* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download WEEK 2: 16 J
Matter wave wikipedia , lookup
Hidden variable theory wikipedia , lookup
Quantum state wikipedia , lookup
EPR paradox wikipedia , lookup
Bremsstrahlung wikipedia , lookup
James Franck wikipedia , lookup
Franck–Condon principle wikipedia , lookup
Double-slit experiment wikipedia , lookup
Relativistic quantum mechanics wikipedia , lookup
Renormalization wikipedia , lookup
History of quantum field theory wikipedia , lookup
Molecular orbital wikipedia , lookup
Delayed choice quantum eraser wikipedia , lookup
Quantum key distribution wikipedia , lookup
Bohr–Einstein debates wikipedia , lookup
Particle in a box wikipedia , lookup
Ultrafast laser spectroscopy wikipedia , lookup
Tight binding wikipedia , lookup
X-ray photoelectron spectroscopy wikipedia , lookup
Astronomical spectroscopy wikipedia , lookup
Auger electron spectroscopy wikipedia , lookup
Wave–particle duality wikipedia , lookup
Theoretical and experimental justification for the Schrödinger equation wikipedia , lookup
Quantum electrodynamics wikipedia , lookup
X-ray fluorescence wikipedia , lookup
Atomic orbital wikipedia , lookup
Atomic theory wikipedia , lookup
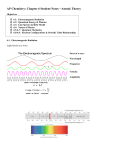
WEEK 2: 16 JAN THRU 22 JAN; LECTURES 5-6 Learning Objectives Interpretation of Line spectrum (What is happening that causes the emission lines?) Know that quantized emissions represent energy transitions Be able to interpret line spectra by assigning lines to transitions Connect line spectra to the energy of emitted photons, the color of light emitted, and its wavelength Bohr Model of the hydrogen atom: where is the electron in the H atom? Qualitative Goals: Connect the line spectrum of hydrogen to the energy level diagram of hydrogen using the Bohr model. Define and describe the energy of an electron in the hydrogen atom + Connect emission and absorption to pictures of electrons in orbit; We are using He ion for this goal Have a qualitative ability to describe emission and absorption using the principal quantum number. Know the sign of the energy change (positive or negative) when emission or absorption occurs. Quantitative Goals Calculate E given ni and nf (Calculate energy of emitted photon.) + Calculate ionization energy for H atom or He (knowing that n = for ionization) Know that the energy change is negative if the photon is emitted. Orbitals: Solutions to Schrödinger equation What is meant by the “electron density” (or the probability of finding an electron)? Know the qualitative meaning of wavefunctions, which are solutions to the Schrödinger Equation. Describe the meaning of probability density, and electron density as the location of an electron. Quantum Numbers Be familiar with the names of principle, angular, and magnetic quantum numbers Define shell, subshell and orbital Know and follow rules for allowed combination of quantum numbers Understand the relationship between quantum numbers and size, shape, and orientation of an orbital Know orbital names. Describe the value of electron density in regions of space using electron density plots or contour diagrams Interpret an electron density plot, and define nodes and lobes of orbitals Make connections between electron configuration and orbitals; know that quantum numbers describe orbitals, and orbitals sufficiently describe the location of the electron ------------------------------------------------------------------------------------------------------------------------ Week 2 CHEM 110 Student Packet - Spring 2013 30 WEEK 2: 16 JAN THRU 22 JAN; LECTURES 5-6 Homework Problems Due: Thurs. Jan 24 1. The brightest emission line in the line spectrum of potassium is at 535nm. What is the energy of the photon emitted? 2. The brightest emission line of an element is 420nm. What is the color of the flame? 3. In the Bohr model for the hydrogen atom, in which orbit does an electron have higher overall energy: n = 1 or n = 5? 4. What is the maximum number of electrons that can have n = 3 and ms = + ½? 5. How many possible orbitals are there with n = 3 and m = 1? 6. Give the set of quantum numbers that describes each of the following orbitals assuming they are all in the third shell. A. Week 2 B. C. D. CHEM 110 Student Packet - Spring 2013 31 WEEK 2: 16 JAN THRU 22 JAN; LECTURES 5-6 7. If the Bohr model is used, what frequency of light would be required for ionization of hydrogen? A. B. C. D. E. 10. Which of the following statements is/are true for the Bohr model of the hydrogen atom? 14 6.17 10 Hz 3 1.31 10 Hz 15 3.29 10 Hz 10 4.31 10 Hz None of the above is within 5% of the correct answer 1. 2. 3. 8. Which of the following electron transitions in a hydrogen atom results in the greatest release of energy from the atom? A. B. C. D. E. A. B. C. D. E. n = 3 to n = 4 n = 1 to n = 3 n = 6 to n = 4 n = 7 to n = 5 n = 2 to n = 5 9. Which of the following transitions in a hydrogen atom results in emission of light? i. ii. iii. iv. v. A. B. C. D. E. Week 2 n=3 n=1 n=6 n=7 n=2 iii only i and ii only ii and iii only iii and iv only i, ii, and v The radius of the orbit increases as the principal quantum number increases. The energy required to ionize the atom increases as the principal quantum number decreases. Light emitted by the excited hydrogen atom corresponds to transitions from orbits of higher principal quantum number to lower principal quantum number. 1 only 1and 2 only 2 and 3 only 1 and 3 only 1, 2, and 3 11. For electron distributions, which of the following statements is/are true? 1. 2. to n = 4 to n = 3 to n = 4 to n = 5 to n = 5 3. A. B. C. D. E. d orbitals have a spherical shape. p orbitals have a high electron density at the nucleus. s orbitals have no electron density at the nucleus. 1 and 2 2 only 2 and 3 3 only None of the statements is true CHEM 110 Student Packet - Spring 2013 32 WEEK 2: 16 JAN THRU 22 JAN; LECTURES 5-6 Recitation Worksheet -----------------------------------------------------------------------------------------------------------------------1. (A) What is the wavelength of one X-ray photon if the frequency is 1.00 × 1019 s−1? (B) What is the energy of one X-ray photon of this frequency? (C) What is the energy of one mole of X-ray photons of the same wavelength? -----------------------------------------------------------------------------------------------------------------------2. A sodium vapor street lamp emits yellow light at wavelength λ = 589 nm. How much energy is released if a mole of photons are emitted? A. B. C. D. E. 2.0 5.4 7.2 1.9 3.5 105 J 108 J 10–13 J 10–4 J 10–19 J HINT 1: What is the relationship needed to find the energy of a single photon? HINT 2: How many photons are in a mole of photons? -----------------------------------------------------------------------------------------------------------------------3. There is a red emission line at 670nm in the line spectrum of Li. What is the energy difference between the energy levels involved in the electronic transition that produces this emission line? A. B. C. D. 2.97 6.70 4.48 9.89 10–19 J 10–7 J 1014 J 10–28 J --------------------------------------------------------------------------------------------------------------------4A. An emission line the hydrogen atom has a wavelength of 93.8 nm. What region in the electromagnetic spectrum is this emission found? 4B. Determine the final value of n associated with this emission? (Hint: Consider E = h and the Rydberg equation, use this to find the value of nf.) 4C. Determine the initial value of n associated with this emission. (Hint: Will this value be higher or lower than nf and why is this the case? What equation will you use to determine this value; what values do you know?) --------------------------------------------------------------------------------------------------------------------- Week 2 CHEM 110 Student Packet - Spring 2013 33 WEEK 2: 16 JAN THRU 22 JAN; LECTURES 5-6 -----------------------------------------------------------------------------------------------------------------------5A. Which of the following electron transitions in a hydrogen atom will emit a photon, which absorb a photon? (How do you know?) A. n = 1 to n = 3 B. n = 4 to n = 3 C. n = 3 to n = 2 D. n = 3 to n = 1 E. n = 2 to n = 3 5B. Which of the above electron transitions in a hydrogen atom will result in emission of light with the longest wavelength? -----------------------------------------------------------------------------------------------------------------------6A. Use the Bohr model and determine the wavelength of light that would ionize a hydrogen atom. (Hint: ionization is the removal of an electron; assume we are removing it from the n = 1 value) 6B. If the electron were in an excited state (n =3) what would the ionization energy be? -----------------------------------------------------------------------------------------------------------------------7. Consider the rules for assigning quantum numbers. Which is not a permissible set of quantum numbers? Identify the orbital (if possible) a) n = 2, ℓ = 0, mℓ = 0 _____ b) n = 3, ℓ = 2, mℓ = 2 _____ _____ c) n = 2, ℓ = 1, mℓ = 1 d) n = 3, ℓ = 3, mℓ = 0 _____ _____ e) n = 4, ℓ = 3, mℓ = 3 Hint: What quantum number(s) gives the electron shell? What quantum number number(s) indicate the subshell? Which quantum number(s) define the orbitals in a subshell? ------------------------------------------------------------------------------------------------------------------------8. Which of the following represents an orbital in which a 3d electron could be found? A. B. C. D. E. 1 only 2 only 4 only 1 and 5 2 and 3 ----------------------------------------------------------------------------------------------------------------------- Week 2 CHEM 110 Student Packet - Spring 2013 34