* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Anatomy of the Heart
Saturated fat and cardiovascular disease wikipedia , lookup
Cardiac contractility modulation wikipedia , lookup
Cardiovascular disease wikipedia , lookup
Heart failure wikipedia , lookup
Cardiothoracic surgery wikipedia , lookup
Electrocardiography wikipedia , lookup
Quantium Medical Cardiac Output wikipedia , lookup
History of invasive and interventional cardiology wikipedia , lookup
Artificial heart valve wikipedia , lookup
Aortic stenosis wikipedia , lookup
Hypertrophic cardiomyopathy wikipedia , lookup
Management of acute coronary syndrome wikipedia , lookup
Cardiac surgery wikipedia , lookup
Myocardial infarction wikipedia , lookup
Lutembacher's syndrome wikipedia , lookup
Coronary artery disease wikipedia , lookup
Mitral insufficiency wikipedia , lookup
Atrial septal defect wikipedia , lookup
Arrhythmogenic right ventricular dysplasia wikipedia , lookup
Dextro-Transposition of the great arteries wikipedia , lookup
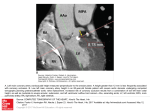
1 Anatomy of the Heart L. Maximilian Buja Embryologic Development . . . . . . . . . . . . . . . . . . . . . . . . . . 3 External Anatomy of the Heart and Great Vessels . . . . . . 7 Pericardium. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 7 Heart . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 8 Summary. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 17 Key Points recently formed cardiac tube is a single chambered structure and is composed of the following components, extending from inferior (caudal) to superior (cephalad): the sinus venosus, which connects to the major veins; the atrium; the ventricle; the bulbus cordis or conus; and the truncus arteriosus, which connects through six pairs of aortic arches to two dorsal aortae1–7 (Fig. 1.1). Initially, the single chambered heart is a straight tube residing in the pericardial cavity. The bulboventricular portion grows much more rapidly than the pericardial cavity. As a result, further extension in a longitudinal direction cannot occur, and the heart tube is forced to bend. The cephalic ventricular portion of the tube bends in a ventral and caudal direction and to the right, whereas the caudal atrial portion progresses in a dorsal and cranial direction and to the left. This process, known as d(dextro)-bulboventricular looping, results in the atrial region establishing a position superior to the ventricular region and the cardiac apex being pointed to the left (Fig. 1.1). The external shape changes are accompanied by a complex process of internal septation that leads to the formation of a four-chambered heart (Fig. 1.2). In the atrium, a septum primum forms and then develops two openings: ostium primum and ostium secundum. A septum secundum then develops on the right of the septum primum. The foramen ovale is formed in the midportion of the developing interatrial septum as a result of the growth and positioning of the septum secundum adjacent to the septum primum. The sinus venosus is incorporated into the superior portion of the developing atria. Ventricular septation is partially accomplished by the upward growth of muscular tissue to form the muscular interventricular septum. Endocardial cushion tissue develops and provides the essential tissue for formation of the atrioventricular valves, the closure of the ostium primum in the atrial septum, and the formation of the membranous interventricular septum. The primitive bulbus cordis contributes several key components of the forming heart. The proximal third of the • External looping and internal septation to form fourchambered heart. • Relationship of embryologic defects to congenital heart disease. • Structure-function relationships of the pericardium. • Distribution of the coronary arteries and anatomic variations. • Definition of anatomic right and left atria and right and left ventricles. • Structure of the four cardiac valves and concept of functional valve apparatus. • Anatomy of the cardiac conduction system and cardiac innervation. This chapter presents basic features of the anatomy of the heart and great vessels, including the embryologic development of these structures and their configuration in the mature state. Basic knowledge of cardiovascular anatomy is essential for effective diagnosis and treatment of cardiovascular diseases. Embryologic Development Basic Embryology Development of the cardiovascular system occurs in the early first trimester fetus. Beginning at about 3 weeks’ gestation, elements of splanchnic mesoderm differentiate into a primitive cardiac tube and pericardial cavity, and vascular channels form and fuse to form blood vessels.1–7 The primitive cardiac tube results from the moving together and fusion of two lateral endothelial heart tubes. Subsequently, the epimyocardial mantle and cardiac jelly develop. These components differentiate into the endocardium, producing the internal endothelial lining of the heart, the myocardium forming the muscular wall, and the epicardium or visceral pericardium producing the outside covering of the heart. The 3 CAR001.indd 3 11/24/2006 10:09:13 AM 4 chapter s us Conoventricular At.r Tr. sulcus Art. At.l. Ventricle Atrium A B 2.08 mm C 3.0 mm III IV VI Con Vent.r. Vent.l. 5.2 mm III IV VI At.r nu s Tr. Art. At.l. Co At.l. us At.r Tr. Art. Vent.r. Vent.l. D 6.0 mm E 8.8 mm FIGURE 1.1. Ventral views of human embryonic hearts that show bending of cardiac tube and establishment of major anatomic components. At., atrium; l., left; r., right; Tr. Art., truncus arteriosus. bulbus cordis forms the trabeculated part of the right ventricle; the midportion, or conus cordis, forms the outflow tracts of both ventricles; and the distal part, the truncus arteriosus, forms the proximal parts of the aorta and pulmonary artery. The junction between the primitive ventricle and the bulbus cordis is designated the primary interventricular foramen. While the ventricular septation process proceeds, the primitive ventricle develops into the major component of the definitive left ventricle, and the proximal one third of the bulbus cordis gives rise to the major component of the definitive right ventricle. Separate aortic and pulmonary channels are formed by separation of the truncus arteriosus by a spiral aorticopulmonary septum (Fig. 1.3). As a result of extensive remodeling of the double aortic arch system, the definitive major vessel system develops into a single aorta with left-sided aortic arch, a pulmonary trunk with right and left main pulmonary arteries, and the ductus arteriosus, which connects the aortic arch and the left pulmonary artery. The heart develops in the fetus as myocardial contractile activity commences, and a functional circulation is established. The foramen ovale in the interatrial septum and the ductus arteriosus remain open. In the fetus, pulmonary vascular resistance is high in the unexpanded lungs and systemic vascular resistance is low. As a result of these anatomic and physiologic features, the fetal circulation involves right to left shunting of blood across the open foramen ovale and the patent ductus arteriosus to provide oxygenated blood from the placenta to the general circulation (Fig. 1.4). After birth, the lungs expand, pulmonary vascular resistance drops, and systemic vascular resistance increases. This leads CAR001.indd 4 to functional closure of the foramen ovale and ductus arteriosus and, subsequently, fibrous closure of these structures. These closures establish complete separation of the rightsided and left-sided circulations in the mature cardiovascular system. Co Ventricle Co n us Truncus arteriosus nu I Co n I II III IV I II 1 Relationship to Congenital Heart Disease Knowledge of normal and abnormal embryologic development of the heart and blood vessels provides an essential basis for and understanding of the morphogenesis of congenital heart disease.8,9 The position of the atria is determined by general body habitus. The anatomic right atrium is defined as the atrium receiving the systemic venous drainage. The anatomic left atrium is defined as the atrium receiving the pulmonary venous drainage. In situs solitus, the right atrium is on the right and the left atrium on the left side of the body. In situs inversus, the anatomic right atrium is on the left side and the anatomic left atrium is on the right side of the body. The right and left ventricles are normally connected to the corresponding atria, but the ventricles may be inverted. Inversion of the ventricles is a major feature of a condition known as congenitally corrected transposition of the great vessels. This condition arises when the primitive heart undergoes levo(l)-bulboventricular looping rather than the usual dextro(d)-bulboventricular looping; hence, the alternative designation of corrected transposition as l-transposition. Other positional anomalies of the heart include dextrocardia and right-sided aortic arch. Abnormalities in atrial septation give rise to three types of atrial septal defects; in order of frequency they are the ostium secundum defect, ostium primum defect, and sinus venosus defect. Ostium secundum defects are located in the midportion of the interatrial septum and result from inadequate formation of septum secundum and/or septum primum to cover the foramen ovale. Ostium primum defects are located in the inferior portion of the interatrial septum and result from a defect in formation of endocardial cushion tissue. Severe endocardial cushion deficiency can lead to a common atrioventricular canal anomaly with ostium primum atrial septal defect, membranous ventricular septal defect, and abnormal atrioventricular valve. The sinus venosus atrial septal defect is located in the superior portion of the interatrial septum and results from defective incorporation of the primitive sinus venosus into the forming heart; this defect is often associated with partial anomalous pulmonary venosus drainage into the right atrium. Abnormalities in ventricular septation give rise to ventricular septal defects, usually in the region of the membranous septum. Formation of the membranous interventricular septum involves contributions from the conal ridges of the bulbus cordis and the endocardial cushions. Deficiencies in contributions from these embryonic structures leads to the formation of membranous ventricular septal defects. Abnormalities in septation of the great vessels give rise to congenital complete transposition of the great vessels, or d-transposition, since the abnormal septation of the great vessels occurs with normal dextro-bulboventricular looping and the atria and ventricles are in normal position. Congenital complete transposition is often accompanied by 11/24/2006 10:09:13 AM 5 a natom y of t h e h e a rt Septum spurium Interatrial septum primum Ostium I Atrio-vent. canal Intervent. septum A B 4–5 mm 6–7 mm Septum II S. sp. Right atrium Ostium II (opening) Ostium I (closing) Septum I Ostium II Septum spurium Septum I Ostium I (closed) Septum II (caudal limb) A_V. canal cushion Intervent. foramen (closes at 15–17 mm.) Left ventricle C D 8–9 mm Septum II S. sp. 12–15 mm Crista Ostium II terminalis in septum I S II Foramen ovale S II Functional outlet F.O. Septum I (valvula F.O.) Atriovent. valves Bundle of his E 25–30 mm F 100 mm. to birth FIGURE 1.2. Longitudinal sections of embryonic heart in frontal plane that show extent of growth of various cardiac septa at progressive stages of development. These diagrams depict the stages of partitioning of the human embryo. Stippled areas indicate the distribution of endocardial cushion tissue; muscle is shown in diagonal hatching, and epicardium in solid black. The lightly stippled areas in the atrioventricular canal in B and C indicate location of dorsal and ventral endocardial cushions of the atrioventricular canal before they have grown sufficiently to fuse with each other in the plane of the diagram. atrial or ventricular septal defects or other congenital lesions. Severe maldevelopment of the heart can give rise to a hypoplastic left heart or hypoplastic right heart and associated valvular atresia. Other anomalies include vascular rings, persistent patent ductus arteriosus, and coarctation of the aorta. Cardiovascular malformations occur in about 0.8% of live births.10 However, the incidence of congenital heart disease is significantly higher because cardiac malforma- tions occur 10 times more often in stillborn than in liveborn infants. Children with congenital heart disease are predominantly male. However, specific defects have a defi nite sex preponderance: females have a higher incidence of patent ductus; arteriosus and atrial septal defect are more common in females; and males have a higher incidence of valvular aortic stenosis, congenital aneurysm of the sinus of Valsalva, coarctation of the aorta, tetralogy of Fallot, and congenital complete transposition of the great arteries.10 CAR001.indd 5 11/24/2006 10:09:14 AM 6 chapter 1 Dextrodorsal truncus ridge Truncus arteriosus communis Dextrodorsal conus ridge Right atrium Pulmonary channel S.-v. truncus ridge Aortic channel S.-v. conus ridge Conovent. flange Arrow in aortic channel Atrioventricular canal Left atrium Conus art. Ventral atriovent. canal cushion Dorsal atriovent. canal cushion Right ventricle Left ventricle Interventricular septum A B FIGURE 1.3. Frontal plane dissections of developing heart show important relations in establishing aortic and pulmonary outlets. The truncus arteriosus has been drawn with its cut end turned Superior vena cava Crista terminalis Septum II Limbus of foramen ovale upward, in order to show the absence of truncus ridges in the early stages and their relationships in later stages. Septum I (remnant) Margin of interatrial foramen II Septum I = valvula foraminis ovalis Orifices of pulmonary veins Valve of inferior vena cava Valve of coronary sinus Tricuspid valve Septum membranaceum Left atrium Mitral valve Tendinous cords Papillary muscle Interventricular septum (muscular portion) Right ventricle FIGURE 1.4. Schematic drawing to show interrelations of septum primum and septum secundum during the latter part of fetal life. Note especially that the lower part of septum primum is positioned so as to act as a one-way valve at the oval foramen in septum secundum. The split arrow indicates that a considerable part of the blood CAR001.indd 6 Trabeculae carneae of left ventricle from the inferior vena cava passes through the foramen ovale to the left atrium while the remainder eddies back into the right atrium to mix with the blood being returned by way of the superior vena cava. 11/24/2006 10:09:14 AM 7 a natom y of t h e h e a rt External Anatomy of the Heart and Great Vessels Pericardium The anatomy of the heart and great vessels has been well documented in previous publications.11–17 The normal location of the heart is in the mediastinum to the left of the midline with the cardiac apex pointed to the left (Fig. 1.5). The heart is rotated and tilted in the chest, and as a result about two thirds of the anterior surface of the heart is composed of the right ventricle, and the left third of the anterior surface is composed of the left ventricle. The right inferior border (obtuse border) of the heart is formed by the right ventricle and the left lateral border is formed by the left ventricle. Located superior to the right and left ventricles are the right and left auricles of the right and left atria, respectively. The anterior superior surface of the heart is constituted by the outflow portion (conus) of the right ventricle and the pulmonary trunk, which extends from right to left as it exits the pericardium. The pulmonary trunk then gives rise to the left and right main pulmonary arteries. The aorta is located posterior to the pulmonary trunk. The aorta takes origin from the left ventricle and is oriented from left to right as it exits the pericardium. The aorta then curves to the left and inferiorly, creating a leftsided aortic arch. The aortic arch gives origin to the right innominate, left common carotid, and left subclavian arteries. The aorta then continues inferiorly as the descending thoracic aorta. Innominate artery Right innominate vein R. int. mam. vein Thymus gland Reflection of pericardium Vena cava sup. Asc. aorta Probe in transv. sinus Right auricle Right atrium The pericardial cavity is a fluid-filled space that surrounds the heart and proximal great vessels. The pericardial space is enclosed by a thin layer of connective tissue that is lined by a single layer of mesothelial cells. There are two components: the visceral and parietal pericardium. The visceral pericardium covers the epicardium of the heart, and the parietal pericardium forms the outer layer. The superior extent of the pericardial cavity representing the transition of visceral to parietal pericardium occurs approximately 2 to 3 cm superior to the heart at the level of the great vessels, thereby enclosing the proximal aorta and pulmonary trunk in the pericardial cavity (Fig. 1.5). The pericardial cavity normally contains about 20 cc of serous fluid, which serves to lubricate the heart and facilitate cardiac motion. The pericardium can be affected by a variety of inflammatory and neoplastic conditions. Hemorrhage into the pericardium may occur as a result of cardiac rupture, usually secondary to acute myocardial infarction, or rupture of the proximal aorta following aortic dissection. The severity of cardiac dysfunction secondary to pericardial disease is influenced acutely by the amount and rapidity of fluid accumulation in the pericardial cavity and chronically by the severity of inflammation and fibrosis. Rapid accumulation of 100 to 200 mL of fluid or blood in the pericardial cavity can induce cardiac tamponade, whereas the slow accumulation of several hundred milliliters can be accommodated in the Left subclavian artery Left innominate vein Pleura Pulmonary artery Pleura Fibrous pericardium Serous pericardium Conus arteriosus Left auricle Coronary sulcus Right ventricle FIGURE 1.5. Ventral view of heart in situ with the pericardial sac opened. CAR001.indd 7 Margo acutus Incisura apicis cordis Pleura Margo obtusus Left ventricle Anterior longitudinal sulcus Apex 11/24/2006 10:09:14 AM 8 chapter cavity with stretching of the pericardial lining before impaired cardiac function develops. Heart Basic Structure The anatomy of the heart has been documented in detail.11–17 The heart is composed of three layers: the epicardium, the myocardium, and the endocardium. The epicardium consists of fatty connective tissue and is lined by the visceral pericardium. The major coronary arteries and veins traverse the epicardium. The myocardium constitutes most of the mass of the heart and is composed of cardiac myocytes, vessels, and connective tissue. The cardiac myocytes represent approximately 80% of the mass but only 20% of the number of cells in the myocardium. The endocardium is divided into the nonvalvular (visceral) and valvular endocardium. The endocardium consists of thin fibrocellular connective tissue, which is lined by a single layer of endothelial cells. Cardiac Dimensions Several sources have provided information regarding dimensions and measurements of the heart.11–17 The weight of the heart varies in relationship to body dimensions, including length and weight. Hudson11 has published a useful guide regarding fresh heart weight in males and females. The adult male heart weight has the following parameters: 0.45% of body weight, average 300 g, range 250 to 350 g. The adult female heart weight has the following parameters: 0.40% of body weight, average 250 g, range 200 to 300 g. Selective measurements of right ventricular and left ventricular weights can be made, and ranges have been established for determination of selective enlargement of right and left ventricles as Right pulmonary veins 1 well as biventricular enlargement.15 The thickness of the walls of the cardiac chambers is as follows: right and left atria, 0.1 to 0.2 cm; right ventricle, 0.4 to 0.5 cm; left ventricle, 1.2 to 1.5 cm (free wall, excluding papillary muscles and large trabeculae). The average circumferences of the cardiac valves are as follows: aortic, 7.5 cm; pulmonic, 8.5 cm; mitral, 10.0 cm; and tricuspid, 12.0 cm. Coronary Vasculature The anatomy and physiology of the coronary circulation have been described in detail.18–25 In the normal heart, oxygenated blood is supplied by two coronary arteries that are the first branches of the aorta. The origin of the left and right coronary arteries from the aorta is through their ostia positioned in the left and right aortic sinuses of Valsalva, which are located just distal to the left and right cusps, respectively, of the aortic valve (Figs. 1.5, 1.6, and 1.7). The left main coronary artery is a short vessel with a length of 0.5 to 1.5 cm. The left main coronary artery divides into left anterior descending and left circumflex branches and, occasionally, a left marginal branch. The left anterior descending coronary artery and its left diagonal and septal branches supply the anterior portion of the left ventricles and interventricular septum. The left circumflex coronary artery and its circumflex marginal branches supply the lateral left ventricle. The right coronary artery supplies the right ventricle and, in about 90% of hearts, it extends posteriorly to give rise to the posterior descending coronary artery. There is considerable variation in the anatomic distribution of the coronary arterial branches. However, in most hearts, branches from both the left circumflex and right coronary arteries contribute to the blood supply of the posterior left ventricle, resulting in a so-called balanced circulation. In about 10% of hearts, the right coronary artery is small, and the left circumflex coronary artery gives origin Left pulmonary veins Left auricle Vena cava superior Left atrium Aorta Posterior and left aortic semilunar valves Right aortic semilunar valve Right coronary artery Small cardiac vein Anterior cardiac veins Preventricular. arteries Right marginal artery Right ventricle Circumflex branch Left coronary artery Left aortic sinus Great cardiac vein Left pulmonary semilunar valve Right and anterior pulmonary semilunar valves Pulmonary artery Conus arteriosus Anterior descending branch of left coronary artery Left ventricle Anterior longitudinal sulcus Incisura apicis cordis CAR001.indd 8 FIGURE 1.6. Cephalic view of the heart with the epicardium removed to expose the injected coronary vessels. 11/24/2006 10:09:15 AM 9 a natom y of t h e h e a rt Vena cava superior Superior right pulmonary vein Superior left pulmonary vein Inferior right pulmonary vein Inferior left pulmonary vein Left atrium Oblique vein of left atrium Interatrial sulcus Terminal sulcus Right atrium Vena cava inferior Valvula venae cavae Small cardiac vein Right coronary artery Right ventricle Middle cardiac vein Posterior descending branch of right coronary artery Great cardiac vein Coronary sinus Posterior vein of left ventricle Diaphragmatic surface of left ventricle Posterior longitudinal sulcus FIGURE 1.7. Dorsocaudal view of the heart with the epicardium removed to expose the injected coronary vessels. to the posterior descending coronary artery and provides the sole blood supply for the posterior left ventricle, creating a left dominant circulation. Rarely, the converse right dominant circulation exists when the left circumflex is small and the posterior left ventricle is supplied primarily by left ventricular branches of the right coronary artery. The major blood supply to the sinoatrial node via the sinus node artery is derived from the proximal right coronary artery in about 60% of hearts and from the left circumflex coronary artery in about 40% of hearts.18 The major atrioventricular nodal artery is derived from the coronary artery that gives rise to the posterior descending branch, which is the right coronary artery in about 90% and the left circumflex coronary artery in about 10% of hearts. The epicardial coronary arteries deliver oxygenated blood to the intramyocardial arteries, arterioles, and capillaries leading to oxygen and substrate extraction in the myocardium (Fig. 1.8). A small amount of unoxygenated blood flows directly into the ventricular cavities via the thebesian veins. However, most desaturated blood traverses the myocardial venules and veins into the epicardial veins, which drain into the coronary sinus located in the inferoposterior region of the right atrium. Collateral blood vessels form during embryologic development of the heart, and they connect different components of the coronary arterial circulation.18–25 The coronary collateral system is composed of four types of vessels: intramural branches of the same coronary artery (homocoronary collaterals); intramural branches of two or more coronary arteries (intercoronary collaterals); atrial branches, which connect with the vasa vasorum of the aorta and other vessels (extra- CAR001.indd 9 cardiac collaterals); and intramural branches, which communicate with the cardiac cavities (arterioluminal vessels). In the normal adult heart, the collateral vessels are thin walled, small channels, usually less than 50 µm in diameter, and they contribute little to total coronary blood flow. In response to coronary arterial narrowing and myocardial ischemia, the capacity of the coronary collateral system can greatly increase. The myocardial collateral vessels can increase in diameter into the range of 200 to 600 µm or Endocardium Myocardium Epicardium Arterioluminal vessel Myocardial sinusoid Intertrabecular space Coronary artery Anastomosis between myocardial sinusoids Arteriosinusoidal vessel Trabeculae carneae Anastomoses between coronary arteries Arteriovenous anastomosis Capillary bed Myocardial sinusoid Venovenous anastomosis Capillaries emptying into myocardial sinusoids Coronary vein Thebesian vein Anastomosis between thebesian veins FIGURE 1.8. Diagram of the ventricular wall, showing the relationship between the various intramural vascular channels. 11/24/2006 10:09:15 AM 10 chapter 1 greater, develop muscular media, and transport significant amounts of blood flow (Fig. 1.9). In addition to the types of collateral vessels described above, collateral channels also can develop proximal and distal to a stenosis in a given coronary artery. Right and Left Atria and Ventricles FIGURE 1.9. Radiograph of postmortem coronary arteriogram demonstrating an extensive coronary collateral system in a case of coronary heart disease. The proximal part of the left anterior descending artery is obliterated by old disease (O). The more distal part of the anterior descending artery (A) has fi lled through a rich anastomotic network (X) that has formed in the substance of the interventricular septum between branches of the anterior (A) and posterior (P) descending arteries. The right and left atria and right and left ventricles have distinctive anatomic features (Figs. 1.10 to 1.17). The anatomic right ventricle is characterized as follows: distinct muscular infundibulum (conus) arteriosus, which separates the right semilunar (pulmonary) valve and the right atrioventricular (tricuspid valve); single large anterior papillary muscle; and coarse trabecular muscles (trabeculae carneae cordis) at the apical and inflow portion of the chamber.11–17 Key landmarks of the right ventricular infundibular (conus) region from inferior to superior are the membranous interventricular septum; the crista supraventricularis, an inverted V-shaped structure with parietal and septal limbs; and the pulmonic valve. The anatomic left ventricle has the following features: fibrous continuity of annulus of left semilunar (aortic) valve and anterior leaflet of left atrioventricular (mitral) valve, two well-developed papillary muscles (anterolateral and posteromedial), and fine trabecular muscles at the apical and inflow portion of the chamber.11–17 These features allow determination of the anatomic right ventricle and anatomic left ventricle in complex congenital anomalies involving displacement of the various components of the heart. Cardiac Valves The four-chambered heart has four valves: the right semiluminar or pulmonic valve; the right atrioventricular or tricuspid valve; the left semilunar or aortic valve; and the left Ligamentum arteriosum Left pulmonary artery Aorta Probe in transverse sinus Right pulmonary artery Right auricle Left pulmonary veins Left atrium Foramina venarum minimarum Right pulmonary veins Interatrial sulcus Limbus fossae ovalis Fossa ovalis Orifice of coronary sinus Valvula sinus coronarii Valvula venae cavae Vena cava inferior Vena cava superior Right ventricle Crista terminalis Left ventricle Right atrium Tricuspid valve: Anterior cusp Medial cusp Posterior cusp Musculus pectinatus Right coronary artery Small cardiac vein CAR001.indd 10 FIGURE 1.10. Right anterior oblique view of the excised heart, with the right atrium opened to show its internal configuration. 11/24/2006 10:09:16 AM 11 a natom y of t h e h e a rt Right coronary artery Ascending aorta Superior vena cava Pulmonary cone Right auricular appendage Crista supraventricularis Crista terminalis Papillary muscle of conus Pulmonary vein Interventricular septum membranaceum Limbus fossae ovalis FIGURE 1.11. Right side of the heart opened in a plane approximately parallel to the septa, to show the interior of the right atrium and the right ventricle. A segment of the septal leaflet of the tricuspid valve has been removed to expose more fully the region of the membranous portion of the interventricular septum. Septal cusp of tricuspid valve (cut) Valve of inferior vena cava (eustachian v.) Anterior papillary muscle (cut) Inferior vena cava Posterior papillary muscle (cut) Valve of coronary sinus (thebesian v.) Aorta Crista supraventricularis Vena cava superior Papillary muscle of conus Conus arteriosus Pulmonary artery Anterior pulmonary semilunar valve Crista terminalis Limbus fossae ovalis Fossa ovalis FIGURE 1.12. Ventral view of the heart with the walls of the right atrium and ventricle opened to show their internal configuration. This heart has an unusually well-developed moderator band, and the position of the foramen ovale is somewhat more cephalic than usual. CAR001.indd 11 Right atrium Right coronary artery Vena cava inferior Tricuspid valve: Anterior cusp Anterior papillary muscle of left ventricle Posterior cusp Medial cusp Left ventricle Trabecula carnea Moderator band Posterior papillary muscle Muscular interventricular septum Chorda tendineae Anterior papillary muscle 11/24/2006 10:09:18 AM 12 chapter FIGURE 1.13. Photograph of opened right heart demonstrating actual structures depicted in the illustrations. Note the muscular infundibulum separating the tricuspid and pulmonic valves. CA, 1 conus arteriosus, i.e., infundibulum; CS, coronary sinus; PV, pulmonic valve; RA, right atrium; RV, right ventricle. Aorta Pulmonary trunk Great cardiac vein Left coronary artery Right semilunar valve of aorta Interventricular septum membranaceum Interventricular septum musculare Superior vena cava Orifice of right coronary artery Right superior pulmonary vein Adherent margin of valv. foram ov. Mitral valve (cut) Coronary sinus Papillary muscle (cut) FIGURE 1.14. Left side of the heart opened in a plane approximately parallel to the septa, to show the interior of the left atrium and left ventricle. A portion of the anterior leaflet of the mitral valve has CAR001.indd 12 Inferior vena cava been removed to expose more fully the region of the membranous portion of the interventricular septum and the aortic orifice. 11/24/2006 10:09:19 AM 13 a natom y of t h e h e a rt Left atrium Valve of inferior vena cava Septum membranaceum (atriovent. part.) Septum membranaceum (interventricular part.) Atrioventricular bundle Tricuspid valve Papillary muscle Mitral valve Papillary muscles Interventricular septum FIGURE 1.15. Frontal section through a heart fi xed in diastole, showing a ventral view of the dorsal portion. The plane of section passes through the septum membranaceum and both atrioventricular ostia. FIGURE 1.16. Photograph showing the relationship of the membranous interventricular septum (lighted) to the tricuspid annulus and septal leaflet of the tricuspid valve. While the membranous interventricular septum is entirely contained in the left ventricle, the superior portion of the interventricular septum extends superior to the tricuspid annulus. This anatomic relationship allows for left ventricular to right atrial shunts in certain pathologic states, such as infective endocarditis of the aortic valve. MIS, membranous interventricular septum; SLTV, septal leaflet of the tricuspid valve. CAR001.indd 13 FIGURE 1.17. Photograph of opened left ventricle showing continuity of the anterior mitral leaflet and the annulus of the aortic valve. AML, anterior mitral leaflet; LC, left cusp of aortic valve; NCC, noncoronary cusp of aortic valve; PPM, posterior papillary muscle; RC, right coronary cusp of aortic valve. 11/24/2006 10:09:21 AM 14 chapter Pulmonary annulus fibrosus 1 Conus ligament Ant. descending br. Right coronary art. Left coronary art. Circumflex br. Left fibrous triangle Aortic annulus fibrosus Atrioventricular bundle Right fibrous friangle Annulus fibrosus of mitral orifice Annulus fibrosus of tricuspid valve Anastomosing branch FIGURE 1.18. Ventricular portion of the heart viewed from above with the atria removed, to show the four cardiac valves and the fibrous triangles, the annuli fibrosi, and the attachment of the ven- Post. descending br. of right coronary a. tricular musculature to them. Epicardial fat has been removed from the atrioventricular sulcus in order to show the coronary arteries. atrioventricular or mitral valve (Figs. 1.18 to 1.21). The pulmonic and aortic semilunar valves each have three cusps separated by three commissures, and the cusps insert into a fibrous connective tissue annulus. The aortic valve cusps are designated as the left and right cusps in relationship to the coronary ostia and a third, noncoronary cusp. With valve closure during diastole, the cusps of the aortic and pulmonic valves make contact along a line about 1 to 2 mm below the free margin. The central points of maximal contact are espe- cially prominent and are known as the noduli of Arantius. The most common congenital valve lesion is a malformed aortic valve, either a unicuspid or bicuspid valve. The tricuspid or right atrioventricular valve has three leaflets (cusps) that are separated by three commissures and insert into a fibrous annulus. The leaflets are designated as the lateral, medial, and anterior leaflets. The leaflets are attached to the ventricular muscle by multiple chordae tendineae, which extend from the ventricular surfaces of the Pulmonary semilunar valve Pulmonary conus Septal cusp of tricuspid valve Ant. desc. branch of left coronary art. Noncoronary cusp of aortic semilunar v. Septum membranaceum Bundle of his Muscular part of interventricular septum FIGURE 1.19. Interior of the ventricular base of the heart, exposed by removal of the apical half of the ventricles. The interventricular septum has been partially removed to show the relations of the CAR001.indd 14 Mitral valve interventricular septum membranaceum, and the mitral, the tricuspid and the aortic valves. 11/24/2006 10:09:24 AM a natom y of t h e h e a rt leaflets to the mural ventricular wall as well as a welldeveloped anterior papillary muscle and a diminutive structure, the papillary muscle of the conus. The mitral or left atrioventricular valve has a highly developed and coordinated group of structures that are referred to as the mitral valve apparatus (Figs. 1.18 to 1.21). The mitral valve apparatus consists of fibrous annulus, anterior and posterior leaflets, anterior and posterior commissures, multiple chordae tendineae, and anterolateral and posteromedial papillary muscles. Chordae tendineae extend from the ventricular surfaces of both leaflets and attach to both papillary muscles. Chordae tendineae exhibit a branching pattern with primary chordae arising from the papillary muscles and branching into secondary and then tertiary chordae, which insert into the valvular leaflets. Mitral valve dysfunction can result from a wide variety of pathologic processes affecting any component of the mitral apparatus, including the myocardium. The anterior mitral leaflet is usually a single structure, whereas the posterior leaflet may be divided into two or three scallops (Fig. 1.21). The anterior mitral leaflet normally has a significantly larger area and longer length from annulus to free margin that the posterior mitral leaflet. As a result, the anterior leaflet contributes about two thirds of the area of leaflet tissue involved in closure of the mitral orifice during systole. Expansion of the posterior leaflet to equal the length 15 FIGURE 1.21. Photograph of mitral valve showing a portion of the anterior mitral leaflet and the posterior leaflet consisting of multiple scallops. AML, anterior mitral leaflet; PML, posterior mitral leaflet. of the anterior leaflet from annulus to free margin is an important anatomic feature of myxomatous degeneration of the mitral valve. Other features include general redundancy, thickening, and a glistening white appearance of leaflet tissue and chordae tendineae. Cardiac Conduction System and Cardiac Innervation FIGURE 1.20. Photograph of left ventricle showing aortic valve and a portion of the mitral apparatus, including the anterior mitral leaflet, chordae tendineae, and papillary muscles. AML, anterior mitral leaflet; APM, anterior papillary muscle; CT, chordae tendineae; LC, left cusp of aortic valve; PPM posterior papillary muscle. CAR001.indd 15 The anatomy of the cardiac conduction system has been defined by meticulous study (Fig. 1.22).26,27 The sinoatrial node is located in the superficial subepicardium of the superior right atrium at the junction of the superior vena cava and the right auricular appendage. The sinoatrial node contains myocytes that are specialized for the generation of electrical impulses and constitute the cardiac pacemaker. The electrical impulse propagates selectively along certain paths in the atria; however, the existence of anatomically distinct conduction pathways in the atria is difficult to demonstrate. Atrioventricular conduction is accomplished by specialized structures, the more proximal atrioventricular node (node of Tawara) and the more distal atrioventricular bundle (bundle of His). The atrioventricular node is positioned in the inferior interatricular septum just anterior and medial to the ostium of the coronary sinus. The specialized conduction tissue then extends through the fibrous skeleton separating the atria and ventricles to connect to the bundle of His, which is located at the apex of the muscular interventricular septum. The His bundle gives origin to the right and left bundle branches, which extend through the subendocardium of the interventricular septum into the right and left ventricular free walls. The heart has a dual innervation from the sympathetic (thoracolumbar) and parasympathetic (craniosacral) divisions of the autonomic nervous system (Fig. 1.23). These nerves interact with the conduction tissue to provide neural modulation of cardiac function. 11/24/2006 10:09:24 AM 16 chapter Superior vena cava Right pulmonary vein Sino-atrial node 1 Left pulmonary vein Left atrial wall Fossa ovalis Mitral valve Coronary orifice Inferior vena cava Septum membranaceum Atrio-ventricular node Bifurcation of bundle Main atrioventricular bundle (bundle of his) Left branch of bundle Right branch of bundle Branch of bundle in moderator band Purkinje fibers under endocardium of papillary musc. Branch under septal endocard. Superior cervical ganglion Superior cardiac nerve Inferior cervical ganglion Vagus nerve Inferior cardiac branch Deep cardiac plexus FIGURE 1.22. Schematic diagram of heart opened frontally, demonstrating the location and relations of the several parts of the sinoatrial and atrioventricular conduction system. Superior cardiac nerve Middle cardiac nerve Superior cardiac branch Inferior cervical ganglion First thoracic ganglion Ansa of vieussens Superficial cardiac plexus Ganglion of wrisberg Pulmonary plexus Right coronary plexus Left coronary plexus FIGURE 1.23. Ventral view of thorax to show the nerve supply to the heart. CAR001.indd 16 11/24/2006 10:09:27 AM a natom y of t h e h e a rt Summary This chapter presented a description and illustration of the embryologic and anatomic features of the heart and great vessels. The embryologic development of the cardiac structures was presented, and the relationship of cardiac development to congenital heart diseases was discussed. Key components of the mature heart were described and illustrated, including the pericardium, the coronary arteries, the four cardiac valves, the right and left atria and ventricles, and the cardiac conduction system and cardiac innervation. The relationship of structure to function was considered. Understanding cardiovascular anatomy is important for the diagnosis and treatment of cardiovascular diseases. References 1. Patten BM. The development of the heart. In: Gould SE, ed. Pathology of the Heart and Blood Vessels, 3rd ed. Springfield, IL: Charles C Thomas, 1968:20–90. 2. Patten BM. Section on heart. In: Human Embryology, 2nd ed. New York: McGraw-Hill, 1953:656–705. 3. Patten BM. The development of the circulatory system. In: Foundations of Embryology. New York: McGraw-Hill, 1958: 484–539. 4. Sadler TW. Cardiovascular system. In: Langman’s Medical Embryology, 5th ed. Baltimore: Williams & Wilkins, 1985: 168–214. 5. Olson EN, Srivastava D. Molecular pathways controlling heart development. Science 1996;272:671–676. 6. Patten BM. Developmental defects at the foramen ovale. Am J Pathol 1938:14:135–161. 7. Kramer TC. The partitioning of the truncus and conus and the formation of the membranous portion of the interventricular septum in the human heart. Am J Anat 1942;71: 343–370. 8. Edwards JE. Congenital malformations of the heart and great vessels. In: Gould SE, ed. Pathology of the Heart and Blood Vessels, 3rd ed. Springfield, IL: Charles C Thomas, 1968: 262–478. 9. Berry CL. Congenital heart disease. In: Pomerance A, Davies MJ, eds. The Pathology of the Heart. Oxford: Blackwell Scientific Publications, 1975:533–578. 10. Friedman WF. Congenital heart disease in infancy and childhood. In: Braunwald E, ed. Heart Disease. A Textbook of Cardiovascular Medicine. 4th ed. Philadelphia: WB Saunders, 1992:887–965. CAR001.indd 17 17 11. Hudson REB. Structure and function of the heart. In: Cardiovascular Pathology, vol 1. Baltimore: Williams & Wilkins, 1965:1–52. 12. Barry A, Patten BM. The structure of the adult heart. In: Gould SE, ed. Pathology of the Heart and Blood Vessels, 3rd ed. Springfield, IL: Charles C Thomas, 1968:91–130. 13. Patten BM. The cardiovascular system. In: Morris’s Human Anatomy, 10th ed. Philadelphia: Blakiston, 1942:582–785. 14. Reiner L. Gross examination of the heart. In: Gould SE, ed. Pathology of the Heart and Blood Vessels, 3rd ed. Springfield, IL: Charles C Thomas, 1968:1111–1149. 15. Davies MJ, Pomerance A, Lamb D. Techniques in examination and anatomy of the heart. In: Pomerance A, Davies MJ, eds. The Pathology of the Heart. Oxford: Blackwell Scientific, 1975:1–48. 16. Ludwig J, Lie JT. Heart and vascular system. In: Ludwig J, ed. Current Methods of Autopsy Practice, 2nd ed. Philadelphia: WB Saunders, 1979:21–50, 668–673. 17. Silver MM, Freedom RM. Gross examination and structure of the heart. In: Silver MD, ed. Cardiovascular Pathology, 2nd ed. New York: Churchill Livingstone, 1991:1–42. 18. James TN. The coronary circulation and conduction system in acute myocardial infarction. Prog Cardiovasc Dis 1968; 10:410–446. 19. Shaper W. The Collateral Circulation of the Heart. New York: American Elsevier, 1971. 20. Crawford T. The anatomy of the coronary arteries. In: Pathology of Ischaemic Heart Disease. London: Butterworths, 1977:4–17. 21. Gregg DE, Patterson RE. Functional importance of coronary collaterals. N Engl J Med 1980;303:1404–1406. 22. Willerson JT, Hillis LD, Buja LM. Pathogenesis and pathology of ischemic heart disease. In: Ischemic Heart Disease: Clinical and Pathophysiological Aspects. New York: Raven Press, 1982: 7–83. 23. Marcus ML. The Coronary Circulation in Health and Disease. New York: McGraw-Hill, 1983. 24. Baroldi G. Diseases of extramural coronary arteries. In: Silver MD, ed. Cardiovascular Pathology, 2nd ed. New York: Churchill Livingstone, 1991:487–563. 25. Boudoulas H, Gravanis MB. Ischemic heart disease. In: Gravanis MD, ed. Cardiovascular Disorders: Pathogenesis and Pathophysiology. St. Louis, MO: Mosby, 1993:14–16. 26. Lev M. The conduction system. In: Gould SE, ed. Pathology of the Heart and Blood Vessels, 3rd ed. Springfield, IL: Charles C Thomas, 1968:180–220. 27. Hudson REB. The conducting system: anatomy, histology and pathology in acquired disease. In: Silver MD, ed. Cardiovascular Pathology, 2nd ed. New York: Churchill-Livingstone, 1991:1367–1428. 11/24/2006 10:09:28 AM CAR001.indd 18 11/24/2006 10:09:29 AM