* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download 5.3 Representative Groups PPT
Boron group wikipedia , lookup
Group 12 element wikipedia , lookup
Alkali metal wikipedia , lookup
Livermorium wikipedia , lookup
Dmitri Mendeleev wikipedia , lookup
Group 3 element wikipedia , lookup
Period 6 element wikipedia , lookup
Alkaline earth metal wikipedia , lookup
Period 5 element wikipedia , lookup
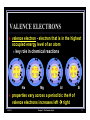
DO NOW: 2/25/14 n Take out your 5.2 HW Questions & Ch. 5 Notes n On Do Now Sheet (write answer in a complete sentence) 1. Compare how did Mendeleev organize the elements in his periodic table to how the elements are organized in the modern periodic table. 2. 2/25/14 Elements that start off each row also start off each of the 7 main ______________________. Chapter 5: The Periodic Table 1 5.3 REPRESENTATIVE GROUPS n Vocabulary: valence electron, alkali metals, alkaline earth metals, halogens, noble gases n Objectives: n Relate the number of valence electrons to groups in the periodic table and to properties of elements in those groups. n Predict the reactivity of some elements based on their locations within a group. n Identify some properties of common A group elements. 2/25/14 Chapter 5: The Periodic Table 2 2/25/14 Chapter 5: The Periodic Table 3 VALENCE ELECTRONS n n 2/25/14 2 numbering schemes A Groups n numbered 1- 8 n # matches # of valence electrons in electron configuration for element in that group Chapter 5: The Periodic Table 4 VALENCE ELECTRONS n valence electron - electron that is in the highest occupied energy level of an atom n key role in chemical reactions Na n 2/25/14 Mg Al Si properties vary across a period b/c the # of valence electrons increases left à right Chapter 5: The Periodic Table 5 VALENCE ELECTRONS n electron configuration determines an element s chemical properties & therefore elements within the same group have similar chemical properties (from 5.2) n elements in a group have similar properties because they have the same # of valence electrons 2/25/14 Chapter 5: The Periodic Table 6 Li VALENCE ELECTRONS Na properties will not be identical because valence electrons are in different energy levels K Rb Cs 2/25/14 Fr Chapter 5: The Periodic Table 7 VALENCE ELECTRONS HYDROGEN QUESTION: n 2/25/14 If hydrogen is a nonmetal gas that seems to have more in common with Group 17 nonmetals than Group 1 metals, why is it located on the left side of the periodic table with the active metals? Chapter 5: The Periodic Table 8 VALENCE ELECTRONS HYDROGEN ANSWER n hydrogen s location is related to its electron configuration & # of valence electrons, not its properties n 2/25/14 has 1 valence electron so it is grouped with other elements that have only 1 valence electron Chapter 5: The Periodic Table 9 GROUP 1A: THE ALKALI METALS 2/25/14 n 1 valence electron n properties: extremely reactive n found in nature only in compounds n example: table salt NaCl Chapter 5: The Periodic Table 10 GROUP 1A: THE ALKALI METALS lithium Least Reactive sodium the reactivity of alkali metals increases from the top of Group 1A to the bottom potassium rubidium cesium francium 2/25/14 Most Reactive Chapter 5: The Periodic Table 11 GROUP 1A: THE ALKALI METALS What properties of sodium are shown in the photos? 2/25/14 Chapter 5: The Periodic Table 12 GROUP 1A: THE ALKALI METALS n 2/25/14 Sodium n soft (unlike most metals) n melting point: 98°C n density < water n reacts with water à n hydrogen gas produced n enough energy released to ignite the hydrogen gas Chapter 5: The Periodic Table 13 GROUP 1A: THE ALKALI METALS DID YOU KNOW? n 2/25/14 Francium n most unstable of the 1st 103 elements n longest lived isotope, francium-223, has a half-life of only 22 minutes n only about one ounce of francium on Earth at any moment Chapter 5: The Periodic Table 14 GROUP 2A: THE ALKALINE EARTH METALS Beryllium No change when added to water. Least Reactive Magnesium reacts with hot water Calcium - n valence electrons: 2 n properties: metals; harder than metals in Group 1A n differences in reactivity among the alkaline earth metals are shown by the way they react with water reacts easily with cold water Strontium reacts easily with cold water Barium reacts easily with cold water Radium 2/25/14 Most Reactive Chapter 5: The Periodic Table 15 THE ALKALINE EARTH METALS: MAGNESIUM & CALCIUM (Pg 141) n magnesium & calcium have essential biological functions & provide materials used in construction & transportation n Mg: helps plants produce sugar & can be used to make light weight bicycle frames n Ca: helps build strong teeth & bones & is the main ingredient in limestone 2/25/14 Chapter 5: The Periodic Table 16 GROUP 3A: THE BORON FAMILY 2/25/14 n valence electrons: 3 n glass containing boron used in lab glassware & some cookware n aluminum is the most abundant metal in Earth s crust n often combined with O in mineral called bauxite n strong, lightweight, malleable, & good conductor Chapter 5: The Periodic Table 17 GROUP 4A THE CARBON FAMILY Carbon Least n valence electrons: 4 Metallic n properties: metallic nature of elements increases from top to bottom within group. 1 nonmetal, 2 metalloids, 2 metals n except for water, most compounds in your body contain carbon n silicon 2nd most abundant element in Earth s crust Silicon Germanium Tin Lead 2/25/14 Most Metallic Chapter 5: The Periodic Table 18 GROUP 5A: THE NITROGEN FAMILY n valence electrons: 5 n properties: Wide range of physical properties n besides nitrogen, fertilizers often contain phosphorus 2/25/14 Chapter 5: The Periodic Table 19 GROUP 6A: THE OXYGEN FAMILY 2/25/14 n valence electrons: 6 n oxygen n most abundant element in Earth s crust n ozone is form of oxygen n sulfur n 1 of 1st elements discovered n large natural deposits Chapter 5: The Periodic Table 20 GROUP 7A THE HALOGENS 2/25/14 n valence electrons: 7 n properties: highly reactive nonmetals (& metalloid); reacts easily with most metals n despite their physical differences, the halogens have similar chemical properties Chapter 5: The Periodic Table 21 GROUP 8A THE NOBLE GASES 2/25/14 n valence electrons: 8 n helium has 2 valence electrons n properties: colorless & odorless & extremely unreactive nonmetals n can be very useful n used to prevent reactions with such highly reactive elements as oxygen. Chapter 5: The Periodic Table 22 2/25/14 Chapter 5: The Periodic Table 23 2/25/14 Chapter 5: The Periodic Table 24 2/25/14 Chapter 5: The Periodic Table 25 LINKS n David s Wizzy Periodic Table - Click on Element to See Atom Structure. http://www.colorado.edu/physics/2000/applets/a2.html n Interactive Periodic Table of Elements. http://www.webelements.com/ http://www.chemicool.com/ http://web.visionlearning.com/ custom/chemistry/custom/CHE1.4-pg-table.shtml 2/25/14 Chapter 5: The Periodic Table 26 LINKS Alkali & Alkali Earth Metals http://www.youtube.com/watch?v=DFQPnHkQlZM http://www.youtube.com/watch?v=uixxJtJPVXk http://www.youtube.com/watch?v=eaChisV5uR0 http://www.youtube.com/watch?v=kyLPS3EHS8M http://www.youtube.com/watch?v=JSUhzcVnuOg http://www.youtube.com/watch?v=MGUPKA_pOEE n Comparing 4 Halogens http://www.youtube.com/watch?v=u2ogMUDBaf4 http://www.youtube.com/watch?v=m0RAE8j6sjY http://vimeo.com/55688451 n Brainiac Video http://www.youtube.com/watch?v=83eq5HyG-tI http://www.teachertube.com/viewVideo.php?video_id=41344 n 2/25/14 Chapter 5: The Periodic Table 27