* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Elevated serum BAFF levels in patients with sarcoidosis: association
Innate immune system wikipedia , lookup
Polyclonal B cell response wikipedia , lookup
Cancer immunotherapy wikipedia , lookup
Management of multiple sclerosis wikipedia , lookup
Autoimmune encephalitis wikipedia , lookup
Multiple sclerosis research wikipedia , lookup
Multiple sclerosis signs and symptoms wikipedia , lookup
Adoptive cell transfer wikipedia , lookup
Pathophysiology of multiple sclerosis wikipedia , lookup
Sarcoidosis wikipedia , lookup
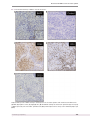
Rheumatology 2013;52:16581666 doi:10.1093/rheumatology/ket186 Advance Access publication 17 May 2013 RHEUMATOLOGY Original article Elevated serum BAFF levels in patients with sarcoidosis: association with disease activity Ikuko Ueda-Hayakawa1, Hirotsugu Tanimura2, Manabu Osawa2, Hiroshi Iwasaka2, Shuichi Ohe2, Fumikazu Yamazaki2, Kana Mizuno2 and Hiroyuki Okamoto1,2 Abstract Objective. The purpose of this study was to determine serum levels of B-cell-activating factor (BAFF) and its clinical association in patients with sarcoidosis. Methods. Serum levels of BAFF from 37 patients and 21 healthy subjects were examined by ELISA. Serum angiotensin-converting enzyme (ACE), lysozyme and IFN-g levels in sarcoidosis patients were also measured. Isolated monocytes cultured with IFN-g, IL-4 or IL-10 and their expression of membrane and soluble BAFF were analysed by flow cytometry or ELISA. Peripheral B cell subsets were analysed by flow cytometry. BAFF expression in the granuloma of the skin was examined by immunohistochemistry. ANAs were determined by indirect IF using HEp-2 cells as a substrate. CLINICAL SCIENCE Results. Serum BAFF levels were significantly elevated in sarcoidosis patients when compared with healthy controls. The frequency of skin and eye involvement was significantly higher in patients with elevated serum BAFF than in patients with normal levels. Serum BAFF levels were correlated with serum levels of ACE, lysozyme and IFN-g. Immunostaining of anti-BAFF in the skin revealed BAFF expression by epithelioid cells of granuloma. In vitro, IFN-g induced membrane-bound BAFF expression on monocytes and secretion of soluble BAFF by isolated monocytes. In the peripheral blood, sarcoidosis patients showed increased naı̈ve B cells with a reciprocal decrease in memory B cells and plasmablasts. Seventeen of 26 (65%) sarcoidosis patients exhibited ANA positivity. Conclusion. Serum BAFF levels can be used as a surrogate marker of disease activity in sarcoidosis patients. Increased BAFF may be related to the pathogenesis of sarcoidosis. Key words: sarcoidosis, B-cell-activating factor (BAFF), B cell, monocyte. Introduction Sarcoidosis is a systemic granulomatous disorder that is characterized by pathological lesions with a discrete, compact, non-caseating epithelioid cell granuloma that are commonly observed in the lungs, lymph nodes, eyes and skin [1]. Once mononuclear inflammatory cells accumulate in the target organ, macrophages aggregate and differentiate into epithelioid and multinucleated giant cells [2]. In addition to predominant involvement of cellular 1 Department of Dermatology, Kansai Medical University, Hirakata, Japan and 2Department of Dermatology, Kansai Medical University, Moriguchi, Japan. Submitted 18 July 2012; revised version accepted 4 April 2013. Correspondence to: Ikuko Ueda-Hayakawa, Department of Dermatology, Kansai Medical University, 2-5-1 Shinmachi, Hirakata, Osaka 573-1010, Japan. E-mail: [email protected] immunity in the pathogenesis of sarcoidosis, an involvement of the humoral immune system in the inflammatory process is indicated by frequently observed hypergammaglobulinaemia [3, 4], autoantibody production [5] and circulating immune complexes [6]. Moreover, the clinical and pathological features of sarcoidosis mimic those of many systemic autoimmune diseases [7]. The TNF family ligand B-cell-activating factor (BAFF) has critical roles in B cell development and function [8]. A role for BAFF and BAFF receptor (BAFFR) in the survival of B cells during B cell maturation has been shown in BAFF- and BAFFR-deficient animals [9, 10]. As BAFF promotes B cell survival, its overexpression could potentially break B cell tolerance by rescuing self-reactive B cells from deletion. Indeed, BAFF-transgenic mice develop severe autoimmune symptoms that are similar to SLE and SS characterized by hypergammaglobulinaemia, ! The Author 2013. Published by Oxford University Press on behalf of the British Society for Rheumatology. All rights reserved. For Permissions, please email: [email protected] Elevated serum BAFF levels in sarcoidosis patients high titres of anti-dsDNA antibodies and GN [11, 12]. In humans, serum levels of BAFF are high in patients with systemic autoimmune diseases such as SLE, RA, SS and SSc [1214]. In this study, we showed that serum BAFF levels were elevated in patients with sarcoidosis. High serum BAFF levels were associated with skin and eye involvement. Serum BAFF levels were also significantly correlated with the serum levels of both angiotensin-converting enzyme (ACE) and lysozyme, as well as with serum interferon (IFN)-g levels. In vitro, IFN-g enhanced membranebound BAFF expression and secretion of the soluble form of BAFF by monocytes from healthy controls, suggesting that IFN-g-induced BAFF expression by monocytes may play a role for elevated serum BAFF production. Immunohistostaining of skin biopsies revealed a local production of BAFF by epithelioid cells in the granuloma. We also showed altered B lymphocyte homeostasis characterized by an expanded frequency of naı̈ve B cells (CD19+CD27 ) and reduced frequency of both memory B cells (CD19+CD27+) and plasmablasts (CD19+/ CD27high) in sarcoidosis patients. A high frequency of ANA positivity was observed in sarcoidosis patients, though there was no correlation between BAFF levels and ANA positivity. From these observations, we suggest that elevated BAFF production may cause B cell alterations and contribute to the pathogenesis in sarcoidosis. Materials and methods Patients Serum samples were obtained from 37 Japanese sarcoidosis patients (5 males and 32 females). The age (mean ± SD) of these patients was 61 ± 12 years. Fresh venous blood samples were centrifuged shortly after clot formation and all samples were stored at 80 C before use. For comparison, serum from 21 age- and sexmatched healthy Japanese volunteers was collected and stored in the same manner. The diagnosis of sarcoidosis was established on the basis of clinical and radiological findings, supported by histological evidence in one or more organs of noncaseating epithelioid cell granulomas in the absence of organisms or particles. Patients with both sarcoidosis and SSc were excluded because BAFF is known to be elevated in CTDs such as SSc. Patients underwent a clinical assessment. Involvement of their organ systems, including skin, lung, heart, eye and muscle, was evaluated as described previously [15]. Cutaneous involvement was assessed by skin biopsy; pulmonary involvement included bilateral hilar lymphadenopathy (BHL), interstitial infiltrate or pulmonary fibrosis based on a chest radiogram and high-resolution CT; cardiac involvement included 18FDG PET and/or 67Ga scintigraphy uptake for inducible arrhythmias; eye involvement included anterior and/or posterior uveitis determined by slit-lamp and funduscopic examination. Involvement of others organs including muscle, liver, spleen, kidney and bone was examined by imaging with www.rheumatology.oxfordjournals.org 18 FDG PET and/or 67Ga scintigraphy. ACE activity was measured using a method based on colourimetry of the quinoneimine dye produced from the substrate hippuryl-Lhistidyl-L-leucine [16]. Lysozyme activity was measured using a turbidimetric method [17]. ACE and lysozyme were considered elevated when the values were >20.0 U/l and 10 mg/ml, respectively. ANAs were determined by indirect IF using HEp-2 cells as a substrate, and a titre of 1:80 or higher was considered positive (MBL, Nagoya, Japan). This study was approved by the ethics committee on human research of Kansai Medical University and informed consent was obtained from all patients. Enzyme-linked immunosorbent assay The levels of BAFF in sera and supernatant of monocytes culture were assessed by an ELISA using commercial kits for the assay of human BAFF (R&D Systems, Minneapolis, MN) according to the manufacturer’s protocol. The mean minimum detectable dose of BAFF was 3.38 pg/ml. Serum IFN-g levels were measured by a commercial ELISA kit (Human IFN-gamma Quantikine ELISA kit system, R&D Systems, sensitivity >8.0 pg/ml) according to the manufacturer’s protocol. All samples were analysed in duplicates. Absorbance was measured at 450 nm. Flow cytometric analysis Heparinized whole blood samples from both healthy donors (n = 11) and sarcoidosis patients (n = 15) were obtained for FACS analysis. Peripheral blood mononuclear cells (PBMCs) were isolated by centrifugation over Lymphoprep (Axis-Shield PoC AS, Oslo, Norway) and washed in PBS. Erythrocytes were lysed using red blood cell lysis buffer (BD Pharmingen, Franklin Lakes, NJ) before use. Cells were stained in ice-cold PBS supplemented with 5% bovine calf serum using a combination of a FITC-conjugated antibody, phycoerythrin (PE)-conjugated antibody, allophycocyanin (APC)-conjugated antibody and 7-amino-actinomycin D (7AAD) (Invitrogen Life Technologies, Carlsbad, CA). Antibodies used in this study included FITC-conjugated anti-CD19 (HIB19; eBioscience, San Diego, CA), FITC-conjugated BAFF (1D6; eBioscience), APC-conjugated anti-CD27 (LG7.F9; eBioscience), PE-conjugated CD14 (M5E2; BD Pharmingen) and APC-conjugated anti-CD3 (UCHT1; eBioscience). Dead and damaged cells were labelled with 7AAD and were eliminated from the analysis. Cells were analysed on a FACSCalibur (BD Biosciences, Franklin Lakes, NJ). Monocyte isolation and cell cultures Monocytes from healthy individuals were isolated from PBMC by negative selection employing antibodies labelled with magnetic beads according to the manufacturer’s instructions (Miltenyi Biotec, Bergisch Gladbach, Germany). For the analysis of BAFF secretion, monocytes were FACS sorted after using a negative monocyte isolation kit. Cell sorting was performed on a FACSAlia III (BD Biosciences). Monocytes were resuspended in 1659 Ikuko Ueda-Hayakawa et al. RPMI 1640 culture medium (Sigma Aldrich, St Louis, MO, USA) containing 10% fetal calf serum, 100 units/ml penicillin and 100 mg/ml streptomycin and maintained at 37 C and 5% CO2. The following recombinant human reagents were used: 5100 ng/ml IFN-g (PeproTech Inc., Rocky Hill, NJ, USA), 100 ng/ml IL-4 (PeproTech Inc.) and 100 ng/ml IL-10 (R&D Systems). Immunohistochemical staining for BAFF BAFF expression was examined by immunohistochemical staining in skin biopsies from both sarcoidosis patients with elevated serum BAFF levels (n = 3; three females) and five normal controls. Sections were stained with rat monoclonal IgG2a antibody specific for human BAFF (Buffy-1, LifeSpan Biosciences, Inc., Seattle, WA, USA), then with horseradish peroxidase-conjugated avidinbiotin complexes (Vectastatin avidin-biotin Complex Methods, Vector Laboratories, Burlingame, CA, USA). Sections were finally developed with DAB [Histofine SAB-PO (M), Nichirei Bioscience, Tokyo, Japan] and counterstained with haematoxylin. Statistical analysis Statistical analysis was performed using the Mann Whitney U test for determining the level of significance of differences between sample means. Fisher’s exact probability test for comparison of frequency and Bonferroni’s test for multiple comparisons were also used. Spearman’s rank correlation coefficient was used to examine the relationship between two continuous variables. P values < 0.05 were considered statistically significant. Results Elevated serum BAFF levels in patients with sarcoidosis Serum BAFF levels were measured in 37 sarcoidosis patients and in 21 healthy controls. Serum BAFF levels were significantly higher in patients with sarcoidosis than in healthy controls (mean ± S.D. of 1.59 ± 0.92 ng/ml vs 0.97 ± 0.21 ng/ml, P < 0.0005, Fig. 1A). Correlation of serum BAFF levels with both ACE and lysozyme levels Because serum ACE and lysozyme have been commonly used as markers of sarcoidosis activity [18, 19], we examined serum ACE and lysozyme levels in patients with sarcoidosis. We found that serum BAFF levels were significantly correlated with both serum ACE and lysozyme levels (P < 0.01 and P < 0.0001, respectively, Fig. 1B). No significant correlation between serum levels of BAFF and serum gammaglobulin levels was observed. These findings indicate that serum BAFF levels are associated with disease activity in sarcoidosis. 1660 Clinical and laboratory characteristics in patients with sarcoidosis showing normal and elevated serum BAFF levels Clinical and laboratory findings were compared between sarcoidosis patients with elevated BAFF levels and those with normal levels. BAFF values higher than the mean + 2 S.D. (1.40 ng/ml) of the control serum samples were considered to be elevated in this study. Elevated serum BAFF levels were found in 38% (14/37) of the patients (Table 1). The frequency of skin involvement was significantly higher in patients with elevated BAFF compared with those with normal levels (P < 0.0005, Table 1). In addition, patients with elevated serum BAFF levels had more frequently eye involvement than those patients with normal levels (P < 0.05). No significant difference was observed in BHL and in muscle involvement between patients with elevated BAFF levels and those without. The frequency of diffuse lung involvement and heart involvement tended to be higher in patients with elevated BAFF than those without. Serum ACE, lysozyme and gammaglobulins except IgM levels also tended to be higher in patients with elevated BAFF levels compared with patients with normal BAFF levels, although no significant difference was observed between the two groups. Serum calcium levels were similar between these two groups. Correlation between serum BAFF and serum IFN-g levels in patients with sarcoidosis Several cytokines have been reported to increase BAFF production [20, 21]. IFN-g has been known to play important roles in the formation of sarcoid lesions in active patients [22]. Thus, we examined the association between levels of serum BAFF and IFN-g. Serum IFN-g levels were significantly elevated in patients with increased BAFF levels compared with those with normal levels (Table 1), and serum levels of BAFF significantly correlated with those of IFN-g (r2 = 0.51, P < 0.0001, Fig. 1C). Thus, these findings suggest that increased IFN-g contributed to the enhanced BAFF production in sarcoidosis patients. IFN-g induced membrane-bound BAFF expression on monocytes and soluble BAFF production by monocytes As it has been reported that IFN-g induced BAFF expression on monocytes [21], we next investigated whether the presence of IFN-g, IL-4 or IL-10 affected BAFF expression on monocytes from healthy controls. These cytokines are known to have differential effects on monocytes: IFN-g is the prototype of activating cytokines, whereas IL-10 and IL-4 exhibit inhibitory effects. Monocytes were enriched by negative selection using the MACS system and were cultured at 15 105 per well in a 96-well plate for 3 days in the presence of IFN-g, IL-4 or IL-10. After 3 days of cytokine treatment, cells were stained with BAFF- and CD14-specific antibodies to determine whether these cytokines could regulate BAFF expression on monocytes. BAFF-specific immunostaining on monocytes was enhanced by treatment with IFN-g but not with either IL-4 or IL10 (supplementary Table S1, available at Rheumatology www.rheumatology.oxfordjournals.org Elevated serum BAFF levels in sarcoidosis patients FIG. 1 Serum BAFF level and its correlation with disease activity in patients with sarcoidosis. (A) Serum levels of BAFF in patients with sarcoidosis and in healthy controls. Serum levels of BAFF were determined by specific ELISA. The dashed lines indicate the cut-off value (mean + 2 S.D. of the control samples). The lines indicate the median. Serum levels of BAFF were significantly elevated in sarcoidosis patients when compared with healthy controls. (B) Correlation of serum BAFF levels with both ACE and lysozyme. Serum ACE and lysozyme levels were significantly correlated with serum BAFF levels in sarcoidosis patients: r2 = 0.20; P < 0.01 and r2 = 0.62; P < 0.0001, respectively. (C) Correlation between serum IFN-g and serum BAFF levels in sarcoidosis patients. Serum IFN-g levels were significantly correlated with serum BAFF levels in sarcoidosis patients: r2 = 0.51; P < 0.0001. Online). Next, we examined production of the soluble form of BAFF by monocytes. Monocytes from healthy controls were purified and cultured with the cytokines for 4 days, and the soluble form of BAFF in the supernatant was www.rheumatology.oxfordjournals.org quantified by ELISA. Levels of the soluble forms of BAFF were significantly elevated in supernatant of monocytes treated with IFN-g (Fig. 2A). Moreover, the numbers of monocytes treated with IFN-g were significantly increased 1661 Ikuko Ueda-Hayakawa et al. TABLE 1 Clinical and laboratory observations in patients with sarcoidosis showing elevated and normal serum BAFF levels Elevated BAFF (n = 14) FIG. 2 IFN-g mediates enhanced BAFF production and proliferation of monocytes. Normal BAFF (n = 23) Age, years 65.8 (9.6) 58.3 (12.7) Sex, n, male:female 1:13 3:20 Disease duration, years 6.1 (5.4) 4.4 (3.6) Skin, % 100*** 86 Lung, % 91 67 Radiographic stage 0/I/II/III 1/5/2/3/(3) 6/11/0/1/(5) or IV/(not determined), n BHL, % 67 70 Diffuse lung, % 17 6 Eyes, % 67* 41 Liver, % 0 0 Nervous system, % 0 0 Heart, % 33 9 Kidney, % 0 0 Bones, % 0 0 Muscles, % 17 14 Lymphadenopathy, % 67 67 ACE, IU/l 20.7 (11.6) 16.3 (7.9) Lysozyme, mg/ml 12.2 (13.2) 6.3 (2.9) Ca, mg/dl 9.3 (0.6) 9.4 (0.3) IgG, mg/dl 1465 (559) 1416 (381) IgA, mg/dl 279 (109) 228 (93) IgM, mg/dl 66 (35) 93 (44) IFN-g, pg/ml 4.9 (6.4)** 0.0 (0.1) Current treatment Oral steroids, n 7 5 MTX, n 1 0 Except where indicated otherwise, values are mean (S.D.). *P < 0.05, **P < 0.0005, ***P < 0.0005 vs sarcoidosis patients with normal BAFF levels. compared with controls, although the numbers of monocytes cultured with IL-4 or IL-10 were comparable to the control (Fig. 2B). These results suggest that IFN-g plays an important role in enhancing BAFF production by monocytes. (A) Production of the soluble form of BAFF by monocytes. Monocytes from healthy controls were purified and cultured with cytokines for 4 days, and the soluble form of BAFF in the supernatant was quantified by ELISA. The level of the soluble form of BAFF was significantly elevated in supernatant of monocytes treated with IFN-g. (B) Proliferation of monocytes by IFN-g. The number of monocytes from healthy individuals treated with IFN-g showed a significant increase compared with the control, whereas the number of monocytes cultured with IL-4 or IL-10 was comparable to the control. Immunohistochemistry of BAFF in sarcoid skin lesions To determine the role of BAFF in the pathogenesis of granuloma, we examined BAFF expression in skin biopsies from three sarcoidosis patients by immunohistochemistry. BAFF staining was observed in granuloma (Fig. 3A). The majority of BAFF-expressing cells were CD68+ epithelioid cells (Fig. 3A and B), which are thought to be derived from monocyte-macrophage lineage cells [2, 23]. On the other hand, BAFF expression was rarely seen in infiltrated lymphocytes (Fig. 3A, C and D). Alteration of B cells subsets in sarcoidosis patients Since BAFF has been known to be an important player in the survival and differentiation of B cells, we examined the frequency and numbers of B cells to determine whether 1662 there were alterations in the peripheral B cell populations in patients with sarcoidosis. Blood samples from healthy donors and patients were analysed by flow cytometry. PBMCs were stained with CD19- and CD27-specific antibodies and classified into three subsets: CD19+CD27 naı̈ve B cell, CD19+CD27+ memory B cell and CD19+/ CD27high plasmablast, as previously described [24, 25]. The frequency of total lymphocytes and absolute number of CD19+ B cells were similar between healthy donors and patients (Table 2). Compared with healthy donors, a significantly higher frequency of naı̈ve B cell (CD19+CD27 ) was observed in sarcoidosis patients (P < 0.05). In contrast, the frequency of memory B cells www.rheumatology.oxfordjournals.org Elevated serum BAFF levels in sarcoidosis patients FIG. 3 Immunohistochemistry of BAFF in sarcoid skin lesions. Representative picture of BAFF staining in skin biopsies from sarcoidosis patients with elevated serum BAFF levels. (A) BAFF, (B) negative control, (C) CD68, (D) CD3, (E) CD20. BAFF staining was observed in granuloma (A). The majority of BAFF-expressing cells were CD68+ epithelioid cells (A, C). BAFF expression was rarely seen in infiltrated lymphocytes (D, E). www.rheumatology.oxfordjournals.org 1663 Ikuko Ueda-Hayakawa et al. TABLE 2 Frequency and number of B cells in healthy donors and sarcoidosis patients Healthy donors (n = 11) Age, years Male:female CD19+ B cell Frequency, % of lymphocytes Absolute number, 104/ml B cell subset, % of total B cells CD19+CD27 (naı̈ve) CD19+CD27+ (memory) CD19+/ CD27high (plasmablast) Sarcoidosis patients (n = 15) 50.3 (13.1) 3:8 58.2 (11.3) 1:14 4.72 (2.05) 86 (41) 5.30 (3.43) 84 (65) 69.41 (7.97) 27.30 (6.98) 3.29 (1.41) 78.55 (15.69)* 19.43 (14.66)* 2.01 (1.33)* Values are mean (S.D.). B cells were gated on CD19+ cells and further distinguished by surface markers as indicated. *P < 0.05. TABLE 3 Characteristics of ANA positivity in 26 sarcoidosis patients Pt Age, years Sex ANA titre 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 60 63 62 50 47 59 42 77 59 62 68 72 63 74 75 37 73 62 60 79 46 74 75 61 70 47 F F F F F F F F F F F F F F M F F M F F F F F F F F <40 <40 <40 <40 40 40 40 40 40 80 80 80 80 80 80 80 80 80 80 80 80 160 160 640 1280 1280 ANA pattern Specific antibody Sp, H Sp, H dsDNA Sp, H Sp, H Sp, H Sp Sp, H Sp Sp Sp, H Sp Sp (80), DS (160) Sp Sp DS DS U1RNP, SSA, SSB ACA ACA Serum BAFF levels, ng/ml IFN-c, pg/ml 2.38 0.98 1.23 1.03 1.03 1.19 1.27 2.34 3.39 0 0 0 9.31 3.13 1.13 2.1 1.38 1.18 1.02 1.93 1.53 2.54 5.55 0.78 1.44 1.9 1.86 0.92 2.15 1.47 0 7.32 0.59 0 0 0 14.68 1.27 19.34 0 0 8.48 1.75 0 0 0 Sp: speckled; H: homogeneous; DS: discrete speckled. (CD19+CD27+) and plasmablasts (CD19+/ CD27high) was significantly reduced (P < 0.05 for each). ANAs in sarcoidosis patients Since overexpression of BAFF could potentially break B cell tolerance by rescuing self-reactive B cells from deletion, we performed an ANA test by indirect IF. We examined ANA in 26 sarcoidosis patients, and a titre of 1:80 or 1664 higher was considered positive (Table 3). Seventeen of the 26 patients (65%) had ANA positivity. Fifteen of these were female. Several IF patterns were observed, including fine speckled (in 14 sera), homogeneous (in 7 sera) and discrete speckled (in 3 sera). Additionally, four patients exhibited some autoantibody specificities including anti-dsDNA, anti-U1RNP, anti-SSA, anti-SSB and ACA antibodies. No correlation was found between serum BAFF levels and the presence of these autoantibodies. www.rheumatology.oxfordjournals.org Elevated serum BAFF levels in sarcoidosis patients Positive ANA titres ranged from 1:80 to 1:1280. None of the patients showed any clinical findings of other CTDs. None of the healthy controls was positive for ANA. Discussion In the current study, serum levels of BAFF were significantly elevated in patients with sarcoidosis (Fig. 1). Elevated ACE and lysozyme, which have been used as activation markers [18, 19], were significantly associated with serum BAFF levels (Fig. 2). In addition, the frequency of skin and eye involvement was significantly higher in patients with elevated BAFF levels compared with those patients with normal levels (Table 1). Close association of elevated serum BAFF with disease activity could provide a novel mechanism for the pathogenesis of sarcoidosis. There was a significant correlation between BAFF and IFN-g in the serum levels of sarcoidosis patients (Fig. 1C), suggesting a linkage between BAFF and IFN-g production. Previously we reported an increase in CD14+CD16+ monocytes coincident with high ACE levels in sarcoidosis patients [26]. Stimulation with IFN-g induced the production of the soluble form of BAFF by monocytes (Fig. 2A) with increased numbers (Fig. 2B), suggesting that IFN-g mediated BAFF production by monocytes underlie the aberrant BAFF levels in sarcoidosis patients. Examination of BAFF expression in the skin from sarcoidosis patients revealed considerable levels of BAFF in granulomas (Fig. 3). The majority of BAFF-expressing cells was CD68+ epithelioid cells (Fig. 3A and B). BAFF expression was rarely seen in infiltrated lymphocytes (Fig. 3A, C and D). BAFF production from the granulomas in the skin could contribute to the increase in serum BAFF levels. We did not find any differences, including the size of granuloma and the infiltration of B and T cells among these patients. Since BAFF is known to be important in the survival and differentiation of B cells [27], we examined the frequency and numbers of B cell subsets in the blood of patients with sarcoidosis. It revealed that there was an increase in the frequency of naı̈ve B cells (CD19+CD27 ) and reciprocal decreases in the frequency of both memory B cells (CD19+CD27+) and plasmablasts (CD19+/ CD27high) in patients with sarcoidosis (Table 2). While our paper was under review, Saussine et al. [28] published similar reports that BAFF serum levels were significantly higher in patients with chronic active sarcoidosis. Consistent with our study, naı̈ve B cells tended to be increased, whereas memory B cells were decreased. Interestingly, Saussine et al. also showed an increase in transitional B cells. Thus, increased naı̈ve B cell numbers in active sarcoidosis patients could be the result of expansion of transitional B cells, which is supported by high levels of serum BAFF. Moreover, Saussine et al. [29] showed an increase in IL10-producing regulatory B cells (Bregs) in active sarcoidosis patients and proposed anti-inflammatory roles for granuloma progression. It has been shown that aberrant expression of BAFF causes the defective selection of autoreactive B cells [30]. Increased serum and/or plasma levels of www.rheumatology.oxfordjournals.org BAFF have been documented in SLE, RA, SSc and SS [1214, 31]. In this study, we detected low levels of but significantly higher ANA positivity in our patients with sarcoidosis. Consistent with our observation, Weinberg et al. [5] reported that ANA positivity was evident in 10 out of 34 (29%) of their sarcoidosis patients. These observations could be the result of dysregulation of self-tolerance. Currently the role of autoantibody production in the pathogenesis of sarcoidosis is unclear. Further study should be required to identify the relationship between the autoantibody production and the pathogenesis of sarcoidosis. Sarcoidosis outcome cannot be predicted from the initial presentation, making it difficult to determine how to treat patients. The monoclonal antibody against BAFF (Belimumab) has completed a placebo-controlled phase III trial in SLE and has been approved by the FDA for the treatment of SLE [32, 33]. Although a precise understanding of the immune processes that initiate the disease is still unclear, we propose that BAFF might be a pathogenic factor in sarcoidosis and that targeting BAFF may represent a new therapeutic strategy for sarcoidosis patients. Rheumatology key messages Serum BAFF levels were significantly elevated in sarcoidosis patients. . Serum BAFF levels can be used as a surrogate marker of disease activity in sarcoidosis. . Increased BAFF may be related to the pathogenesis of sarcoidosis. . Funding: This work was supported by a Grant-in-Aid for Young Scientists (B) from the Ministry of Education, Culture, Sports, Science and Technology. Disclosure statement: The authors have declared no conflicts of interest. Supplementary data Supplementary data are available at Rheumatology Online. References 1 Agostini C, Adami F, Semenzato G. New pathogenetic insights into the sarcoid granuloma. Curr Opin Rheumatol 2000;12:716. 2 Newman LS, Rose CS, Maier LA. Sarcoidosis. N Engl J Med 1997;336:122434. 3 Buckley CE, Nagaya H, Sieker HO. Altered immunologic activity in sarcoidosis. Ann Intern Med 1966;64:50820. 4 Hunninghake GW, Crystal RG. Mechanisms of hypergammaglobulinemia in pulmonary sarcoidosis. Site of increased antibody production and role of T lymphocytes. J Clin Invest 1981;67:8692. 5 Weinberg I, Vasiliev L, Gotsman I. Anti-dsDNA antibodies in sarcoidosis. Semin Arthritis Rheum 2000;29:32831. 1665 Ikuko Ueda-Hayakawa et al. 6 Daniele RP, McMillan LJ, Dauber JH et al. Immune complexes in sarcoidosis: a correlation with activity and duration of disease. Chest 1978;74:2614. 21 Nardelli B, Belvedere O, Roschke V et al. Synthesis and release of B-lymphocyte stimulator from myeloid cells. Blood 2001;97:198204. 7 Pettersson T. Rheumatic features of sarcoidosis. Curr Opin Rheumatol 1997;9:627. 22 Shigehara K, Shijubo N, Ohmichi M et al. Increased circulating interleukin-12 (IL-12) p40 in pulmonary sarcoidosis. Clin Exp Immunol 2003;132:1527. 8 Batten M, Groom J, Cachero TG et al. BAFF mediates survival of peripheral immature B lymphocytes. J Exp Med 2000;192:145366. 9 Schiemann B, Gommerman JL, Vora K et al. An essential role for BAFF in the normal development of B cells through a BCMA-independent pathway. Science 2001;293: 21114. 10 Mackay F, Schneider P, Rennert P et al. BAFF AND APRIL: a tutorial on B cell survival. Annu Rev Immunol 2003;21: 23164. 11 Mackay F, Woodcock SA, Lawton P et al. Mice transgenic for BAFF develop lymphocytic disorders along with autoimmune manifestations. J Exp Med 1999;190:1697710. 12 Groom J, Kalled SL, Cutler AH et al. Association of BAFF/BLyS overexpression and altered B cell differentiation with Sjogren’s syndrome. J Clin Invest 2002;109: 5968. 13 Cheema GS, Roschke V, Hilbert DM et al. Elevated serum B lymphocyte stimulator levels in patients with systemic immune-based rheumatic diseases. Arthritis Rheum 2001; 44:13139. 14 Matsushita T, Hasegawa M, Yanaba K et al. Elevated serum BAFF levels in patients with systemic sclerosis: enhanced BAFF signaling in systemic sclerosis B lymphocytes. Arthritis Rheum 2006;54:192201. 15 Iannuzzi MC, Rybicki BA, Teirstein AS. Sarcoidosis. N Engl J Med 2007;357:215365. 16 Kasahara Y, Ashihara Y. Colorimetry of angiotensin-I converting enzyme activity in serum. Clin Chem 1981;27: 19225. 17 Ensink FTE, van Haeringen NJ. Pitfalls in the assay of lysozyme in human tear fluid. Ophthal Res 1977;9: 36673. 18 Lieberman J. Elevation of serum angiotensin-convertingenzyme (ACE) level in sarcoidosis. Am J Med 1975;59: 36572. 23 Mizuno K, Okamoto H, Horio T. Heightened ability of monocytes from sarcoidosis patients to form multinucleated giant cells in vitro by supernatants of concanavalin A-stimulated mononuclear cells. Clin Exp Immunol 2001;126:1516. 24 Chu VT, Enghard P, Schürer S et al. Systemic activation of the immune system induces aberrant BAFF and APRIL expression in B cells in patients with systemic lupus erythematosus. Arthritis Rheum 2009;60:208393. 25 Sato S, Fujimoto M, Hasegawa M et al. Altered blood B lymphocyte homeostasis in systemic sclerosis: expanded naive B cells and diminished but activated memory B cells. Arthritis Rheum 2004;50:191827. 26 Okamoto H, Mizuno K, Horio T. Circulating CD14+ CD16+ monocytes are expanded in sarcoidosis patients. J Dermatol 2003;30:5039. 27 Batten M, Groom J, Cachero TG et al. BAFF mediates survival of peripheral immature B lymphocytes. J Exp Med 2000;192:145366. 28 Meyer-Bahlburg A, Andrews SF, Yu KO et al. Characterization of a late transitional B cell population highly sensitive to BAFF-mediated homeostatic proliferation. J Exp Med 2008;205:15568. 29 Saussine A, Tazi A, Feuillet S et al. Active chronic sarcoidosis is characterized by increased transitional blood B cells, increased IL-10-producing regulatory B cells and high BAFF levels. PLoS One 2012;7:e43588. 30 Mackay F, Woodcock SA, Lawton P et al. Mice transgenic for BAFF develop lymphocytic disorders along with autoimmune manifestations. J Exp Med 1999;190: 1697710. 31 Zhang J, Roschke V, Baker KP et al. Cutting edge: a role for B lymphocyte stimulator in systemic lupus erythematosus. J Immunol 2001;166:610. 19 Pascual RS, Gee JB, Finch SC. Usefulness of serum lysozyme measurement in diagnosis and evaluation of sarcoidosis. N Engl J Med 1973;289:10746. 32 Navarra SV, Guzmán RM, Gallacher AE et al. Efficacy and safety of belimumab in patients with active systemic lupus erythematosus: a randomised, placebo-controlled, phase 3 trial. Lancet 2011;377:72131. 20 Moore PA, Belvedere O, Orr A et al. BLyS: member of the tumor necrosis factor family and B lymphocyte stimulator. Science 1999;285:2603. 33 Sanz I. Connective tissue diseases: targeting B cells in SLE: good news at last! Nat Rev Rheumatol 2011;7: 2556. 1666 www.rheumatology.oxfordjournals.org