoronary ultrasound in survivors of acute myocardial infarction and unstable angina. Circulation 1997; 96: 1461–9. [16] Möhlenkamp S, Behrenbeck TR, Pump H, et al. Reproducibility of two coronary calcium quantification algorithms in patients with different degrees of calcification. Int J Card Imaging 2001; 17: 133–42. [17] Wexler L, Brundage B, Crouse J, et al. Coronary artery calcification: pathophysiology, epidemiology, imaging methods, and clini- Electron beam computed tomography [18] [19] [20] [21] [22] [23] [24] [25] [26] [27] [28] [29] [30] [31] [32] [33] cal implications. A statement for health professionals from the American Heart Association. Writing Group. Circulation 1996; 94: 1175–92. Austen WG, Edwards JE, Frye RL, et al. A reporting system on patients evaluated for coronary artery disease. Report of the Ad Hoc Committee for Grading of Coronary Artery Disease, Council on Cardiovascular Surgery, American Heart Association. Circulation 1975; 51: 5–40. Kajinami K, Seki H, Takekoshi N, Mabuchi H. Coronary calcification and coronary atherosclerosis: site by site comparative morphologic study of electron beam computed tomography and coronary angiography. J Am Coll Cardiol 1997; 29: 1549–56. Tanenbaum SR, Kondos GT, Veselik KE, Prendergast MR, Brundage BH, Chomka EV. Detection of calcific deposits in coronary arteries by ultrafast computed tomography and correlation with angiography. Am J Cardiol 1989; 63: 870–2. Rumberger JA, Sheedy PF III, Breen JF, Schwartz RS. Coronary calcium, as determined by electron beam computed tomography, and coronary disease on arteriogram. Effect of patient’s sex on diagnosis. Circulation 1995; 91: 1363–7. Kaufmann RB, Peyser PA, Sheedy PF, Rumberger JA, Schwartz RS. Quantification of coronary artery calcium by electron beam computed tomography for determination of severity of angiographic coronary artery disease in younger patients. J Am Coll Cardiol 1995; 25: 626–32. Budoff MJ, Georgiou D, Brody A, et al. Ultrafast computed tomography as a diagnostic modality in the detection of coronary artery disease: a multicenter study. Circulation 1996; 93: 898–904. Schroeder S, Kopp AF, Baumbach A, et al. Noninvasive detection and evaluation of atherosclerotic coronary plaques with multislice computed tomography. J Am Coll Cardiol 2001; 37: 1430–5. Loecker TH, Schwartz RS, Cotta CW, Hickman JR Jr. Fluoroscopic coronary artery calcification and associated coronary disease in asymptomatic young men. J Am Coll Cardiol 1992; 19: 1167–72. Janowitz WR, Agatston AS, Kaplan G, Viamonte M Jr. Differences in prevalence and extent of coronary artery calcium detected by ultrafast computed tomography in asymptomatic men and women. Am J Cardiol 1993; 72: 247–54. Bild DE, Folsom AR, Lowe LP, et al. Prevalence and correlates of coronary calcification in black and white young adults: the Coronary Artery Risk Development in Young Adults (CARDIA) Study. Arterioscler Thromb Vasc Biol 2001; 21: 852–7. Anderson KM, Castelli WP, Levy D. Cholesterol and mortality: 30 years of follow-up from the Framingham study. JAMA 1987; 257: 2176–80. Schmermund A, Baumgart D, Möhlenkamp S, et al. Natural history and topographic pattern of progression of coronary calcification in symptomatic patients: an electron-beam CT study. Arterioscler Thromb Vasc Biol 2001; 21: 421–6. Montenegro MR, Eggen DA. Topography of atherosclerosis in the coronary arteries. Lab Invest 1968; 18: 586–93. Farb A, Burke AP, Tang AL, et al. Coronary plaque erosion without rupture into a lipid core. A frequent cause of coronary thrombosis in sudden coronary death. Circulation 1996; 93: 1354–63. Taylor AJ, Burke AP, O’Malley PG, et al. A comparison of the Framingham risk index, coronary artery calcification, and culprit plaque morphology in sudden cardiac death. Circulation 2000; 101: 1243–8. Schmermund A, Schwartz RS, Adamzik M, et al. Coronary atherosclerosis in unheralded sudden coronary death under age [34] [35] [36] [37] [38] [39] [40] [41] [42] [43] [44] [45] [46] [47] [48] [49] [50] F53 50: histopathologic comparison with ‘healthy’ subjects dying out of hospital. Atherosclerosis 2001; 155: 499–508. Burke AP, Farb A, Tashko G, Liang YH, Virmani R. Is calcification a marker of stable or unstable coronary plaques? [abstract]. Circulation 1998; 98(suppl):I-655. Keelan PC, Bielak LF, Ashai K, et al. Long-term prognostic value of coronary calcification detected by electron-beam computed tomography in patients undergoing coronary angiography. Circulation 2001; 104: 412–7. Detrano R, Hsiai T, Wang S, et al. Prognostic value of coronary calcification and angiographic stenoses in patients undergoing coronary angiography. J Am Coll Cardiol 1996; 27: 285–90. Detrano RC, Wong ND, Doherty TM, et al. Coronary calcium does not accurately predict near-term future coronary events in high-risk adults. Circulation 1999; 99: 2633–8. Arad Y, Spadaro LA, Goodman K, Newstein D, Guerci AD. Prediction of coronary events with electron beam computed tomography. J Am Coll Cardiol 2000; 36: 1253–60. Raggi P, Callister TQ, Cooil B, et al. Identification of patients at increased risk of first unheralded acute myocardial infarction by electron-beam computed tomography. Circulation 2000; 101: 850–5. Georgiou D, Budoff MJ, Kaufer E, Kennedy JM, Lu B, Brundage BH. Screening patients with chest pain in the emergency department using electron beam tomography: a follow-up study. J Am Coll Cardiol. 2001; 38: 105–10. Smith SC Jr, Amsterdam E, Balady GJ, et al. Prevention Conference V: beyond secondary prevention: identifying the highrisk patient for primary prevention: tests for silent and inducible ischemia: Writing Group II. Circulation 2000; 101:E12–6. Callister TQ, Cooil B, Raya SP, Lippolis NJ, Russo DJ, Raggi P. Coronary artery disease: improved reproducibility of calcium scoring with an electron-beam CT volumetric method. Radiology 1998; 208: 807–14. Maher JE, Bielak LF, Raz JA, Sheedy PF II, Schwartz RS, Peyser PA. Progression of coronary artery calcification: a pilot study. Mayo Clin Proc 1999; 74: 347–55. Möhlenkamp S, Pump H, Baumgart D, et al. The progression of atherosclerosis is accelerated in patients with obstructive vs. nonobstructive coronary artery disease. Eur Heart J 1998; 11: 126. Callister TQ, Raggi P, Cooil B, Lippolis NJ, Russo DJ. Effect of HMG-CoA reductase inhibitors on coronary artery disease as assessed by electron-beam computed tomography. N Engl J Med 1998; 339: 1972–8. Achenbach S, Ropers D, Maffert R. Lipid-lowering with cerivastatin significantly reduces the progression of coronary calcification measured by electron beam tomography. Eur Heart J 2001; 22(suppl): 252. Raggi P, Callister TQ, Davidson M, et al. Aggressive versus moderate lipid-lowering therapy in postmenopausal women with hypercholesterolemia: rationale and design of the Beyond Endorsed Lipid Lowering with EBT Scanning (BELLES) trial. Am Heart J 2001; 141: 722–6. Schmermund A, Erbel R, Silber S. Age and gender distribution of coronary artery calcium measured by four-slice computed tomography in 2,030 persons with no symptoms of coronary artery disease.Am J Cardiol 2002; 90: 168–73. Becker CR, Knez A, Leber A, Treede H, Haberl R, Reiser MF. Angiography with multi-slice spiral CT. Detecting plaque, before it causes symptoms. MMW Fortschr Med 2001; 143: 30–2. Kachelriess M, Ulzheimer S, Kalender WA. ECG-correlated imaging of the heart with subsecond multislice spiral CT. IEEE Trans Med Imaging 2000; 19: 888–901. Eur Heart J Supplements, Vol. 4 (Suppl F) September 2002